72 Early Synovitis and Early Undifferentiated Arthritis
What Is Early Synovitis?
Recent data suggest that early therapy and achieving low disease activity improve long-term outcomes in inflammatory arthritis.1,2 These observations have increased awareness among rheumatologists of the importance of early diagnosis and treatment. As a result, considerable pathophysiologic research and clinical trials have focused on identifying early synovitis as soon as possible. Stratifying patients into well-characterized diseases, such as rheumatoid arthritis (RA), is essential so that appropriate therapy can be initiated. Equally important, identifying patients with a high chance for spontaneous remission is needed to avoid overtreating individuals with transient undifferentiated synovitis.
The window for modifying disease outcomes might be narrow, and maximum benefit might require interventions even earlier; some evidence suggests that treatment within months might be beneficial. For this reason, several early arthritis cohorts in recent years strictly restrict the inclusion criterion of symptom duration and include only patients with a symptom onset of less than 12 weeks; this is referred to as very early synovitis. Even this definition probably does not capture the earliest phase of disease, as circulating antibodies might appear years before the onset of symptoms in RA, and biomarkers reflecting bone destruction are elevated before arthritis is present.3,4 Animal models show that histologic evidence of inflammation and activation of signaling pathways can occur well before clinically apparent disease.
Although the term synovitis generally refers to a swollen joint detected by physical examination, alternative definitions are also applied. Physical examination might not be sensitive enough, and clinically undetectable synovitis may be present and relevant to identify. Therefore, some studies consider tenderness in the absence of swollen joints as synovitis. For instance, early arthritis clinics might include patients with tenderness but no swollen joints. Others determine synovitis not by physical examination but through the use of imaging modalities such as ultrasound (US) or magnetic resonance imaging (MRI). Different ultrasound techniques are used; of these, traditional B-mode grayscale ultrasonography provides information on synovial thickness, and Doppler imaging measures the synovial blood flow. Ultrasound is easily accessible, rapid, and inexpensive. An important consideration is the reproducibility between readers. Whereas some studies found good or acceptable inter-reader agreement, other studies observed considerable variation between readers. US appears to be valuable, especially in the absence of abnormalities, and the negative predictive value of a normal US result is high. The prognostic implication of abnormal US findings is less clear.5 More long-term studies are needed to establish which US characteristics have a high positive predictive value for the development of persistent or erosive arthritis.
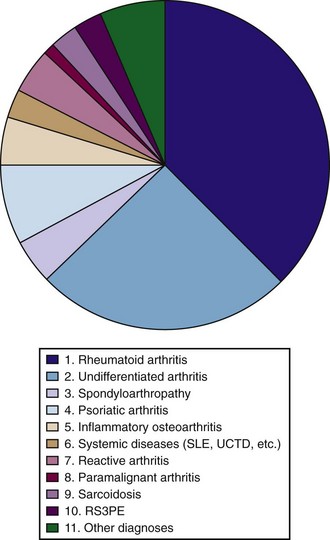
Until more data become available, the term early synovitis generally refers to synovitis that is detected by physical examination. It is important to note that early synovitis refers to a disease symptom but does not reflect any specific diagnosis. The spectrum of final diagnoses in a population of early synovitis patients is presented in Figure 72-1.
Early Arthritis Clinics
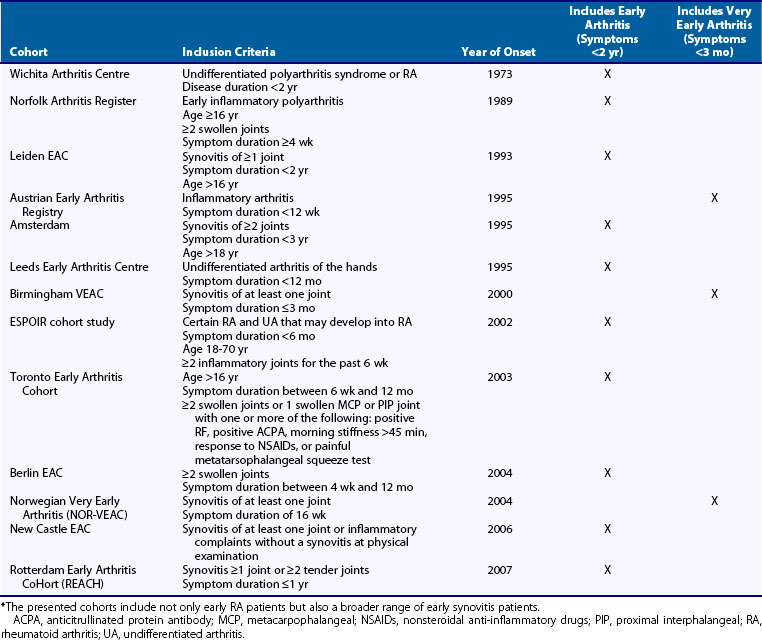
Early arthritis clinics are population-based inception cohorts in which patients with synovitis of early onset are included and followed prospectively. These cohorts often contain a wealth of phenotypic information, and the longitudinal design allows the study of factors related to a beneficial or severe course of the disease. Such studies might increase our understanding of the processes involved in arthritis progression. A summary of existing early arthritis cohorts is presented in Table 72-1. Inclusion criteria have changed over time, in parallel with changing interpretations of the term early. Whereas the early arthritis clinics started in or before the 1990s allow inclusion of patients with symptoms up to 2 years, recently started early arthritis clinics focus on very early arthritis and include patients with symptoms less than 12 weeks.
What Is Undifferentiated Arthritis?
European early arthritis cohorts observed that 35% to 54% of the patients included do not meet criteria for other diseases and are considered as UA6,7; in the remaining early synovitis patients, a definitive diagnosis could be established at first visits. The frequency of UA is dependent on symptom duration at the time of the first visit to a rheumatologist. The longer symptoms exist, the more likely that sufficient characteristics are evident to support a definitive diagnosis, and the lower the prevalence of UA will be. In addition, the prevalence of UA will change when classification criteria for rheumatologic diseases change. For example, when the 1987 American College of Rheumatology (ACR) criteria for RA were applied to new-presenting early synovitis patients in the context of an early arthritis clinic in Europe (e.g., Leiden, the Netherlands), about 20% of those patients directly fulfilled the 1987 ACR criteria and thus were classified as RA. At present, few studies evaluating the performance of the 2010 ACR/European League Against Rheumatism (EULAR) criteria are yet available. Initial data from the Leiden clinic suggest that this percentage is increased when the 2010 criteria for RA are applied.8 The new criteria for RA not only may lead to a lower prevalence of UA, they also may lead to changed patient characteristics in the UA group in that those patients who resembled RA most closely may have departed from the UA population and now are potentially classified as RA.
Characteristics of Undifferentiated Arthritis
Characteristics at first presentation between patients who present with early UA and early RA using the 1987 criteria are somewhat different. Patients with recent-onset UA are younger (mean age, 48 vs. 57 years) and are less frequently female (58% vs. 66%) than early RA patients. UA patients generally have fewer swollen joints and have a greater likelihood of asymmetric synovitis. UA and RA patients do not differ in the acuteness of the start of their complaints, body mass index (BMI), or frequency of a positive family history for RA.9 UA patients have fewer bone erosions at baseline; in a recent study, erosions were present in 18% of UA patients and 35% of RA patients.10 Important differences were also noted with regard to autoantibodies. UA patients are RF positive in only 14% and anticitrullinated protein antibody (ACPA) positive in 12% of cases, whereas both of these values are 55% in patients with early RA.5,6 According to both the 1987 and the 2010 classification criteria for RA, the presence of ACPAs in patients with arthritis is not equal to the classification of RA. Particularly in the case of monoarthritis or oligoarthritis, patients may not fulfill criteria for RA and are labeled as UA. However, early UA patients with ACPAs have a chance of about 70% of classifying under the 1987 ACR criteria 1 year later.
Remission Rates in Undifferentiated Arthritis and Rheumatoid Arthritis
The natural disease course of UA is variable, depending on the inclusion criteria and the duration of symptoms in several inception cohorts. Spontaneous remission occurs in 40% to 55% of UA patients. In contrast, the remission rate in RA is at most 10% to 15%.12,13 In these studies, remission was defined as the absence of swollen joints for 1 year or longer after discontinuation of eventual disease-modifying antirheumatic drug (DMARD) therapy and/or discontinuation of participation in the outpatient clinic because the rheumatologist classified this patient as being in remission. Longer-term follow-up is needed to determine whether symptoms recur many years later. In addition to the difference in frequency of spontaneous remission in UA and RA, the disease duration when spontaneous remission is achieved differs as well. A recent study observed that within UA, the median disease duration until spontaneous remission is 17 months, whereas in RA patients, it takes a median period of 40 months before remission is achieved.9 Thus, the chance of achieving a natural remission is reduced as the disease process matures. This supports the notion that chronicity might be more easily reversed in the undifferentiated phase of disease.
Joint Destruction in Undifferentiated Arthritis and Rheumatoid Arthritis
A recent study evaluated the subgroup of UA patients who developed RA later in time. Radiologic data were compared with those of patients who at first presentation of the disease directly fulfilled the 1987 ACR criteria for RA. Radiographic progression rates between these groups were not different. Health Assessment Questionnaire (HAQ) scores and Disease Activity Score (DAS) measures also were not different between these two groups.14 Thus, although UA patients as a group have a higher rate of spontaneous remission compared with patients with RA, the subgroup of UA patients who progress to RA have an equally severe course compared with patients classified as RA earlier in the disease. Studies that compare the rate of joint destruction in the total group of early UA patients versus that of patients with early RA have not been performed.