16 Mast Cells
Basic Biology of Mast Cells
Development and Tissue Distribution
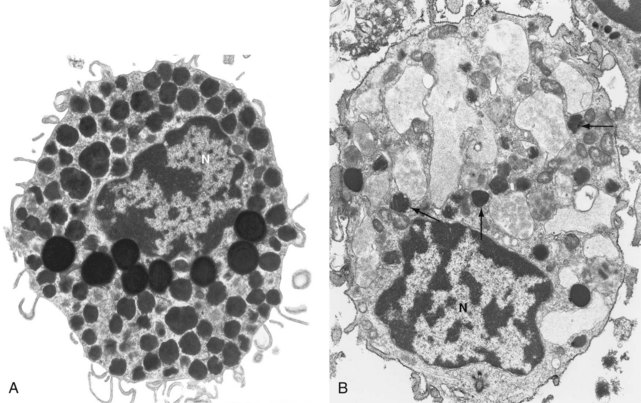
Mast cells are distinctive in appearance. Ranging in size from 10 to 60 µM and with a centrally located round or oval nucleus, their abundant cytoplasm is filled with multiple small granules. They were named Mastzellen in 1878 by the German pathologist Paul Ehrlich, who believed incorrectly that they were overfed connective tissue cells (mästen, German, “to feed or fatten an animal”).1 Electron microscopy reveals that the plasma membrane of mast cells exhibits multiple thin cytoplasmic extensions, providing a broad interface with surrounding tissue (Figure 16-1A). The tissue distribution of mast cells is extensive; within tissue, mast cells tend to cluster around blood vessels and nerves, and near epithelial and mucosal surfaces. They are also found in the lining of vulnerable body cavities such as the peritoneum and the diarthrodial joint. Given this localization, mast cells are among the first immune cells to encounter pathogens invading into tissue from the external world or via the bloodstream, consistent with their role as immune sentinel cells.2
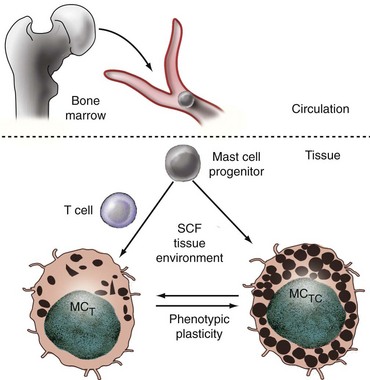
Mast cells are of hematopoietic origin, arising in the bone marrow and depositing in tissues after migrating through the bloodstream3,4 (Figure 16-2). Unlike most other myeloid cells, such as monocytes and neutrophils, mast cells do not terminally differentiate in the bone marrow but rather circulate as committed progenitors, bearing the surface signature CD34+/c-kit+/CD13+.5 Further developmental details have been worked out most extensively in the mouse. Upon entering the tissues, murine mast cells may mature into classic granulated cells or may remain as ungranulated progenitors, awaiting local signals to differentiate fully. Comparison of murine lung and intestine has demonstrated that these tissues use distinct pathways to regulate the constitutive and inducible recruitment of mast cell progenitors, illustrating that mast cell homing is a precisely controlled process.6 Tissue homing is modulated prominently by lymphocytes, including regulatory T cells (Tregs).7
Once resident in tissues, mast cells may live for many months.8 Unlike other myeloid lineage cells such as macrophages and neutrophils, mature mast cells remain capable of mitotic division, although recruitment of circulating progenitors appears to greatly exceed local replication as a pathway to expand the number of mast cells in a tissue.9 Mechanisms of reducing mast cell numbers include apoptosis, demonstrated in tissue mast cells deprived of the cytokine stem cell factor, a critical survival signal for mast cells.10,11 Under certain conditions, mast cells may emigrate via the lymphatics, appearing in draining lymph nodes much in the manner of dendritic cells.12
Mast Cell Heterogeneity: Common Progenitor, Multiple Subsets, and Phenotypic Plasticity
Although all types of mast cells derive from a common progenitor lineage, the phenotype of fully differentiated tissue mast cells is heterogeneous. Human mast cells are conventionally divided into two broad classes based on the protease content of their granules (see Figure 16-2).13 MCTC display rounded granules containing the enzymes tryptase and chymase; the smaller and more irregularly shaped granules of MCT contain tryptase but not chymase.14 MCTC also express other proteases, including carboxypeptidase and cathepsin G. MCC cells bearing only chymase have been reported but are controversial. These subtypes differ in tissue distribution. MCTC tend to be found in connective tissue, such as normal skin, muscle, intestinal submucosa, and synovium; MCT predominate in mucosal sites, including the lining of the gut and respiratory tract, although in fact both are present in many locations.15,16 Beyond protease signature, other differences between these subsets include their profiles of cytokine elaboration and cell surface receptor expression; however, tissue-specific phenotypic differences are noted within each type.
The relationship between MCTC and MCT mast cells is controversial. Are they committed subsets, akin to CD4 and CD8 lymphocytes, or functional states that mast cells assume under the influence of the microenvironment? In the mouse, where an analogous distinction exists between connective tissue mast cells (CTMCs) and mucosal mast cells (MMCs), evidence for phenotypic plasticity is strong. Both in culture and in vivo, single CTMCs may differentiate into (or give rise to) MMCs and vice versa.17,18 Mast cells with intermediate protease expression are found, and serial observations suggest that exposure to an inflammatory stimulus can induce progressive change from one class to another, although whether this occurs at a single-cell level has not been definitively established.19 Similarly, in murine and human mastocytosis, clonally expanded mast cells display divergent phenotypes depending on the tissue of residence.20,21 In aggregate, these data favor the hypothesis that mast cells assume a particular phenotype under the control of local signals but can change radically if conditions change.
Stem Cell Factor
One of the most important signals from tissue to local mast cells is stem cell factor (SCF).10 The receptor for SCF, c-kit, is expressed widely on hematopoietic lineages early in differentiation, but among mature lineages, mast cells are one of the few cell types that express c-kit at a high level. Stimulation of mast cells by SCF promotes maturation and phenotypic differentiation, blocks apoptosis, and induces chemotaxis. It may also activate mast cells directly to release mediators. In both mouse and humans, SCF remains an irreplaceable survival signal for tissue mast cells. Accordingly, mice with defects in SCF or c-kit are strikingly deficient in mature tissue mast cells (examples include W/Wv, Sl/Sld, and Wsash strains). Similarly, clonal mast cells obtained from patients with systemic mastocytosis commonly exhibit activating mutations in c-kit.22
SCF occurs in two alternate forms resulting from differential mRNA splicing: soluble and membrane bound.10 The importance of this latter form is clear from Sl/Sld mice, which lack only the membrane-bound isoform yet exhibit very few tissue mast cells.23 SCF is synthesized by multiple lineages, including mast cells themselves. Expression by fibroblasts is likely especially important, given the intimate physical contacts observed between fibroblasts and mast cells in situ. Rodent mast cells co-cultured with fibroblasts demonstrate enhanced survival, connective tissue phenotypic differentiation, and heightened capacity to elaborate proinflammatory eicosanoids—effects mediated at least in part by direct contact, including interactions between SCF and c-kit.24,25 The extent of similar regulation in human mast cells is uncertain.26 Expression of SCF has also been documented on other lineages, including macrophages, vascular endothelium, and airway epithelium, and is likely a critical pathway by which tissues modulate the local mast cell population.
T Lymphocytes and Other Cells
It is interesting to note that T lymphocytes exert a profound effect on mast cell phenotype. SCID mice lacking T cells fail to develop mucosal mast cells, a defect that may be corrected by T cell engraftment.27 An analogous observation has been made in humans deficient in T cells as the result of congenital immunodeficiency or acquired immunodeficiency syndrome (AIDS). Intestinal biopsy in these patients shows that mucosal mast cells (MCT) are strikingly reduced, but connective tissue (MCTC) mast cells are present in normal numbers.28 The pathways by which T cells exert this striking effect are not defined, although it is clear that T cell cytokines such as interleukin (IL)-3, IL-4, IL-6, IL-9, and transforming growth factor (TGF)-β may have profound effects on the phenotype of mast cells matured in culture.29–31 By contrast, interferon (IFN)-γ inhibits mast cell proliferation and may induce apoptosis. These observations imply that cells recruited to an inflamed tissue may profoundly impact the phenotype of local mast cells. The rheumatoid synovium may well exemplify this phenomenon: Normally populated by MCTC mast cells, large numbers of MCT are identified in the inflamed synovium, typically in regions rich in infiltrating leukocytes, while MCTC reside in deeper, more fibrotic areas of the joint.32 It is interesting to note that Treg cells can also directly impact mast cell function, including receptor expression and degranulation.33,34
Other cells beyond T cells may potentially interact with mast cells in the tissues. In particular, fibroblasts and mast cells commonly demonstrate close physical interactions.35 Beyond SCF, fibroblasts elaborate cytokines such as the IL-1 family member IL-33, which can exert determinative effects on mast cell protease expression and effector phenotype.36,37
Different Functions for MCT and MCTC Mast Cells?
The preservation of distinct types of mast cells in multiple species implies distinct and nonoverlapping roles for these subtypes. However, our understanding of functional differences between MCT and MCTC remains limited. One hypothesis is that MCT play a proinflammatory role and MCTC specialize in matrix remodeling.38 This hypothesis makes sense of (1) the promotion of MCT development by T cells patrolling the tissues; (2) the partitioning of MCT and MCTC mast cells to inflamed and fibrotic areas respectively; and (3) the preferential expression of the proinflammatory mediators IL-5 and IL-6 by MCT and the profibrotic IL-4 by MCTC.39 Not all observations fit comfortably into this dichotomy, however. For example, the potently proinflammatory anaphylatoxin receptor C5aR (CD88) is expressed on MCTC but not on MCT.40 Ultimately, too little is known about the actual functional importance of these subsets to permit firm conclusions.
Mast Cell Activation
IgE
The canonical pathway to mast cell activation is via IgE and its receptor FcεRI. With a Ka of 1010/M, this receptor is essentially constantly saturated with IgE at typical serum concentrations.41 Such binding not only sensitizes mast cells to the target antigen but also helps to promote mast cell survival and, in some cases, cytokine production.42,43 Cross-linking of FcεRI-bound IgE by multivalent antigen induces a brisk and vigorous response. Within minutes, granules within the mast cell fuse together and with the surface membrane create a set of labyrinthine channels that allow rapid release of granule contents (see Figure 16-1B).44 This compound exocytosis event, termed anaphylactic degranulation, is followed within minutes by the elaboration of eicosanoids newly synthesized from arachidonic acid cleaved from internal membrane lipids. Finally, signals transduced via FcεRI induce the transcription of new genes and the elaboration of a wide range of chemokines and cytokines (Figure 16-3). Upon termination of the stimulation event, the surface membrane closes over the granule-formed channels; these subsequently bud off within the cytoplasm, re-creating discrete granules using the original membranes.44 These granules become recharged with mediators through a process that occurs gradually over days to weeks.45
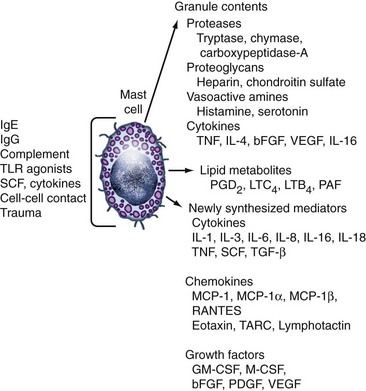
Figure 16-3 Mediator production by human mast cells (partial list). The set of mediators liberated upon activation will vary depending on the state of differentiation of the mast cell and the nature of the stimulus. See Reference 96 for a complete mediator list and references. bFGF, basic fibroblast growth factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; Ig, immunoglobulin; IL, interleukin; LTB4, leukotriene B4; LTC4, leukotriene C4; MCP-1, monocyte chemoattractant protein-1; M-CSF, macrophage colony-stimulating factor; PAF, platelet-activating factor; PDGF, platelet-derived growth factor; PGD2, prostaglandin D2; RANTES, released upon activation, normal T cell expressed and secreted; SCF, stem cell factor; TARC, thymus and activation-regulated chemokine; TGF-β, transforming growth factor-β; TLR, Toll-like receptor; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.
IgG and Immune Complexes
IgE is only one among many pathways of mast cell activation. One key trigger for mast cell activation in both human and mouse is IgG, acting via receptors for the Fc portion of IgG (FcγR). The importance of this pathway was demonstrated first in mice rendered genetically deficient in IgE. Contrary to expectations, these animals remained susceptible to anaphylaxis mediated through IgG and the low-affinity IgG receptor FcγRIII.46,47 The human counterpart of this receptor, FcγRIIa, is equally capable of inducing activation of human mast cells.48 Human mast cells exposed to IFN-γ may also be induced to express the high-affinity IgG receptor FcγRI, rendering them susceptible to IgG-mediated activation, although expression of this receptor in vivo has not been shown.49
These IgG receptors contribute to involvement of mast cells in IgG-driven diseases. Thus, in the mouse, mast cells participate in IgG-mediated immune complex peritonitis, the cutaneous Arthus reaction, and experimental murine bullous pemphigoid.50–52 Activation via Fc receptors also mediates mast cell participation in antibody-mediated murine arthritis.53,54
Soluble Mediators and Cell-Cell Contact
Mast cells may coordinate with immune and nonimmune lineages via mechanisms beyond antibody response, including soluble mediators and surface receptors. Examples of such signals include the cytokine tumor necrosis factor (TNF) and the neurogenic peptide substance P, which can induce mast cell degranulation.55,56 Physical contact with other cells can also induce mast cell activation. For example, CD30 on lymphocytes can interact with CD30L on mast cells to induce the production of a range of chemokines.57 It is interesting to note that ligation of CD30L does not induce the release of granule contents or lipid mediators, illustrating the selectivity of response of which mast cells are capable.
Danger and Injury
Mast cells are equipped to recognize danger in the absence of guidance from other lineages via a range of pathogen receptors, including multiple Toll-like receptors (TLRs) and CD48, a surface protein recognizing the fimbrial antigen FimH.58 These receptors are implicated in the response of mast cells to pathogens.59 Mast cells may also be activated through complement, including the anaphylatoxins C3a and C5a.54,56 Finally, mast cells can respond directly to physical stimuli such as trauma, temperature, and osmotic stress.60 Together, these receptors enable mast cell involvement in a broad range of immune and nonimmune processes.
Inhibitory Signals for Mast Cells
As with other immune lineages, mast cells are subject to both negative and positive regulation. Examples of inhibitory receptors on the surface of mast cells include the IgG receptor FcγRIIb and the integrin-binding immunoglobulin superfamily member gp49b1. The importance of these receptors is demonstrated in genetically deficient animals. Mice lacking FcγRIIb demonstrate a striking propensity to activation via both IgG and IgE (which bind with low affinity to FcγRIIb as well as to FcεRI),61,62 but gp49b1-null mice are unusually susceptible to IgE-mediated anaphylaxis.63 Of note, no human orthologue of gp49b1 is known, thus the relevance of this pathway in modulating MC activity in humans remains unclear. Nevertheless, modulating the surface expression of inhibitory receptors serves as an important mechanism for regulation of the activation threshold of mast cells in tissues.64
Mast Cell Mediators
Granule Contents: Proteases, Amines, Proteoglycans, and Cytokines
Mature mast cells package a range of mediators in their granules, ready for immediate release through fusion with the surface membrane. The most abundant of these are the neutral proteases, named for their enzymatic activity at neutral pH, but vasoactive amines, proteoglycans such as heparin, and pre-formed cytokines play distinct roles in the biologic consequences of mast cell degranulation. The release of these mediators is not all or none. In addition to anaphylactic degranulation, mast cells may release only a few granules at a time in a process termed piecemeal degranulation.65 Further, mast cells can release one type of granule but not another.66 Alternately, mast cells may be induced to elaborate cytokines and chemokines with no release of granule contents, as illustrated by activation via CD30L.57 Thus, although the mast cell is well equipped to release large volumes of pre-formed mediators, it is equally capable of responses tailored to the activating stimulus.
Tryptase
Named for its enzymatic similarity to pancreatic trypsin, tryptase is the most abundant granule protein in human mast cells.67 It is an essentially specific marker for mast cells, synthesized in scant amounts by basophils but by no other lineage.68 The enzyme found in granules is the β-isomer, which is enzymatically active upon formation of a homotetramer that relies on the scaffolding function of the proteoglycan heparin.69 Mast cells also synthesize α-tryptase, a protein incapable of forming homotetramers and so enzymatically inactive. Unlike β-tryptase, the α-isomer is not stored in granules but is constitutively released into the circulation, where its function is unknown. The distinction between tryptase isomers is important for diagnostic reasons: As a marker of degranulation, systemic levels of β-tryptase serve as a marker of recent anaphylaxis.70 By contrast, α-trypsin levels reflect total body mast cell load and serve as a useful biomarker in systemic mastocytosis.71 Tryptase directly cleaves structural proteins such as fibronectin and type IV collagen and enzymatically activates stromelysin, an enzyme responsible for activating collagenase.72 Tryptase also promotes hyperplasia and activation of fibroblasts, airway smooth muscle cells, and epithelium. Cleavage of protease-activated receptors such as PAR-2 may contribute to some of these activities,73–75 although other studies have documented PAR2–independent tryptase activation of mesenchymal cells.76 In aggregate, these effects suggest an important role for tryptase in matrix remodeling. A further contribution to the inflammatory milieu is suggested by the capacity of tryptase to promote neutrophil and eosinophil recruitment and to cleave C3, C4, and C5 to generate anaphylatoxins.77–79 It is interesting to note that tryptase can potentially downregulate inflammation by cleaving IgE and IL-6.80,81
Chymase
This chymotrypsin-like neutral protease is found in the MCTC subset of human mast cells, packaged within the same granules as tryptase.14 Similar to tryptase, chymase can cleave matrix components and activate stromelysin, although it can also activate collagenase directly, suggesting a role in matrix remodeling.82 Chymase can influence cytokine function, with the capacity to cleave pro-IL-1β to generate active cytokine, as well as to inactivate proinflammatory cytokines such as IL-6 and TNF.80,83,84
Vasoactive Amines
Human mast cells are capable of synthesizing and storing the biogenic amines histamine and serotonin, implicated in vascular leak.85 Histamine, by far the more abundant, is a vasoactive amine found in both MCT and MCTC mast cells, although it is not unique to this lineage. Histamine is involved in the wheal-and-flare response to cutaneous allergen challenge via augmented vascular permeability, transendothelial vesicular transport, and neurogenic vasodilation. These effects are mediated principally via the H1 receptor. Three other histamine surface receptors, H2 through H4, are distributed widely on immune and nonimmune lineages, with effects as diverse as gastric acid secretion, Langerhans cell migration, and B cell proliferation.86
Heparin and Chondroitin Sulfate E
These large proteoglycans enable the ordered packing of mediators within human mast cell granules.87,88 Negatively charged carbohydrate side chains complex tightly with positively charged proteins, allowing very high concentrations of β-tryptase and other proteases. Heparin, produced exclusively by mast cells, facilitates the activity of tryptase by making possible proteolytic self-activation within the granule and stabilizing the active tetrameric form of this enzyme.89 Heparin also has a wide range of effects beyond the mast cell. Heparin is potently angiogenic.90 Heparin binding activates antithrombin III, providing the basis for use as an anticoagulant, while inhibiting chemokines and both classical and alternative pathways of complement activation, as well as the function of Treg cells.91,92 The physiologic role of these extracellular activities of mast cell–derived heparin is uncertain.
Pre-Formed Cytokines
Mast cells are able to store certain cytokines in their granules for rapid release. The first of these to be documented was TNF.93 In the mouse, this pool of TNF is implicated in the rapid recruitment of neutrophils to the peritoneum during peritonitis.50,94 Other cytokines that may be stored in granules include IL-4, IL-16, basic fibroblast growth factor (bFGF), and vascular endothelial growth factor (VEGF).
Newly Synthesized Mediators: Lipid Mediators, Cytokines, Chemokines, and Growth Factors
Lipid Mediators
Within minutes of activation, mast cells begin to release metabolites of membrane phospholipids. This process is rapid because the relevant enzymes, beginning with phospholipase A2, responsible for harvesting phospholipids from the outer leaflet of the nuclear membrane, are already present in the cytoplasm and need only to be activated through signals mediated by calcium flux and the phosphorylation of intracellular messengers. The hallmark prostaglandin of human mast cells is prostaglandin D2 (PGD2), which is capable of inducing bronchoconstriction, vascular leak, and neutrophil recruitment. Smaller quantities of other prostaglandins as well as thromboxane are also made. Mast cell–derived leukotrienes have similar but generally more potent activity. Leukotriene C4 (LTC4) is the major leukotriene species generated by human mast cells; together with its metabolites LTD4 and LTE4, it serves as a potent inducer of vascular leak. Smaller quantities of the chemotaxins LTB4 and platelet-activating factor (PAF) are also generated. The particular profile of lipid mediators produced by mast cells can change with local environmental signals and the resulting state of differentiation. Thus, mast cells from skin generate PGD2 in excess of LTC4, and both species are elaborated in roughly equal proportions by mast cells isolated from lung and osteoarthritic synovium.95
Cytokines, Chemokines, and Growth Factors
Within hours of activation, mast cells begin to elaborate newly synthesized mediators as the end result of induced gene transcription and translation. The range of such mediators is broad (see Figure 16-3). They include the canonical proinflammatory mediators TNF, IL-1, and IL-6; the Th2 cytokines IL-4, IL-5, IL-10, and IL-13; chemotactic factors including IL-8, MIP-1α, and regulation upon activation normal T cell expressed and secreted (RANTES); and growth factors for fibroblasts, blood vessels, and other cells such as bFGF, VEGF, and platelet-derived growth factor (PDGF).96 As noted earlier, some of these may also be stored pre-formed in granules for rapid release. The panel of mediators generated depends on the state of differentiation as well as the activating signal, and may occur in the absence of degranulation.
Role of Mast Cells in Health and Disease
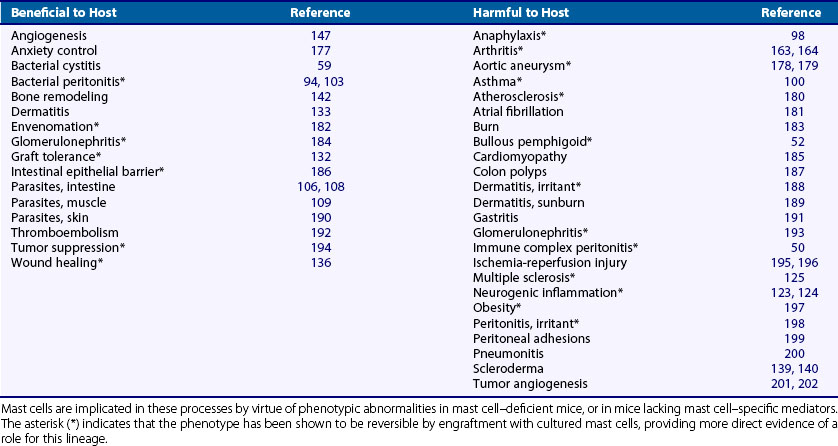
Our understanding of the role of mast cells in health and disease has been aided greatly by the availability of mice lacking mast cells through defects in the SCF/c-kit axis. Although these mice exhibit multiple phenotypic abnormalities, they are viable, excluding an obligate basal role for mast cells in the structure and function of most tissues. Yet under physiologic stress, such as imposed by experimental models of disease, multiple differences from wild-type become evident. In many cases, these abnormalities may be corrected by engraftment with cultured mast cells,97 directly implicating mast cells in a remarkably broad range of disease processes (Table 16-1). Interpretation of such experiments is limited by incomplete physiologic restoration of the mast cell compartment and by residual effects of deficient c-kit signaling in other lineages. However, together with in vitro experiments and careful observation of normal subjects, animal experiments in mast cell–deficient mice have contributed greatly to recent progress in our understanding of mast cell physiology and pathophysiology.
Mast Cells in Allergic Disease: Anaphylaxis, Allergic Disease, and Asthma
Mast cells are the primary mediator of systemic anaphylaxis. This is demonstrated in mast cell–deficient mice, in which resistance to IgE-mediated anaphylaxis may be restored by engraftment with mast cells.98 In humans, participation of mast cells in anaphylaxis has been documented through the detection of elevated serum levels of β-tryptase, a specific marker of mast cell degranulation.70 Mast cells accumulate in atopic mucosal tissues, where they degranulate upon exposure to antigen and contribute prominently to tissue edema and the overproduction of mucus.41 Mast cells also accumulate in the asthmatic airway, including within the smooth muscle lining the airways, and have been implicated by human and animal data in airway hyperreactivity and mucosal changes.99,100
Mast Cells in Nonallergic Inflammation
Pathogen Defense: Mast Cells as Sentinels of Innate Immunity
Mast cells are competent defensive cells against bacteria. They express TLRs and other receptors against bacterial antigens, and upon activation are able to phagocytose bacteria and generate antimicrobial molecules such as cathelicidin.101,102 However, given their relatively small numbers, the most important function of mast cells in immune defense is to serve as sentinels, monitoring for early traces of infection and rapidly mobilizing neutrophils and other inflammatory cells when needed. Such a role has been clearly demonstrated in mouse models of bacterial peritonitis, in which mast cell–deficient animals exhibit high mortality. This susceptibility correlates with delayed recruitment of neutrophils via TNF and leukotrienes; both neutrophil influx and survival may be restored by correction of the mast cell deficit, although in severe infection, mast cell TNF may actually contribute to mortality.94,103–105 Clearance of bacteria from the lung is delayed in mast cell–deficient mice and can be similarly restored.94 Analogous observations have been made in other models of bacterial infection.58 Thus, mast cells may play an important role in defense of the host against bacterial infection.
Mast cells are also implicated in the defense against parasites. Mast cell–deficient animals exhibit abnormal clearance of multiple parasites from gut and skin, in a manner promoted by IgE.106,107 The mechanism of this defense remains uncertain but may include direct attack upon pathogens, recruitment of inflammatory lineages such as neutrophils and eosinophils, and lysis of tight junctions in the mucosal lining to facilitate the expulsion of helminths.106,108,109
Mast Cells and the Adaptive Immune Response
In addition to recruiting innate effector cells, mast cells mobilize T and B lymphocytes, the adaptive arm of the immune system.96 Mast cells may express MHC II, as well as co-stimulatory molecules such as CD80 and CD86, rendering them effective antigen-presenting cells for CD4 T cells. Mast cells can also mobilize and potentiate CD8 T cell responses.110 They may migrate from peripheral tissues to lymph nodes carrying antigen and may contribute to the recruitment of T cells to lymph nodes via mediators such as MIP-1β and TNF, as well as suppression of Treg responses.12,111,112 Indeed, infection-induced lymph node hyperplasia is abrogated in the absence of mast cells. Further, mast cells can recruit CD4 and CD8 effector T cells to peripheral tissues via leukotriene B4, among other mediators.113–115 Finally, mast cells can contribute to the migration of cutaneous Langerhans cells and other dendritic cells to lymph nodes via mediators including histamine.116–118 By means of the inducible expression of CD40L and cytokines, mast cells may stimulate B cells and induce class switching to IgA or IgE.119,120 The physiologic importance of these effects will vary with circumstances. For example, under some conditions delayed-type hypersensitivity responses in skin are mast cell dependent, but under others mast cells appear to play no role.96 The potential importance of the mast cell in adaptive immunity is highlighted by the recent demonstration that mast cell activators are effective vaccine adjuvants.121
Neurogenic Inflammation
In addition to their perivascular localization, mast cells cluster near and even within peripheral nerves. A discrete function for them in these locations has not yet been identified, although the potential for bidirectional neuroimmune interaction is clear. Mast cell mediators such as histamine may directly activate neurons, and mast cells residing near stimulated neurons may be induced to degranulate.122 Indeed, vascular leak and neutrophil infiltration arising from infiltration of skin with the neurogenic mediator substance P are mediated by mast cells.123,124 Thus neurons may recruit mast cells as local effectors to initiate neurogenic inflammation.
Autoimmune Disease
Reconstitution experiments in mast cell–deficient mice have implicated mast cells in a variety of pathologic conditions (see Table 16-1). These include murine models of autoimmune diseases such as bullous pemphigoid, multiple sclerosis, scleroderma, and inflammatory arthritis. In pemphigoid, mast cells triggered via IgG antibodies against a hemidesmosomal antigen recruit neutrophils that are responsible for blister formation.52 The role of mast cells in murine experimental autoimmune encephalomyelitis (EAE) is more complex. Although the resistance of W/Wv mice to EAE corrects with mast cell engraftment, these cells fail to repopulate the brain and spinal cord, indicating that mast cells are not obligate local effector cells in this model.125,126 One mechanism for this activity appears to be promotion of the adaptive immune response, because mast cell engraftment into W/Wv animals improves T cell responses to immunization with the inciting myelin antigen.127,128 The contribution of mast cells to human scleroderma remains unknown. The participation of mast cells in arthritis is discussed in detail later.
Mast Cells as Anti-Inflammatory Cells
Within the last few years, it has become evident that mast cells may also help moderate the immune response. One mechanism for this effect is degradation of proinflammatory mediators. Mast cell proteases may cleave and inactivate the cytokines IL-5, IL-6, IL-13, and TNF, as well as endothelin-1 and the anaphylatoxin C3a.78,84,129,130 The importance of this activity has been demonstrated in a murine sepsis model, in which mast cells reduced mortality by restraining excess inflammation in a protease-dependent manner.130 More broadly, mast cells are capable of producing mediators such as IL-10 that have immunosuppressive activity; even otherwise proinflammatory mediators such as TNF and granulocyte-macrophage colony-stimulating factor (GM-CSF) can be immunosuppressive under appropriate circumstances.131 Thus, mast cells promote immunologic tolerance to skin grafts and limit tissue inflammation related to ultraviolet-light injury.81,132,133