113 Rheumatic Manifestations of Human Immunodeficiency Virus Infection
In the years since acquired immunodeficiency syndrome (AIDS) was initially described in 1981, the human immunodeficiency virus (HIV) pandemic has become one of the leading global health crises. According to new data in the Joint United Programme on HIV/AIDS (UNAIDS) 2008 report, the AIDS epidemic seems to be slowing down globally, but new cases are continuing to increase at alarming rates in certain regions, such as southern Africa, eastern Europe, and central and eastern Asia. An estimated 33 million people worldwide are living with HIV (Figure 113-1). Approximately 2.7 million people became newly infected with HIV in 2007, and 2 million people died.
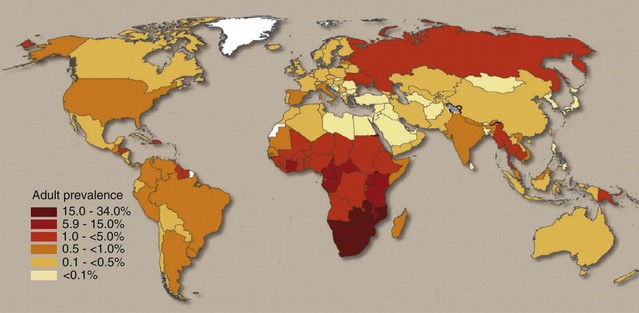
Figure 113-1 A global view of human immunodeficiency virus (HIV) infection—33 million living with HIV in 2007.
(Extracted from 2008 Report on the Global AIDS Epidemic, UNAIDS, 2008.)
Progress in dealing with the HIV epidemic, including progress in education and in public health awareness, has undoubtedly influenced the decrease in prevalence seen among young people in some countries in recent years. As availability of newer treatment strategies and better access to health care result in increased life expectancy in the next decade, it is expected that HIV infection will be managed increasingly as a chronic illness, and complications such as musculoskeletal and rheumatic conditions associated with HIV infection and its treatment are expected to increase (Tables 113-1 and 113-2).
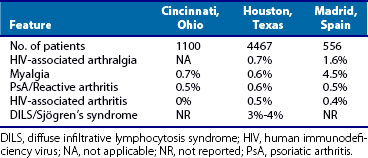
Table 113-1 Rheumatic Diseases Associated with or Occurring in Patients with Human Immunodeficiency Virus (HIV) Infection
| Unique to HIV Infection | Encountered in HIV-Infected Patients | Ameliorated by HIV Infection but Worsening or Reappearing with IRIS |
|---|---|---|
IRIS, immune reconstitution inflammatory syndrome.
HIV-Associated Bone and Joint Disease
HIV-Associated Arthralgia
More than 5% of HIV-positive patients may have otherwise unexplained arthralgia. Arthralgias and myalgias also form a part of the constitutional symptoms of HIV seroconversion. Whether arthralgia can be attributed to circulating viral and host immune complexes owing to HIV infection per se or to other infections (e.g., hepatitis C) has not been determined. The pathogenesis is unclear but may involve cytokines or transient bone ischemia.1 However, patients presenting with arthralgia alone rarely progress to inflammatory joint disease. The most appropriate treatment consists of non-narcotic analgesics and reassurance.
Painful Articular Syndrome
Painful articular syndrome is a self-limited syndrome lasting less than 24 hours, associated with few objective clinical findings, and characterized by severe bone and joint pain.2 It occurs predominantly in the late stages of HIV infection. Its cause is unknown, and no evidence of synovitis has been found in these patients. The knee is most commonly affected, but the elbow and shoulders also can be involved. Radiographic features are nonspecific; occasionally, periarticular osteopenia is seen. Treatment is symptomatic.
HIV-Associated Arthritis
The first reports of a seronegative arthritis associated with HIV infection appeared in 1988, with frequencies of 12%. HIV-associated arthritis seems to be most common in sub-Saharan Africa, where HIV infection is pandemic. In the Congo, where the seroprevalence of HIV infection is 7% to 8%, AIDS is the leading cause of aseptic arthritis (60% of cases).3 This is usually an oligoarthritis (Table 113-3), predominantly involving the lower extremities, and tends to be self-limited, lasting less than 6 weeks.2,4 Most commonly involved are the knees (84%), ankles (59%), and metatarsophalangeal joints (23%) in the lower limbs, and the wrists (41%), elbows (29%), and metacarpophalangeal and interphalangeal joints (25%) in the upper limbs, similar to other viral arthritides. Some patients have been reported as having a longer course, with joint destruction.5,6
Table 113-3 Contrasting Features of Human Immunodeficiency Virus (HIV)–Associated Arthritis and Reactive Arthritis
| Feature | HIV-Associated Arthritis | HIV-Associated Reactive Arthritis |
|---|---|---|
| Joint involvement | Asymmetric oligoarthritis/polyarthritis | Asymmetric oligoarthritis/polyarthritis |
| Mucocutaneous involvement | Absent | Present |
| Enthesopathy | Absent | Frequent |
| Synovial fluid white blood cell count | 500-2000/µL | 2000-10,000/µL |
| Synovial fluid cultures | Negative | Negative |
| Microorganisms in synovial membranes | HIV virus (?) | Chlamydia* |
| HLA-B27 association | Absent | 70%-90%† |
* Shown in non–HIV-associated reactive arthritis. Reports of such infections in patients with HIV-associated reactive arthritis are lacking.
The origin is unclear; no association has been noted with HLA-B27 or any other known genetic factor. Synovial fluid cultures are typically sterile, although one report described the presence of tubuloreticular inclusions, suggesting a viral origin, possibly HIV itself.2,4 Radiographs of affected joints are usually normal, except in uncommon cases with prolonged symptoms, in which joint space narrowing and destruction can occur. Treatment includes nonsteroidal anti-inflammatory drugs (NSAIDs) and, in more severe cases, low-dose glucocorticoids. Hydroxychloroquine and sulfasalazine also have been used.7
Reactive Arthritis Occurring in HIV Infection
Early reports in the United States suggested that reactive arthritis occurred more commonly in the setting of HIV infection; however, later studies showed that this may be reflective of the sexually active nature of the population at highest risk for HIV infection.8 This contention is not borne out in studies from sub-Saharan Africa, where HLA-B27 is rare, as were reports of spondyloarthritis before the HIV epidemic. With the arrival of AIDS, a dramatic upsurge in the prevalence of reactive arthritis and undifferentiated spondyloarthritis, and less often psoriatic arthritis,6,9 was seen, suggesting a pathogenic role of HIV infection.
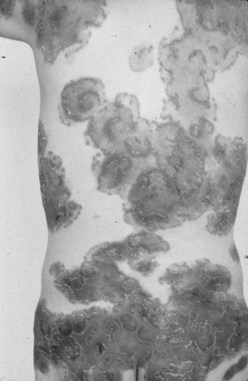
The typical presentation is a seronegative lower extremity peripheral arthritis, usually accompanied by enthesitis (dactylitis, Achilles tendinitis, and plantar fasciitis). Mucocutaneous features are common, especially keratoderma blennorrhagicum (Figure 113-2) and circinate balanitis. Extensive psoriasiform skin rashes can occur. The clinical overlap makes it difficult sometimes to distinguish HIV-associated reactive arthritis from psoriatic arthritis.10 Urethritis occurs in similar frequency as in HIV-negative reactive arthritis. Axial involvement and uveitis seem to be less common but do occur. Longitudinal studies from Africa have described an aggressive course with a poor prognosis.11,12
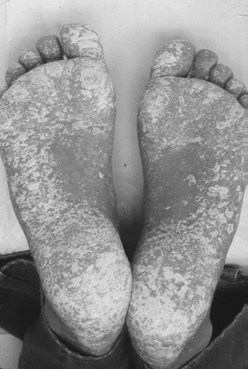
Figure 113-2 Keratoderma blennorrhagicum in a patient with reactive arthritis and human immunodeficiency virus infection.
HLA-B27 is found in 80% to 90% of patients with HIV-associated reactive arthritis, at least among whites.10 Studies from Africa have found most patients to be HLA-B27 negative, however.6,9 Some studies suggest that the presence of HLA-B27 antigen may slow the progression to AIDS.11,12 In asymptomatic HIV-infected, HLA-B27–positive individuals, cytotoxic T lymphocyte response is dominated by recognition of a gag-encoded p24 protein epitope that is not seen in HIV-positive, HLA-B27–negative individuals.13,14 Other human leukocyte antigen (HLA) class I antigens that have been associated with a better outcome in HIV infection have been implicated in psoriasis and psoriatic arthritis and include HLA-B13 and HLA-B17 (B57, B58).12,13 HLA-B*5703 was protective against HIV progression in a Zambian population but conferred susceptibility to spondyloarthritis.15
Treatment
Treatment is similar to that for HIV-negative patients with reactive arthritis. NSAIDs are the mainstay; in particular, indomethacin is recommended, not only for its efficacy, but also for its inhibition of HIV replication that has been observed in vitro, which seems to be unique to this NSAID.16 Patients frequently have an inadequate response to NSAIDs alone. Sulfasalazine has been shown to be effective in some studies at doses of 2 g/day, and one study suggested that it ameliorated HIV infection.17,18 Methotrexate was initially believed to be contraindicated because of its immunosuppressive effect, but with careful monitoring of HIV viral loads, CD4+ counts, and the patient’s clinical status, more recent studies have suggested a place for methotrexate in the treatment of reactive arthritis and psoriatic arthritis occurring in HIV infection.19
Hydroxychloroquine has been reported to be efficacious not only in treating HIV-associated reactive arthritis, but also in reducing HIV replication in vitro and in reducing HIV viral loads in vivo.20 Arthritis and the cutaneous lesions of HIV-associated reactive arthritis and psoriatic arthritis have been found to respond to etretinate (0.5 to 1 mg/kg/day),21 although because of the side effects of this drug, its use should be reserved for patients unresponsive to other treatments. Tumor necrosis factor blockers have been used,22,23 although these agents should be used with extreme caution and only in patients with CD4+ counts greater than 200/µL and HIV viral load less than 60,000 copies/mm3.24,25 One prospective study of eight HIV patients with spondyloarthritis or rheumatoid arthritis found tumor necrosis factor blockers effective and safe for up to 5 years when these precautions were followed at initiation of therapy.25
Psoriasis and Psoriatic Arthritis
The extent of skin involvement with psoriasis can be extensive (Figure 113-3) in HIV-positive patients, especially in patients not on antiretroviral treatment. Of note, cutaneous T cell lymphoma can resemble psoriasis and should be considered in the differential diagnosis of psoriasis in HIV-positive individuals.26 A report from Zambia found 27 of 28 African patients with psoriatic arthritis to be HIV positive.27 The arthritis was predominantly polyarticular, lower limb, and progressive. Psoriasis was commonly an extensive guttate-plaque admixture and, in contrast to the articular disease, was nonremittive with the onset of AIDS.28 Antiretroviral treatment has been shown to be effective in treating HIV-associated psoriasis and its associated arthritis.29 Phototherapy may improve the skin rash but also may enhance viral replication, worsen HIV disease, and increase the risk of skin cancer. Other agents reported to be efficacious include cyclosporine (although renal function must be monitored carefully) and etretinate. Methotrexate also can be used, albeit with caution.19 Tumor necrosis factor blockers can be used in patients with refractory disease, and a number of patients have shown dramatic improvement in skin lesions and in arthritis,24,25 although with the usual precautions (see earlier) because frequent polymicrobial infections while on the drug resulted in its discontinuation in some patients.30
Undifferentiated Spondyloarthritis
Symptoms of reactive arthritis or psoriatic arthritis such as enthesopathy (plantar fasciitis, Achilles tendinitis) are observed in patients who do not otherwise develop full-blown disease.31 Treatment is symptomatic (NSAIDs, intralesional corticosteroid injections), although sulfasalazine should be considered in patients with more extensive disease.
Avascular Necrosis of Bone
Most cases of osteonecrosis have occurred after the introduction of highly active antiretroviral therapy (HAART).32 Dyslipidemia associated with protease inhibitors has been implicated most frequently, although no controlled studies have been performed to establish whether antiretroviral drugs per se predispose to this.33 Other contributing factors include alcohol abuse and use of corticosteroids, megestrol acetate, antiphospholipid antibodies,34 and intravenous drug abuse,35 as well as HIV itself. The most common presenting symptom of osteonecrosis is pain on weight bearing and activity. Some patients may be asymptomatic, and the diagnosis is made based on incidental findings in radiologic studies. Most patients tend to present when subchondral collapse already has occurred. Radiographs, computed tomography (CT), magnetic resonance imaging (MRI), and nuclear medicine studies have been used successfully to diagnose osteonecrosis, as in HIV-negative patients.
Hypertrophic Pulmonary Osteoarthropathy
Hypertrophic pulmonary osteoarthropathy affects bones, joints, and soft tissues and can develop in HIV-infected patients with Pneumocystis jiroveci pneumonia. It is characterized by severe pain in the lower extremity; digital clubbing; arthralgia; nonpitting edema; and periarticular soft tissue involvement of the ankles, knees, and elbows. The skin over the affected areas is glistening, edematous, and warm. Radiography reveals extensive periosteal reaction and subperiosteal proliferative changes in the long bones of the lower extremity. A bone scan shows increased uptake along the cortical surfaces. Treatment of P. jiroveci pneumonia usually alleviates this condition.36
Osteopenia and Osteoporosis
Osteopenia and osteoporosis occur more than three times as commonly in HIV-infected patients regardless of antiretroviral treatment37 and can result in pathologic fractures. One meta-analysis found 15% of HIV-positive patients to have osteoporosis, and 52% to have osteopenia.38 Abnormal bone metabolism was attributed to the HIV infection itself by some authors.38 Risk factors for the development of osteopenia include use of protease inhibitors, longer duration of HIV infection, high viral load, high lactate levels, low bicarbonate levels, increased alkaline phosphatase levels, and lower body weight before antiretroviral therapy.39 Vitamin D deficiency is also common in HIV patients, with reported frequency of 47% for moderate to severe vitamin D deficiency in one cohort.40 A retrospective review of 211 HIV-positive patients found vitamin D deficiency to be associated with concomitant hepatitis C infection, previous AIDS, and higher CD4+ counts.41 Clinicians should have a low threshold to screen for vitamin D deficiency and should provide adequate repletion as necessary. Bisphosphonates and, in patients with HIV wasting syndrome, testosterone, have been used to preserve bone density.42
HIV-Associated Muscle Disease
Muscle involvement in HIV infection varies from uncomplicated myalgias or asymptomatic creatine kinase elevation to severe, disabling, HIV-associated polymyositis or pyomyositis (Table 113-4). HIV seroconversion also can coincide with myoglobinuria and acute myalgia, suggesting that myotropism for HIV may be present early in the infection.
Table 113-4 Myopathies Associated with Human Immunodeficiency Virus (HIV) Infection
| HIV-Associated Myopathies | Myopathies Secondary to Antiretrovirals | Others |
|---|---|---|
| HIV polymyositis | Zidovudine myopathy | Opportunistic infections involving muscle (toxoplasmosis) |
| Inclusion body myositis | Toxic mitochondrial myopathies related to other NRTIs | |
| Nemaline myopathy | Tumor infiltrations of skeletal muscle | |
| Diffuse infiltrative lymphocytosis syndrome | HIV-associated lipodystrophy syndrome | Rhabdomyolysis |
| HIV wasting syndrome | Immune reconstitution syndrome related to HAART | |
| Vasculitic processes | ||
| Myasthenia gravis and other myasthenic syndromes | ||
| Chronic fatigue and fibromyalgia |
HAART, highly active antiretroviral therapy; NRTIs, nucleoside reverse transcriptase inhibitors.
Myalgia and Fibromyalgia
One-third of HIV-positive outpatients report myalgias,43 and 11% describe fibromyalgia.44 Fibromyalgia is associated with longer disease duration and a history of depression. Treatment is similar to that for fibromyalgia in the non-HIV setting.
Noninflammatory Necrotizing Myopathy and HIV-Related Wasting Syndrome
Severe wasting from chronic infection, malignancy, malabsorption, and nutritional deficiency often accounts for weakness and disability in patients with AIDS. This wasting leads to loss of lean body and muscle mass. Cachexia and muscle wasting associated with HIV constitute slim disease in Africa. A noninflammatory necrotizing myopathy of unclear pathogenesis has been described, accounting for 42% of patients diagnosed with myopathy.45 Even in patients without significant wasting, muscle biopsy specimens have shown diffuse atrophy, mild neurogenic atrophy, or thick filament loss without conspicuous inflammation. Whether this condition is immune mediated, as some have suggested,46 or whether it is due to metabolic or nutritional factors remains unclear. Corticosteroids have been reported to restore muscle strength and mass.47
Nemaline Myopathy
Nemaline myopathy is a rare disorder that has been described in some HIV-positive patients, in addition to its occurrence as a congenital disorder. Nemaline myopathy represents a nonspecific myofibril alteration resulting from Z band disruption.48 Muscle biopsy specimens disclose prominent, randomly distributed atrophic type 1 fibers with numerous intracytoplasmic rod bodies in the centers of the fibers, corresponding to nemaline rods at electron microscopy. Necrotic fibers and inflammatory infiltrates usually are not found. Some patients have been described to have associated monoclonal gammopathy.49 Although no inflammation is noted, corticosteroids may be useful. In addition, two cases of successful treatment with intravenous immunoglobulin (IVIG) have been reported.50
HIV-Associated Polymyositis
HIV-associated polymyositis most typically manifests early in the course of HIV infection and may be the presenting feature. In one large series of HIV-positive outpatients from a county clinic in Texas, the frequency was 2.2 per 1000.51 The pathogenesis of HIV-associated polymyositis is unclear—possibly stemming from direct viral invasion (leading to a cytopathic effect and subsequent muscle necrosis), as suggested in one pathologic study,52 or from an autoimmune response of the HIV host, as suggested by another study.53
The most common manifestation is a subacute, progressive proximal muscle weakness occurring in the setting of an elevated creatine kinase. Myalgia is not a prominent presenting feature. Skin involvement is unusual, as is involvement of extraocular muscles and facial muscles. On the other hand, only a handful of cases of dermatomyositis in HIV have been reported, usually in the setting of advanced immunodeficiency.54
It has been suggested that creatine kinase levels may be less elevated or even normal in some HIV-associated polymyositis patients. One retrospective report from sub-Saharan Africa found that creatine kinase elevations were fourfold lower in those with HIV-associated polymyositis than in patients with polymyositis without HIV infection.55
On MRI, T2-weighted studies with or without fat saturation show high signal intensity without rim enhancement, in contrast to pyomyositis, in which rim enhancement is seen.56 MRI also is helpful in guiding muscle biopsy—the definitive diagnostic test. Electromyographic studies reveal myopathic motor unit potentials with early recruitment and full interference patterns and fibrillation potentials, positive sharp waves, and complex repetitive discharges indicative of an irritative process. Light microscopy of muscle biopsy specimens shows interstitial inflammatory infiltrates of variable intensity accompanied by degenerating and regenerating myofibrils, similar to those seen in polymyositis without HIV (Figure 113-4). Concomitant vasculitis rarely occurs. In specimens from HIV-positive and HIV-negative patients with myositis, the predominant cell populations were CD8+ T cells and macrophages invading or surrounding healthy muscle fibers that express major histocompatibility complex (MHC) class I antigens on their cell surfaces.57 Endomysial infiltrates in specimens from HIV-positive patients differed from those of patients with polymyositis without HIV infection only by a significant reduction of CD4+ cells.53
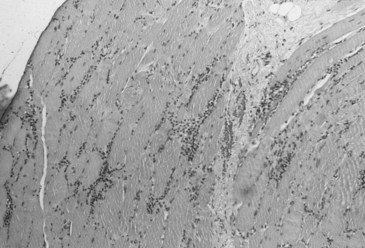
Figure 113-4 Muscle biopsy specimen from a patient with human immunodeficiency virus–associated polymyositis.
Treatment is similar to that provided for other inflammatory myopathies. Creatine kinase elevation and muscle weakness respond to moderate-dose glucocorticoids.51 Refractory cases may require immunosuppressive agents, such as methotrexate, azathioprine, or mycophenolate mofetil. Intravenous immunoglobulin has been used with some success. These agents should be used with caution, however, with careful monitoring of the patient’s clinical status, CD4+ counts, and HIV mRNA levels.
Creatine kinase elevation is commonly encountered in outpatients with HIV infection, secondary to HIV per se, behaviors associated with higher risk for HIV infection (e.g., cocaine use), or HIV treatment.51 In most patients, these elevations are transient and are of little consequence, but they require careful follow-up for any sign of clinical deterioration before electrodiagnostic and biopsy studies are undertaken.
Inclusion Body Myositis
Inclusion body myositis has been recognized as a complication of HIV infection.58 This condition is clinically, histologically, and immunologically identical to sporadic inclusion body myositis. Muscle biopsy specimens suggest two concurrently ongoing processes—an autoimmune process mediated by cytotoxic T cells and a degenerative process manifested by vacuolated muscle fibers and deposits of amyloid-related proteins. Of particular interest has been the finding of elevated mRNA levels and constitutive expression of Toll-like receptor 3, which is known to mediate inflammatory stimuli from pathogens and endogenous danger signals, and to link the innate and adaptive immune systems, in muscle fibers of patients with HIV-associated inclusion body myositis in close proximity with infiltrating mononuclear cells.59 One review of four cases of HIV-associated inclusion body myositis found that involved CD8+ cells surrounding muscle fibers were virus-specific and may cross-react with antigens on the surface of muscle fibers, suggesting that HIV may trigger a viral-specific inflammatory response that can lead to inclusion body myositis.60
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree