123 Tumors and Tumor-like Lesions of Joints and Related Structures
Non-Neoplastic Lesions
Synovial and Ganglion Cysts
Synovial cysts are common and are formed from the synovial lining of a joint, tendon, or bursa. They are non-neoplastic lesions and are caused by herniation of the synovium through the joint capsule or tendon sheath into neighboring tissues, or by expansion of a pre-existing bursa. In adults, synovial cysts frequently develop in association with a variety of joint disorders, including trauma, osteoarthritis, crystal arthropathies, infection, and rheumatoid arthritis or one of its variants. Most synovial cysts have an anatomic relationship to a joint, and most originate in the posterior aspect of the knee, where they are known as a popliteal or Baker’s cyst, followed in frequency by the shoulder and hip. These lesions may also arise in the spine, where they develop from the facet joints, most commonly in the lower lumbar region. The posteromedial region of the knee may be prone to the development of synovial cysts because the synovium-lined joint capsule in this anatomic site may not provide adequate structural support.1 Synovial cysts of the posterior knee joint are purported to affect 2.4% of children, who, in contrast to adults, are usually asymptomatic and have an otherwise normal knee joint.2
Synovial cysts can enlarge as they become increasingly distended with synovial fluid.1–4 Consequently, they may manifest as a periarticular mass, produce progressive joint pain and swelling, limit joint mobility, and compress adjacent neurovascular structures. An example of the last-mentioned item occurs in the spine, where synovial cysts that arise from facet joints may impinge or extend into spinal nerves, causing radicular pain.5–7 Other complications of synovial cysts, which sometimes can produce dramatic clinical findings, are acute rupture and secondary infection.
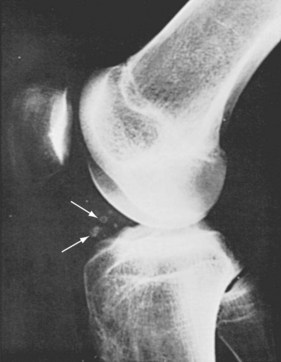
A variety of radiographic techniques have been used to image synovial cysts. Imaging modalities that provide the greatest quantity of diagnostic information include arthrography, ultrasonography, computed tomography (CT), and magnetic resonance imaging (MRI).1–4 All of these modalities reveal synovial cysts to be simple or septated thin-walled structures associated with joints and periarticular structures and filled with fluid, whose density is similar to that of water (Figure 123-1).
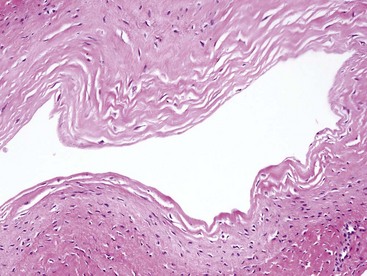
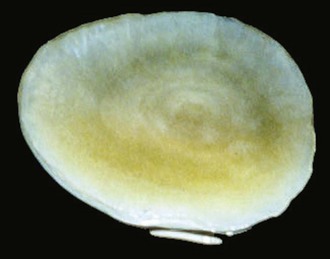
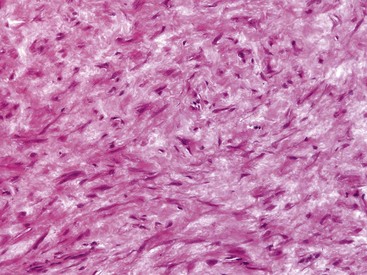
Grossly, synovial cysts usually range in size from 1 to 10 cm. Their inner surface is smooth, glistening, and translucent; however, prior hemorrhage or secondary infection may distort this surface by virtue of attached blood clot and inflammatory debris or the generation of granulation tissue. The cyst wall comprises an inner surface lined by flattened or plump cuboidal synoviocytes arranged one or several cell layers thick, which are surrounded by an outer sheath of fibrous tissue (Figure 123-2). Sometimes the synovial lining cells may be hyperplastic and form papillary fronds, and occasionally scattered subsynovial collections of hemosiderin-laden macrophages are indicative of previous hemorrhage. Facet joint cysts often contain abundant amorphous debris surrounded by macrophages and are associated with severe changes of the ligamentum flavum.
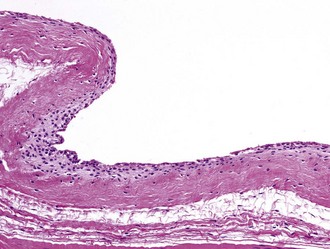
Figure 123-2 Wall of a synovial cyst, composed of an inner lining of synoviocytes overlaying a layer of dense, fibrous tissue.
Treatment of synovial cysts varies and depends on their location and associated symptoms. These cysts may be managed successfully with conservative therapy; however, in certain situations, surgical excision is required.1–7
Ganglion cysts have been recognized for centuries; Hippocrates described them as being composed of “mucoid flesh.”8 They are more common than synovial cysts and arise from tendon sheaths, ligaments, menisci, joint capsules, and bursae.8 Occasionally, they develop de novo in the subchondral areas of bone, and rarely, they arise within nerve or skeletal muscle and lack communication with a joint. Intraneural ganglia have been shown to develop from dissection of joint fluid along an articular branch of a nerve.9 Ganglion cysts are distinguished from synovial cysts by virtue of the fact that they lack a surface lining of synoviocytes. A variety of hypotheses have been proposed to explain their pathogenesis, but none have been proven.8 The most accepted theory is that ganglia develop from mucoid cystic degeneration of periarticular structures. They are commonly associated with repetitive motion activities, inflammatory arthritides, and trauma.
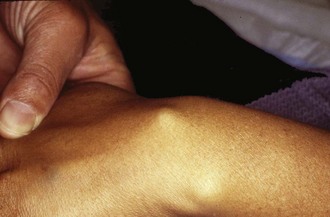
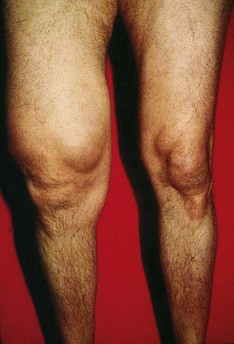
Most ganglia arise along the dorsal and volar aspects of the wrists and fingers and the dorsum of the feet.8,10 They usually are asymptomatic and typically manifest as a slowly growing, mobile, firm mass that moves with the structure from which it has arisen (Figure 123-3). Ganglia may be painful if traumatized and can compress adjacent neurovascular structures, producing a variety of symptoms. The radiographic characteristics of ganglia are similar to those of synovial cysts, and they appear on images as small, fluid-filled cystic structures.1,10
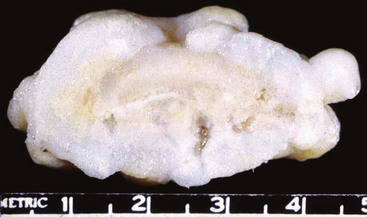
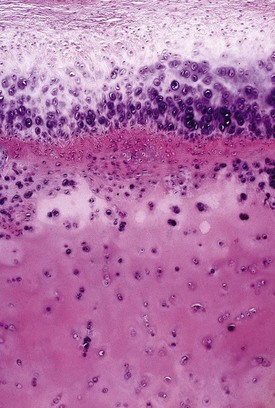
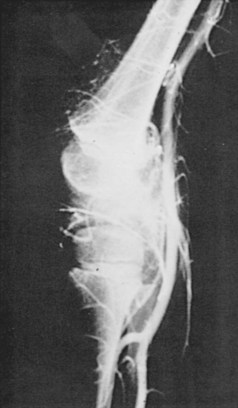
Macroscopically, most ganglia are round, but they may form elongate cylindrical structures if they track along a tendon sheath. Ganglia are uniloculated or multiloculated, have thin walls, and are filled with translucent mucoid fluid (Figure 123-4). The cyst lacks an inner cell lining, and the bulk of the wall consists of dense fibrous tissue, which is usually surrounded by areolar tissue (Figure 123-5). In many instances, the cyst wall is distorted by variable quantities of reactive myxoid tissue and muciphages, which result from small ruptures and extravasation of fluid.
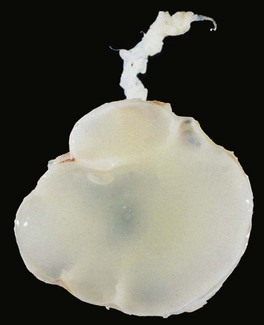
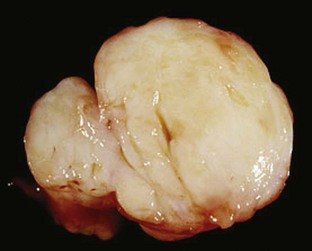
Figure 123-4 Intact ganglion cyst with thread-like pedicle that attaches to a periarticular structure.
Loose Bodies
Loose bodies and joint mice are generic terms for free-floating structures within a joint cavity. They are the most common tumor-like lesions of joints and may be exogenous, such as fragments of a bullet, or endogenous, such as pieces of articular cartilage, osteophytes, menisci, ligaments, or bone.11–13 When not otherwise specified, the term loose bodies refers to detached pieces of articular cartilage or subchondral bone (osteoarticular loose bodies) or both that lie free within the joint, or that have become secondarily embedded in the synovium. Loose bodies can cause pain, crepitance, and locking, and they can limit joint range of motion.
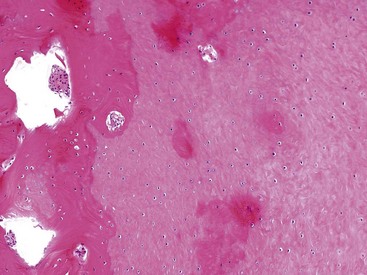
These cells produce layers of newly formed fibrocartilage and hyaline cartilage, which may undergo endochondral ossification and are deposited on the surface of the loose body (Figure 123-6). These layers of newly formed tissue surround the centrally located loose body, similar to the cambium layers of a tree, and provide a mechanism for the whole structure to increase gradually in size and become significantly larger than the initial osteochondral defect from which they originated (Figures 123-7 and 123-8). As the loose body enlarges, the innermost portion of original articular cartilage cannot be supported adequately by diffusion of synovial fluid, and it dies and calcifies. This combination of events causes the loose body to appear on x-rays as dense speckled and ring-like calcifications (Figure 123-9). Radiographically and histologically, the differential diagnosis includes synovial chondromatosis. Treatment is simple excision, which can be done arthroscopically.12,13
Intra-articular Ossicles
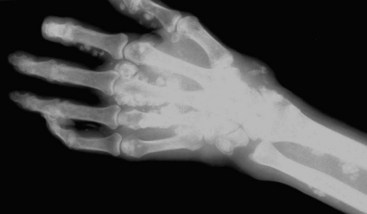
Small bony nodules normally occur in the knees of some rodents14 and other mammals and may rarely occur in humans.15–17 In rodents, they are constantly found in the anterior portions of the joint and frequently in the posterior portions as well. In humans, nodules develop within the substance of the meniscus of the knee joint adjacent to its attachments to the tibia.
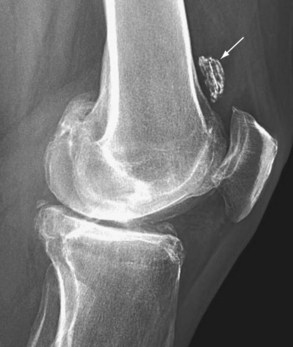
The exact origin of such structures is unknown, although they probably are true sesamoid bones, as seen in rodents, or they may represent ossification secondary to local injury. This latter possibility is supported by the fact that previous knee trauma has been noted in numerous reported cases. The main symptom of meniscal ossicles is pain after exertion, such as walking or prolonged standing, with relief when the knee is at rest. Radiographs may reveal an intra-articular calcification that can be confused with a loose body. MRI shows the ossicle to be a corticated marrow-containing structure that has increased signal intensity on T1-weighted images and decreased signal intensity on T2-weighted images.15,17 The ossicle is located within the lateral or the medial meniscus and appears as a small (≈1 cm in diameter), palpable bony nodule (Figure 123-10).16 If the ossicle is symptomatic, it should be excised; however, if it is an incidental finding, it can be managed conservatively.15,17
Neoplasms
Fatty Lesions of the Synovium
Although the subsynovial connective tissue of diarthrodial joints is rich in fat, a true lipoma of the synovium is rare. When these rare tumors develop, they most frequently affect the knee joint and the synovial sheaths of tendons of the hands, ankles, and feet, where they are more common in the extensor than in the flexor synovial sheaths.18,19 Synovial lipomas can be sessile or pedunculated, and when pedunculated, they may produce pain if they twist on their stalks and become secondarily ischemic. Synovial lipoma, similar to its subcutaneous counterpart, comprises lobules of mature white adipocytes that are delineated by a thin fibrous capsule.
A more common but still unusual fatty lesion of the joint is lipoma arborescens, also known as villous lipomatous proliferation and lipomatosis of the synovium.20,21 This disorder is characterized by a diffuse increase in the quantity of subsynovial fat, which bulges into the overlying synovial lining, producing a villous architecture. It is uncertain whether the proliferating fat is neoplastic (lipomatosis) or is a manifestation of a hyperplastic or reactive process. Affected patients are usually adults, but sometimes adolescents and rarely children develop the lesion.22 Lipoma arborescens causes chronic effusions, pain, and swelling, and restricts joint motion.21 The duration of symptoms is often long, and symptoms have been reported to be present for as long as 30 years; however, acute onset has been documented.
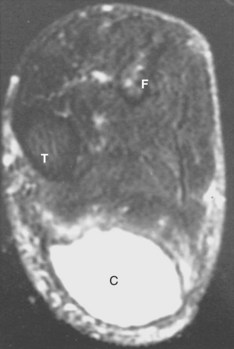
Lipoma arborescens most commonly arises in the knee (Figure 123-11), especially the suprapatellar portion, although it also has been observed in the hip, ankle, and wrist joints. It is typically localized to one joint, but several cases of bilateral knee involvement have been described.21 Laboratory studies are unremarkable, and the joint fluid is clear and yellow.21 Plain films show joint fullness, and findings of osteoarthritis are often present. Arthrography reveals multiple lobulated filling defects, which on CT represent a villonodular mass of low signal intensity that on MRI has the density of fat (Figure 123-12).23 At surgery, the affected synovium has a prominent villous or villonodular architecture and is tan-yellow (Figure 123-13). Histologically, the lesion comprises sheets of mature adipocytes admixed with nutrient blood vessels, all of which are partially compartmentalized by fibrous septa, and is covered on its intra-articular surface by several layers of synovial cells (Figure 123-14). Synovectomy may relieve the symptoms and prevent effusions, but associated osteoarthritis may be progressive.21
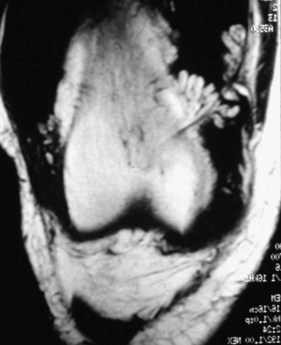
Figure 123-12 Magnetic resonance image of lipoma arborescens shows villonodular mass in the knee joint.
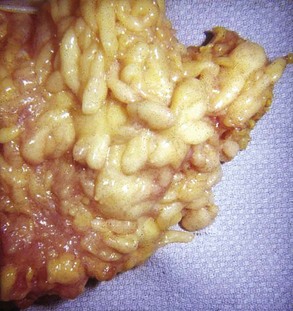
Figure 123-13 Lipoma arborescens composed of a villonodular mass of fatty tissue covered by glistening synovium.
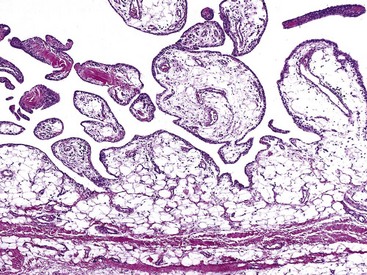
Figure 123-14 Lipoma arborescens with subsynovial compartment filled with mature adipocytes and covered by synoviocytes.
The clinicopathologic differential diagnosis includes diffuse tenosynovial giant cell tumor, synovial chondromatosis, and synovial hemangioma. These lesions can be distinguished easily from lipoma arborescens by their distinct histologic features. Another disorder that should be included in the differential diagnosis is Hoffa’s disease—a condition of irritation, inflammation, and hyperplasia of the synovial lining in regions where fat is normally present, such as adjacent to the patella or patellar ligament.24
Vascular Lesions of the Synovium
Benign vascular tumors of the synovium are rare. Their growth pattern can be localized or diffuse. They tend to predominate in adolescence and young adulthood, but symptoms frequently can be traced back to childhood.25 The joint most commonly involved is the knee, but hemangiomas also have been described in the elbow, the ankle, the tarsometatarsal and temporomandibular joints, and the tendon sheaths of the wrist and ankle.26,27 Unusual complications of synovial hemangiomas include a secondary destructive arthritis and Kasabach-Merritt syndrome.
Synovial hemangiomas produce a variety of symptoms, including unilateral, intermittent joint pain and enlargement, which may result in limitation of motion, locking, buckling, and hemarthrosis, especially after minimal trauma.26 Classically, the affected joint diminishes in size if sufficiently elevated to allow the blood to drain out of the lesion. On physical examination, the joint is swollen and doughy, and nearby cutaneous hemangiomas may be evident. Joint aspiration frequently yields bloody fluid. Preoperative diagnosis of localized hemangioma is difficult, and the differential diagnosis includes localized tenosynovial giant cell tumor, and in the knee includes discoid meniscus, meniscal tears, cysts, and ossicles.21 The diffuse hemangioma is more easily identified, but it can mimic diffuse tenosynovial giant cell tumor and hemophilic arthropathy.
Radiographic evaluation may show nothing more than a vague soft tissue shadow indicative of a swollen synovium and a distended joint capsule or regional osteoporosis in patients who have had long-term symptoms and recurrent hemarthrosis. Rarely, calcified phleboliths are apparent; however, they are associated more often with a soft tissue arteriovenous malformation with secondary joint involvement than with an isolated intra-articular hemangioma (Figure 123-15). Arthrography may show an intra-articular filling defect, and arteriography may be negative in small localized capillary hemangiomas, but contrast material may collect in the more diffuse lesions that contain cavernous or large ectatic vascular spaces (Figure 123-16). CT reveals a lobulated soft tissue mass with mild enhancement after contrast injection.21 MRI may show the tumor to have a low to isointense signal on T1-weighted images and high signal intensity on T2-weighted images.28
Macroscopically, the localized hemangioma tends to be small, but larger lesions (8 cm) have been documented.28 It may be poorly defined, well circumscribed, sessile, or stalked, and ranges in color from red to dark blue-purple. Microscopically, the hemangioma is usually of the cavernous or venous type, with large dilated blood-filled vessels lined by cytologically benign endothelial cells. In arteriovenous hemangioma, which grows in a diffuse fashion, the entire synovium may be edematous and beefy red or stained brown by hemosiderin, and consists of prominent tortuous, congested vessels that may penetrate the joint capsule and extend into neighboring soft tissues. Histologically, the vessels recapitulate architecturally abnormal arteries, veins, and capillaries; have abnormal interconnections; and are arranged in a disorganized tangle.
Fibroma of Tendon Sheath
Fibroma of tendon sheath is an uncommon benign neoplasm that clinically mimics tenosynovial giant cell tumor but is morphologically distinct. Fibroma of tendon sheath first was identified as a clinicopathologic entity in 1936, and since that time, more than several hundred cases have been reported.29,30 A translocation t(2;11) (q31-32;q12) that has been identified in this tumor is likely related to its molecular genesis.
Fibroma of tendon sheath usually arises from the tendons and sheaths of the flexor surfaces of the distal extremities; approximately 70% of cases involve the fingers or hand. Of the fingers, the thumb is affected most frequently, followed in descending order by the index and middle fingers.29 Less commonly, large diarthrodial joints such as the knee and rarely the elbow and ankle are sites of origin.29–31 Patients range in age from infants to the elderly, but the median age is the early 4th decade of life.29,30 Most series report a male predominance, with the largest study of 138 cases having a male-to-female ratio of 3 : 1.29 Patients present with a slow-growing, painless mass that usually has been noted for several months to a year.30 In 6% to 10% of cases, a history of antecedent trauma is reported. Tumors developing in large joints can be painful, can restrict range of motion, and may be palpable.32
Plain x-rays show soft tissue fullness; rarely is evidence of bony erosion noted.29 CT or MRI shows a solid, well-circumscribed mass of soft tissue density that usually has low signal intensity on T1-weighted and variable intensity on T2-weighted images. At surgery, the tumors are often attached directly to the tendon or tendon sheath. They are rubbery, oblong, well circumscribed, or encapsulated; average 1.5 to 1.8 cm in greatest dimension; and have a tan-white cut surface (Figure 123-17).29,30
Microscopically, fibroma of tendon sheath is multilobular, with clefts interposed between adjacent lobules. The lobules are composed of spindle and stellate fibroblasts enmeshed in a collagenous and sometimes myxoid stroma (Figure 123-18). Immunohistochemically, the tumor cells have the staining profile of myofibroblasts, and ultrastructurally, the cells have features of fibroblasts and myofibroblasts.33
The natural history of fibroma of tendon sheath consists of slow growth that eventually ceases. The treatment of choice is surgical excision, but a 24% recurrence rate has been reported.29
Synovial Chondromatosis
Synovial chondromatosis is an uncommon condition characterized by the formation of multiple nodules of hyaline cartilage within the subsynovial connective tissue. If the cartilage nodules undergo endochondral ossification, the term synovial osteochondromatosis is appropriate. It is unclear whether the proliferating cartilage is metaplastic or neoplastic; however, more recent cytogenetic abnormalities involving chromosome 6 found in the cartilage of these lesions support a neoplastic process.34 Regardless, synovial chondromatosis is benign and does not metastasize.
Synovial chondromatosis most commonly affects middle-aged men, with an average age in the 5th decade of life.35 Middle-aged women are more likely to develop the disease in the temporomandibular joint. The genders are equally affected with regard to hand and foot involvement, and patients with hand and foot involvement are usually in their 6th decade.
Patients commonly describe joint pain, swelling, stiffness, crepitance, and limitation of motion with a locking or grating sensation on movement.35 Symptoms usually are long-standing, recurrent, and progressive.
Synovial chondromatosis typically arises in large diarthrodial joints. The knee is affected in more than 50% of cases, usually as a monoarticular condition.35 Other common sites include the hip, elbow, shoulder, and ankle. Infrequently, synovial chondromatosis arises in the small joints of the hands and feet and in the temporomandibular joint.36 When cartilage nodules develop in the synovial lining of bursae, tendons, and ligaments, this is known as extra-articular synovial chondromatosis.32 The extra-articular variant most commonly affects the fingers, followed by the toes, hand, wrist, foot, and ankle, and more than one synovial sheath may be involved.32
Plain x-ray findings largely depend on whether the cartilage nodules are calcified or ossified, and whether they erode adjacent bony structures. Visible calcifications are absent in 5% to 33% of cases; however, most often, multiple oval intra-articular radiodensities range in size from a few millimeters to several centimeters (Figure 123-19).37 The pattern of mineralization varies and may appear as irregular flecks that represent calcified cartilage or show a trabecular architecture, which is a manifestation of endochondral ossification. Lesions that are not mineralized can be seen on an arthrogram because they produce multiple filling defects. In approximately 11% of cases, nodules erode the neighboring skeleton, especially along the anterior aspect of the distal femur.
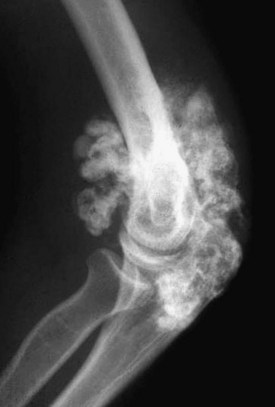
Figure 123-19 Synovial chondromatosis of the elbow. Multiple large calcified bodies fill the joint space and are adjacent to bone.
CT may show mass-like nodules in the synovium that have a density similar to skeletal muscle. CT also can detect small calcifications and erosions before they are apparent on plain films. MRI shows that the nodules of cartilage have low signal intensity on T1-weighted sequences and high intensity on T2-weighted sequences; this reflects the high water content of the hyaline cartilage.37 Areas of calcification or mineralized bone have a low signal intensity on T1-weighted and T2-weighted sequences. CT and MRI scans are helpful in identifying the intra-articular source of the lesion and its anatomic extent. In long-standing disease, the bones adjacent to involved joints may be osteoporotic and may show changes of secondary osteoarthritis.
The cartilage in extra-articular synovial chondromatosis has similar radiographic changes.37 Nodules of cartilage are frequently mineralized and may appear as a linear arrangement of small calcific densities that are aligned along the sheath and that can span many joints (Figure 123-20).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree