58 Imaging Modalities in Rheumatic Diseases
Practical Use of Imaging in Inflammatory Joint Diseases
![]() Video available on the Expert Consult Premium Edition website.
Video available on the Expert Consult Premium Edition website.
The present chapter focuses on inflammatory joint diseases, such as rheumatoid arthritis (RA), psoriatic arthritis (PsA), ankylosing spondylitis (AS), other types of axial spondyloarthritis (SpA), gout, and osteoarthritis (OA). Other aspects, including nuclear medicine and capillaroscopy in connective tissue diseases and vasculitides, are also briefly discussed. The reader is kindly referred to the chapters on individual diseases for imaging aspects of other rheumatologic diseases, and to textbooks of musculoskeletal radiology1 for a more detailed description of the different imaging modalities, including the technical aspects. This chapter outlines the virtues of CR and describes its current major importance in diagnosis and follow-up of rheumatologic diseases, but also, by putting emphasis on newer imaging modalities, particularly MRI and US, stresses that rheumatology has entered a time of exciting and expanding therapeutic and imaging possibilities.
Conventional Radiography
Key Points
X-ray is relatively inexpensive, easily available, and reliable.
The main disadvantage of x-ray is its low sensitivity, particularly for soft tissue changes.
The first roentgen ray image was an “x-ray” of the hand. Since then, imaging of musculoskeletal structures has always been an important role of conventional radiography (x-rays).1 The simple radiograph is relatively cheap, is available worldwide, and produces images that are almost identical regardless of technical parameters or whether the image is analogue or digital. The reliability of the image means that, despite the limitations of radiography, advances in clinical practice can rely on the good old x-ray, with the knowledge that it is largely independent of technologic advances. For many years to come, radiographs will appear much the same as they do now.
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is the archetypal inflammatory joint disease that primarily targets synovium and peripheral joints. It is a systemic inflammatory disorder in which the typical clinical manifestations are usually symmetric, and the radiographic signs usually follow this pattern. As in all of radiology, the observed distribution of disease is often characteristic of the underlying cause; in RA, the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints of the hands, the wrists, and the metatarsophalangeal (MTP) joints of the feet are most often involved.1,2
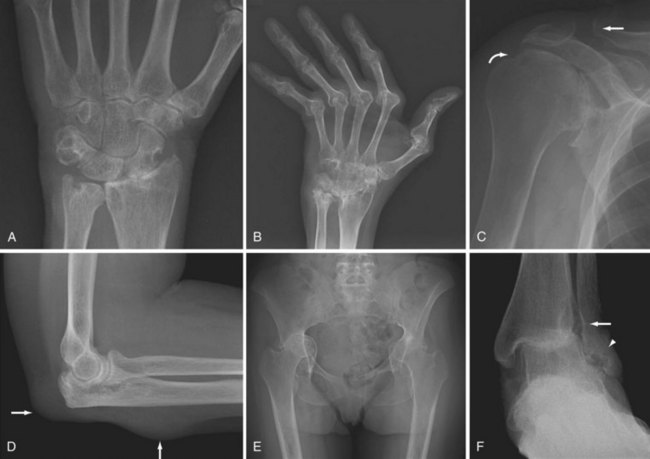
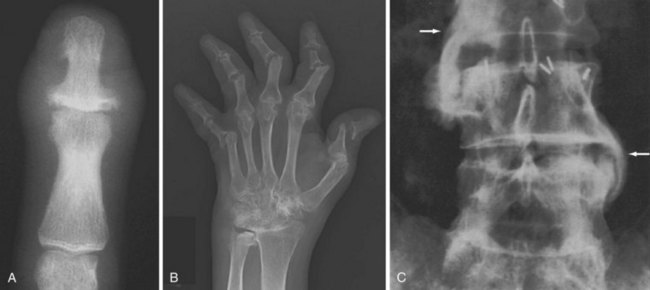
Juxta-articular osteoporosis is a characteristic feature of RA best seen in early disease in small peripheral joints. Later, generalized osteoporosis is usually present and is exacerbated by disuse. Erosion of bone (Figure 58-1) is a characteristic feature of RA and usually appears first at the margins of the joint, where the proliferating synovium lies directly on the surface of the bone between the edge of the cartilage-covered articular surface and the capsular attachment—the “bare areas.” These marginal erosions may be subtle and first appear as disruption of the thin white cortical line, especially at the radial aspect of the metacarpal heads, where they may be seen best on dedicated radiographic projections such as the ball-catcher’s view of the hands. These erosive changes are a good indication of the aggressiveness of the arthritis. In large joints, synovial proliferation may be very severe before bony erosion is detectable on x-ray. This is especially notable in the knee, where pain and swelling due to arthritis and bursitis occur long before erosion. In addition to bare-area erosions, two other types of erosions have been described in RA. Compressive erosion refers to remodeling of inflamed and osteoporotic bone with gradual invagination of one bone into another, typified by protrusio acetabuli of the hip (Figure 58-1E). Surface erosion of bone may also be seen, usually resulting from inflammation of an adjacent tendon sheath. A typical location for this is at the outer margin of the ulnar styloid process secondary to extensor carpi ulnaris tenosynovitis.
Joint malalignment and deformity (see Figure 58-1) are very common in RA and occur as the result of laxity and disruption of capsule, ligaments, and tendons. These deformities are most characteristic in the hand, wrist, foot, and neck. Note that these malalignments may be transient and can reduce during positioning for radiographs. Late disease is associated with severe deformity, as is seen in arthritis mutilans.
Spine
Cervical spine involvement is common in RA and merits specific attention. It occurs mainly after several years of disease in rheumatoid factor–positive patients with severe peripheral RA and may cause severe pain, instability, and, ultimately, spinal cord compression.3 Prevalences of radiologic cervical involvement up to 70% have been reported but are much lower in recent studies in accordance with the fact that intensive disease-modifying antirheumatic drug (DMARD) therapy has been shown to reduce cervical radiologic progression.4
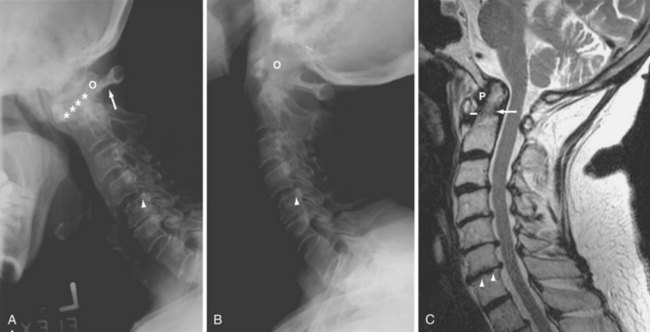
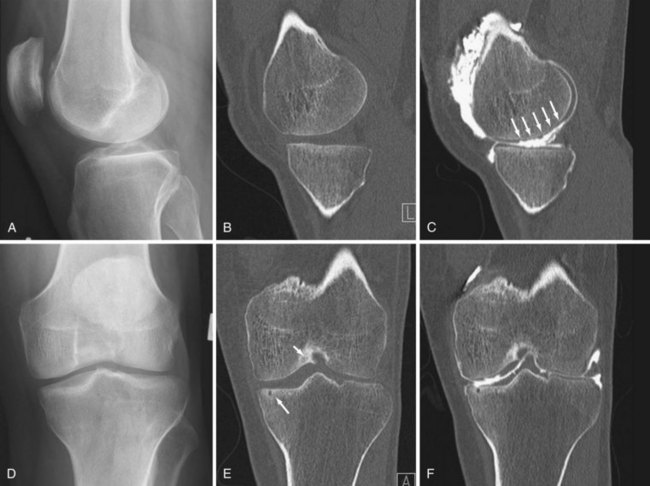
The upper cervical region is most affected, although abnormalities throughout the cervical spine are frequent (Figure 58-2). Upper cervical changes include odontoid process (dens) erosion and atlantoaxial subluxations (anterior, vertical [known as cranial settling], lateral, and posterior); subaxial manifestations include abnormalities of apophyseal and discovertebral joints, subluxation, and dislocation.
Atlantoaxial subluxation is caused by laxity or rupture of the transverse ligament. The characteristic finding is anterior subluxation (i.e., abnormal separation between the anterior arch of the atlas and the odontoid process of the axis). Reports of the exact lower limit of abnormal distance vary, but a distance greater than 2.5 mm is considered abnormal, as is distance that changes significantly between flexion and extension.1 Cranial settling is diagnosed by radiographic measurement of the relationship between the tip of the odontoid process and landmarks at the base of the skull.1 It may lead to protrusion of the dens through the foramen magnum and compression of neurologic structures. Significant atlantoaxial subluxations may require surgery.
Subaxial involvement is frequent and manifests as varying degrees of bone erosion, joint space narrowing, and subluxation of facet joints, as well as disk space narrowing and spinous process erosion. Subaxial subluxations may also lead to neurologic deficits.1
MRI is useful for providing supplementary information on cervical spine involvement (Figure 58-2C), including spinal cord compression (see MRI section).
Use in Diagnosis, Monitoring, and Prognostication
Diagnosis
Characteristic x-ray findings are part of the American College of Rheumatology (ACR) 1987 classification criteria for RA.5 X-ray also has a role in the recent ACR/European League Against Rheumatism (EULAR) 2010 classification criteria, in that patients who display bone erosions typical of RA plus at least one clinically swollen joint fulfill the criteria for and are classified as having RA.6 X-ray can be helpful in differentiating RA from other joint conditions such as osteoarthritis, psoriatic arthritis, and neoplasm.2
Monitoring
In routine clinical management and in clinical trials of RA, x-ray evaluation focuses on joint space narrowing and bone erosion in hands, wrists, and forefeet as measures of structural joint damage.7–9 Validated scoring methods for radiologic damage (the Larsen method and the Sharp method and their modifications) are available and are used extensively in clinical trials.8–10 The van der Heijde and Genant modifications of the Sharp score are generally considered the methods that are most sensitive to change, but they are also the most time-consuming.11 For clinical practice, the less time-consuming simple erosion narrowing score (SENS), derived by counting joints with bone erosion and joints with joint space narrowing, is available12,13 but is seldom used.
Prognostication
Early bone erosion correlates with poor long-term radiographic and functional outcomes,14 and early progression in x-ray erosion is related to future impairment in physical function.15 In early undifferentiated arthritis, the presence of x-ray erosion increases the risk of developing persistent arthritis.16 However, radiographic erosions are present in only a minority of patients with early RA, with prevalences of 8% to 40% at 6 months,17–21 and the absence of erosion in early disease is not necessarily associated with good outcomes. Therefore at this stage, x-ray is not effective for identifying future “nonprogressors” (i.e., patients who will not show increasing structural joint damage).22
Ankylosing Spondylitis
Sacroiliac Joints
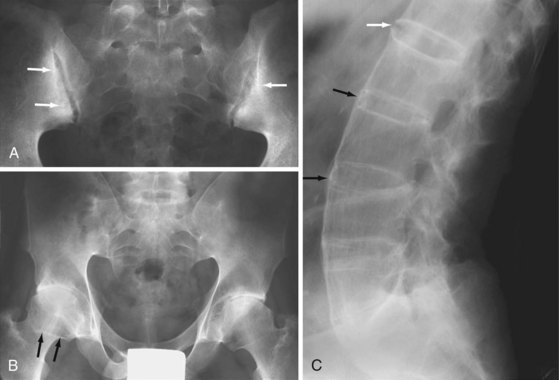
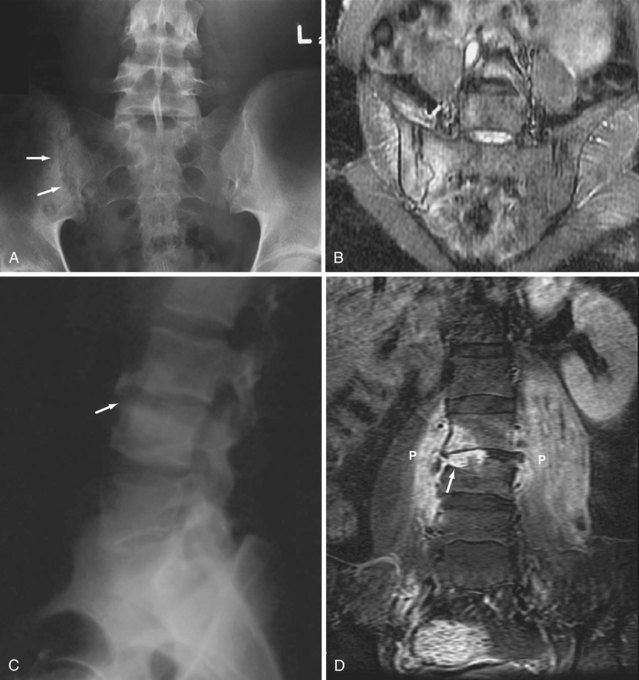
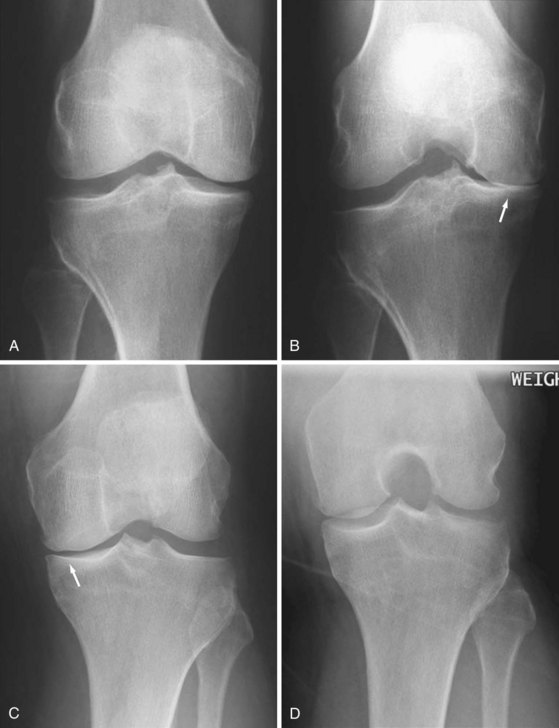
Erosion and ankylosis of the sacroiliac (SI) joints are the hallmarks of spondyloarthritis.1,23 Sacroiliitis is usually the first manifestation and is characteristically bilateral and symmetric in AS.24,25 Early radiographic findings predominate on the iliac side of the cartilage compartment, with erosion of subchondral bone causing loss of definition of the articular surfaces, usually accompanied by variable degrees of adjacent osteoporosis and surrounding reactive sclerosis. Bone erosion may result in the radiographic observation of focal joint space widening (Figure 58-3A), and as the disease progresses, the definition of the joint is completely lost with radiographic superimposition of erosion, sclerosis, and new bone formation, which fills in the erosions and the original cartilaginous “joint space.” The joint may disappear completely in late disease with ankylosis and remodeling of the bone (Figure 58-3B). The ligamentous compartment of the SI joint is frequently affected by bony erosion and entheseal proliferation, although these may be difficult to see radiographically.24,25
Spine
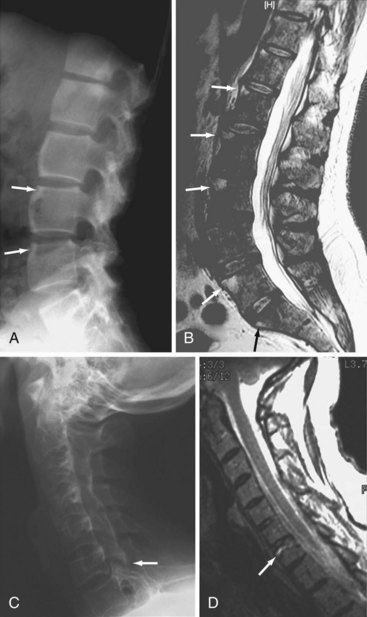
Early radiographic manifestations of AS in the spine are most often due to enthesitis at the edges of the discovertebral joints.24 Focal sclerosis (the “shiny corner”) and erosion (the “Romanus lesion”) develop at the attachment of the annulus fibrosus to the anterior corner of the vertebral end plate and are characteristic features of early AS (Figure 58-4A).26 The anterior borders of the vertebrae may appear straight or “squared” owing to periosteal proliferation of new bone filling in the normal concavity or erosion at the anterosuperior and anteroinferior vertebral margins. This observation is much easier to make in the lumbar spine, where normal vertebrae are always concave anteriorly in comparison with the thoracic and cervical spine, where the normal contour is much more variable and may be square or occasionally convex.
The hallmark of spinal disease in AS is the development of characteristic bony spurs known as syndesmophytes (see Figure 58-3C). These start as thin, vertically oriented projections of bone that develop as the result of ossification within the outer fibers of the annulus fibrosus of the intervertebral disk. Syndesmophytes are radiographically visible on the anterior and lateral aspects of the spine, starting from the corner of the vertebra. Progressive growth will bridge the intervertebral disk causing ankylosis, and extensive syndesmophyte formation produces a smooth, undulating spinal contour—the “bamboo spine.” The syndesmophytes that characterize AS must be differentiated from other spinal and paraspinal bone formations. Degenerative bony spurring in spondylosis deformans arises several millimeters from the discovertebral junction, is typically triangular in shape, and has a horizontally oriented segment of variable length at the point of origin. In diffuse idiopathic skeletal hyperostosis (DISH), bone formation in the anterior longitudinal ligament results in a flowing pattern of ossification that is usually thick, and the sacroiliac joints are not involved.
Erosion of the vertebral end plate is common in later stages of ankylosing spondylitis and may be focal or diffuse. It is also seen when pseudoarthrosis develops following a fracture in a previously ankylosed spine. Changes in the apophyseal joints are common and start with ill-defined erosion and sclerosis, but they may be difficult to see. Capsular ossification or intra-articular bony ankylosis frequently occurs in late disease (see Figure 58-4). The ankylosed spine is highly susceptible to fracture (Figure 58-4C and D), which should always be suspected in cases of unexplained pain exacerbation. Enthesitis at the interspinous and supraspinous ligamentous attachments is very common with bone formation, causing whiskering and interspinous ankylosis.
Use in Diagnosis, Monitoring, and Prognostication
Although the modified New York criteria are actually classification criteria, they are the most commonly used criteria for the diagnosis of AS and are based on clinical features and radiographic sacroiliitis.23 According to these criteria, AS may be diagnosed if, in addition to the presence of one clinical criterion, grade 2 sacroiliitis (minimal sacroiliitis: loss of definition of the joint margins, minimal sclerosis, joint space narrowing, and erosions) or higher occurs bilaterally, or grade 3 (moderate sacroiliitis: definite sclerosis on both sides of the joint, erosions, and loss of joint space) or grade 4 (complete bony ankylosis) occurs unilaterally.23 Owing to the requirement for these radiographic structural changes, the median duration of disease before diagnosis has been 7 to 10 years.27 Definitions of radiographic changes according to the New York criteria are included in the Assessment of Spondyloarthritis International Society (ASAS) classification criteria for axial spondyloarthritis,28 the European Spondyloarthritis Study Group (ESSG) criteria for spondyloarthritis,29 and the modified New York criteria.23
Radiography of the spine is not included in the classification criteria but may be useful for following structural disease progression in patients with spinal involvement. Bone changes seen in patients with axial SpA develop slowly and often are not present in patients with early disease; generally, only minor changes can be observed in 1 to 2 years. Different scoring methods, all based on assessment of lateral views, have been developed to quantify changes in the spine of patients with AS—the Stoke AS Spine Score (SASSS), the Bath AS Radiology Index (BASRI), and the modified Stoke AS Spine Score (mSASSS). A comparative study of these three methods concluded that all measures were reliable, but that mSASSS was more sensitive to change.30 These spine scores are used primarily in clinical research.
Psoriatic Arthritis
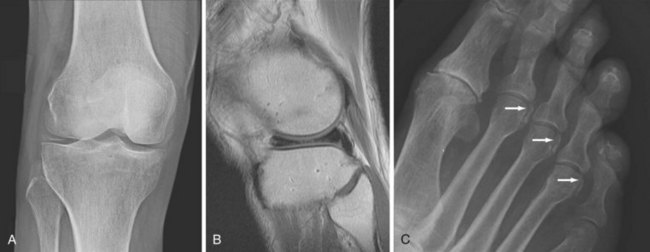
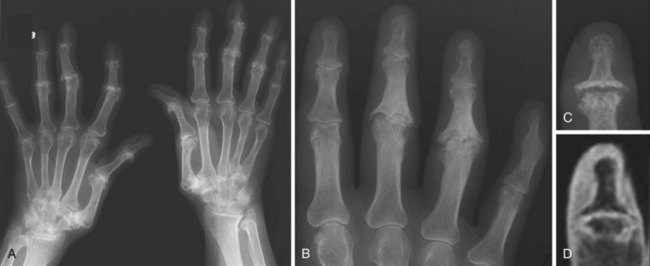
The presentation of psoriatic arthritis (PsA) is quite variable but is often distinctive. Peripheral joint involvement is common, and up to half of patients with PsA have evidence of joint damage on radiographs within 2 years of presentation.31 The hand and the wrist are most often involved and are affected in up to three-quarters of patients with PsA, but the pattern of joint involvement varies from patient to patient and over time in the individual patient. Radiographic appearances are usually asymmetric and may have a ray distribution, with affected joints involving a single digit, including the distal interphalangeal (DIP) joints (Figure 58-5), which are seldom involved in RA. Clinically, this manifests as dactylitis and is a common early presentation of PsA. Osteoporosis is frequently absent, and a distinguishing feature of PsA is the propensity for bone proliferation. This may be seen in the form of periostitis of the shafts of the phalanges, which is sometimes the first radiographic manifestation, or irregular bony spurring at joints or entheses. Fluffy new bone formation adjacent to marginal joint erosions may result in a particularly characteristic “whiskering” appearance. Severe marginal erosion of the heads of metacarpals or phalanges may produce the appearance of a whittled pencil; if combined with deep central erosion of phalangeal bases, this may be referred to as the “pencil-in-cup” appearance. Ankylosis of joints occurs frequently in advanced disease. Large joints are less often affected, and the findings are usually similar to those of RA. Spinal involvement is frequent in PsA. It may be seen in early disease, and sacroiliitis may be demonstrable in up to 75% of patients.32 Changes may be extensive and are more likely to be asymmetric than in AS.
Use in Diagnosis, Monitoring, and Prognostication
Although RA is characterized mainly by osteodestructive lesions, in PsA osteodestructive and osteoproliferative manifestations may coexist not only in the same patient, but even in the same joint.33 In particular, osteoproliferative lesions on radiography are characteristic and are included in the new classification criteria (CASPAR) for PsA.34 The presence of juxta-articular new bone formation, which appears as ill-defined ossification near joint margins (but excluding osteophytes) on radiographs of the hand or foot, is one of five criteria.34
Structural joint damage on conventional radiography is an important outcome measure in PsA. Different radiographic scoring methods of peripheral joints have been developed (e.g., the Sharp-van der Heijde modified scoring method for PsA, which is a detailed scoring system for evaluating erosion and joint space narrowing), and osteolysis and pencil-in-cup phenomena are assessed separately.11 Scoring systems are used primarily in clinical trials. No specific scoring systems are available for spine and sacroiliac joints in PsA; these areas can be monitored as in AS (see later).
Gout
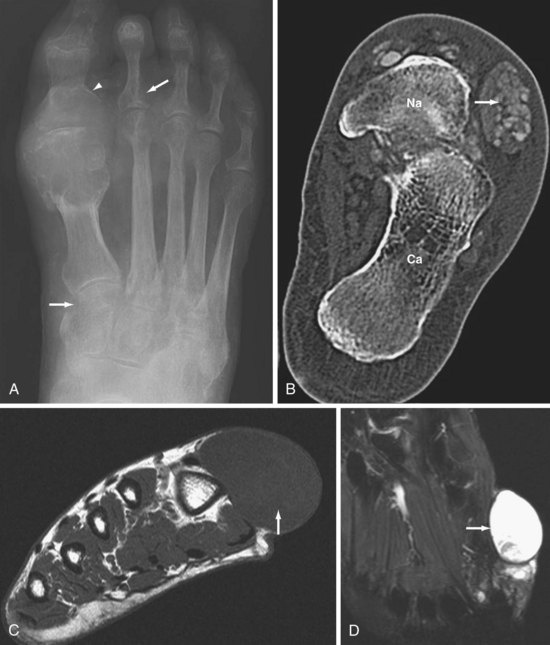
Radiography is not helpful in the diagnosis of acute gouty arthritis because findings are limited to the soft tissues and are nonspecific. Chronic tophaceous gout is an asymmetric arthritis that frequently affects the feet, hands, wrists, elbows, and knees. The most common site of involvement is the first metatarsophalangeal (MTP) joint (Figure 58-6). Radiologic changes in long-standing gout are related to tophi and their effects on soft tissue and bone.35
Tophi appear as eccentric, nodular soft tissue masses around the joints with an amorphous increased density or patchy calcification.35 Intraosseous tophi cause well-marginated subchondral cystic lucencies or intraosseous calcifications. Erosion of adjacent bone is common at the periosteal or endosteal surface. Erosions may be intra-articular, para-articular, or remote from the joint and typically are well defined with smooth sclerotic borders. An overhanging margin of bone at the edge of the erosion is a characteristic feature of gout. The joint space and bone density are usually preserved until late in the disease. Bursal inflammation commonly produces soft tissue swelling around the olecranon and in a prepatellar location.
Use in Diagnosis, Monitoring, and Prognostication
The 1977 Criteria for the Classification of Acute Arthritis of Primary Gout36 include the conventional radiographic features of asymmetric swelling and subcortical cysts without erosion. However, conventional radiographic features of acute gout have low clinical utility owing to lack of sensitivity and specificity. In the chronic phase, the characteristic pattern with asymmetric, erosive polyarticular disease and tophi appearing as nodular soft tissue masses with amorphous increased density or patchy calcification may be diagnostically useful.
A specific gout radiographic scoring method has been developed and validated and may improve sensitivity to change in longitudinal studies.37 However, radiography is not a sensitive method for monitoring gout manifestations, compared with US and MRI, because inflammatory changes and tophi volumes cannot be assessed.38
Calcium Pyrophosphate Dihydrate Crystal Deposition Disease
The term CPPD crystal deposition disease refers to a variety of clinical situations, including asymptomatic crystal deposition, pseudogout, pyrophosphate arthropathy, and other, less frequent presentations, such as pseudo-RA. Chondrocalcinosis is a general term that refers to calcification of hyaline or fibrocartilage regardless of its origin (Figure 58-7).
An acute attack of pseudogout is often a monoarthropathy, and typical radiographic findings of soft tissue swelling and joint effusion are nonspecific, mimicking gout and infection. Often chondrocalcinosis is not radiographically visible. X-ray changes in more chronic pyrophosphate arthropathy are very similar to those in OA, with joint space narrowing, sclerosis, and subchondral cysts, with or without intra-articular or periarticular calcinosis.39
The patellofemoral, radiocarpal, and second and third MCP joints are typical sites of involvement in pyrophosphate arthropathy. Distribution is usually bilateral and may be symmetric. In addition to the usual target sites, arthritic changes may be observed in non–weight-bearing joints and sacroiliac joints.40
Pyrophosphate arthropathy may be associated with extensive and rapid subchondral bone collapse and fragmentation with intra-articular loose bodies, resembling neuropathic osteoarthropathy (pseudo-Charcot). Tumorous CPPD crystal deposits that resemble gouty tophi are observed occasionally, most frequently in digits, and are referred to as tophaceous pseudogout.41
Septic Arthritis
Septic arthritis usually is monoarticular and typically is associated with rapid onset of symptoms, systemic illness, obvious local clinical signs, and laboratory evidence of acute inflammation. The earliest clinical sign is symmetric soft tissue swelling around a joint due to soft tissue edema, hypertrophied synovium, and joint effusion. However, this may be difficult to discern on x-ray, and periarticular osteopenia may be the first radiographic feature. Initial widening of the joint space may be seen, as in septic arthritis of the pediatric hip. Marginal erosions develop quickly and have a very similar appearance to other forms of erosive inflammatory arthritis. As the infection progresses, hyaline cartilage and the subchondral cortex are rapidly destroyed, with progressive narrowing of the joint space (Figure 58-8). In late stages, ankylosis of the joint may be seen. Adjacent osteomyelitis occurs with increasing frequency as the infection becomes chronic. With granulomatous infection, the characteristic triad (Phemister’s triad) of radiographic findings consists of marked osteoporosis, marginal erosions, and absent or mild joint space narrowing. Periostitis and bone production are less common.
Use in Diagnosis, Monitoring, and Prognostication
The utility of radiography is limited in septic arthritis, and immediate joint aspiration (directly or by image-guided techniques) is required for early diagnosis; more advanced imaging techniques are required for assessment of associated osteomyelitis or other complications.42 However, radiography still should be routinely performed because it may assist in interpretation of other studies and offers a satisfactory overall assessment of bone morphology and background disease.
Osteoarthritis
Osteoarthritis (OA) primarily targets hyaline cartilage, and its distribution is profoundly influenced by mechanical factors. OA is typically asymmetric in distribution both within and between joints. The most characteristic sites of osteoarthritis include hips, knees (Figure 58-9), proximal and distal interphalangeal joints of the hand, first carpometacarpal and trapezioscaphoid joints of the wrist (Figure 58-10), and first metatarsophalangeal joints.1
Joint space narrowing due to thinning of hyaline cartilage is a key feature of OA. Unlike the inflammatory arthropathies, resultant narrowing of the radiographic joint space is asymmetric, with cartilage thinning more pronounced in areas subject to greater mechanical stress, such as one compartment of the knee. Accurate assessment of cartilage loss is hugely influenced by radiographic projection, particularly in the lower limbs. Sensitive detection of cartilage thinning requires apposition of the most affected surfaces, which requires the joint to be optimally positioned with sufficient mechanical stress to bring the surfaces into contact. In the knee, for example, angular deformity tends to reduce on supine radiography, and severe cartilage loss may not be detected at all (see Figure 58-9). Bilateral weight-bearing projections are more consistent for the detection and measurement of cartilage loss, and the erect extended anteroposterior (AP) view has been established as the preferred projection for diagnostic utility. However, no view is completely reliable until global loss of cartilage occurs in late disease. Weight bearing in flexion is the most sensitive projection for cartilage thinning in the knee and is most reliable for detection of change, although the ideal degree of flexion for a specific individual varies according to the distribution of cartilage loss (see Figure 58-9).43,44
Osteophytes, the most characteristic abnormality of OA, are more specific than joint space narrowing and often are the first radiographic sign of the disease. They start as osseous metaplasia of the hyaline cartilage best seen at the articular margins but also occur centrally on the articular surface. They can become large and often dominate the radiographic appearance. Thus, osteophytes are the key criterion in defining the presence of OA.45
Use in Diagnosis, Monitoring, and Prognostication
Conventional radiography is the standard method for diagnosis and follow-up of OA. Radiographic changes are essential in the ACR criteria for classification of OA. A patient with radiographic osteophytes, knee pain, and age above 50 years, stiffness for less than 30 minutes, or crepitus should be classified as having OA.45 However, newer imaging modalities, in particular MRI, are providing additional information, which can be used in clinical trials and in enhancing our understanding of the disease.
Various scoring systems have been developed to grade the severity of OA. Among the most well known is the Kellgren and Lawrence five-point scale, which has been used widely in OA research studies.46 Atlas-based scoring systems have been developed that provide more accurate discrimination between individual radiographic features of OA.46,47 However, whichever system is used, the reliability is variable. In addition, quantitative measurement of joint space width as a surrogate for cartilage thickness has been employed in research studies, particularly in cases of knee OA, but also in hip and hand OA.
Computed Tomography
Key Points
CT is more sensitive for these purposes than x-ray and on most occasions MRI.
The role of CT of the axial skeleton is limited by a relatively large amount of ionizing radiation.
In practice, CT is rarely used unless x-ray is unclear and MRI is unavailable.
Rheumatoid Arthritis
The exquisite inherent contrast between bone and soft tissue that is afforded by CT makes it a gold standard reference for the detection of bone erosion; as such, CT is ideally suited to the investigation of erosion in inflammatory arthritides. Modern CT with isotropic voxel acquisition and three-dimensional visualization allows accurate detection and quantification of bone erosion with good intraobserver agreement, whereas radiography is limited by its two-dimensional projection and superimposition of structures. However, CT is very limited in its ability to visualize soft tissue changes, and even with contrast enhancement and complex subtraction techniques, CT remains inferior to MRI and ultrasound for assessment of synovial changes such as thickening and hyperemia. Furthermore, detection of erosion on CT and MRI shows very good agreement generally, although CT is slightly more sensitive.48,49
Use in Diagnosis, Monitoring, and Prognostication
CT is not currently used in routine clinical practice for the diagnosis of RA. However it could be useful for diagnosis in that CT appears to be the most sensitive technique for detection of erosion,48,49 and CT of the feet is very simple with a very low radiation dose. CT is used for problem solving in specific cases, such as in examination of the cervical spine if MRI is unavailable or contraindicated.
Use of CT for longitudinal assessment of damage progression has potential merit.49,50 A problem with CT in comparison with MRI or US is its inability to demonstrate improvement in soft tissue pathology. No CT data are available for prognostication in RA. Current use of CT in RA is very limited, although its ability to define erosion so clearly offers opportunities for future developments in early diagnosis and clinical trial research.
Ankylosing Spondylitis/Axial Spondyloarthritis
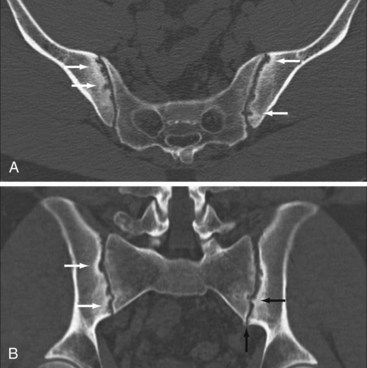
CT allows visualization of the same pathologic processes as conventional radiography—erosion, osteoporosis/sclerosis, and new bone formation/ankylosis—with the added benefit of multiplanar imaging free from superimposition of overlying structures (Figure 58-11).
Gout
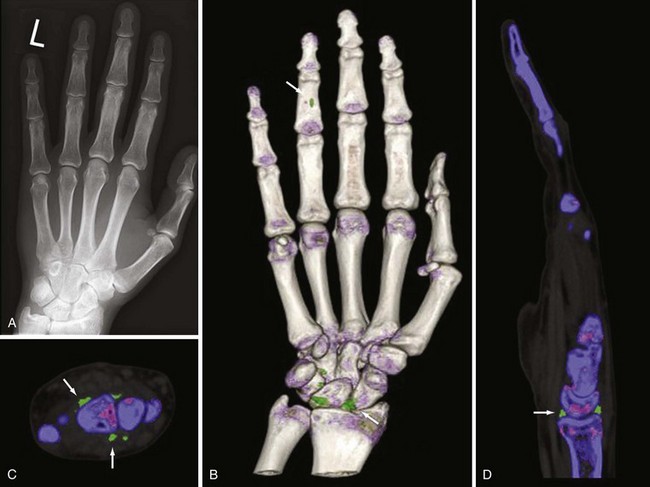
Acute and chronic tophaceous gout is associated with deposition of urate crystals in soft tissues and the development of tophi. Chronic tophi are often partially calcified, and all CT scanners are sensitive for the deposition of calcium in soft tissues (see Figure 58-6). The condition frequently involves intraosseous deposition of crystals, which is much better visualized on CT than on radiography.51 CT shows quite well soft tissue swelling due to tophi, and because anatomic bone detail is very good on CT, erosions are clearly delineated. Dual-energy CT is a new technologic development whereby images can be reconstructed based on a two-material decomposition algorithm that can separate calcium from monosodium urate (Figure 58-12).52
Calcium Pyrophosphate Dihydrate Crystal Deposition Disease
CT is not used in the management of uncomplicated peripheral CPPD, although calcifications can be visualized. CPPD can involve the cervical spine and may cause neck pain. In such patients, CT may visualize CPPD deposition at the craniovertebral junction, often in the transverse ligament of the atlas (crowned dens syndrome).55,56 Calcific deposits may also be found in other periodontoid structures and in the ligamenta flava in some patients, as may osseous abnormalities of the odontoid process such as subchondral cysts or erosions. CT may also be used for verifying the presence of calcifications at other rare locations (e.g., the temporomandibular joints).57
Septic Arthritis
CT is rarely used for the investigation of septic arthritis. Acute septic arthritis of large joints is a surgical emergency, and although x-ray, fluoroscopy, and ultrasound may be useful for diagnosis or for image-guided aspiration, no use for CT in the peripheral skeleton is known. In chronic septic arthritis and infectious diskitis, CT may be used to investigate complications, particularly osteomyelitis. However MRI or a variety of scintigraphic studies are generally preferred.42 In complex cases where MRI scanning is problematic, contrast-enhanced CT can be very useful in detecting abscess formation in deep soft tissues that are not easily assessed by ultrasound. CT guidance may be preferred for spine biopsy as well.
Osteoarthritis
Even though CT can detect bone changes such as osteophytes more accurately than x-ray,58 overall it offers little additional diagnostic utility in peripheral joints. It generally has poor soft tissue contrast capability, hyaline cartilage cannot be directly visualized, and spatial resolution of bone detail is inferior to that of radiography. It can be used in specific problem-solving situations where anatomy is complex and/or poorly visualized on x-ray, such as in the lumbar spine facet joints, but this is not usually the case in the peripheral joints. CT is used occasionally to assess extent of disease, as in the orthopedic assessment of large joints when detailed evaluation of bone morphology may be required preoperatively. CT scanning with intra-articular contrast material (CT arthrogram) may be the single most accurate modality for detecting cartilage thinning, fissuring, flaps, or volume59 (Figure 58-13), as well as loose intra-articular bodies. This requires both a minimally invasive procedure and radiation exposure and is not widely used. CT arthrography may be used as a problem-solving tool in patients who are not able to undergo MRI.
Magnetic Resonance Imaging
Technical Aspects
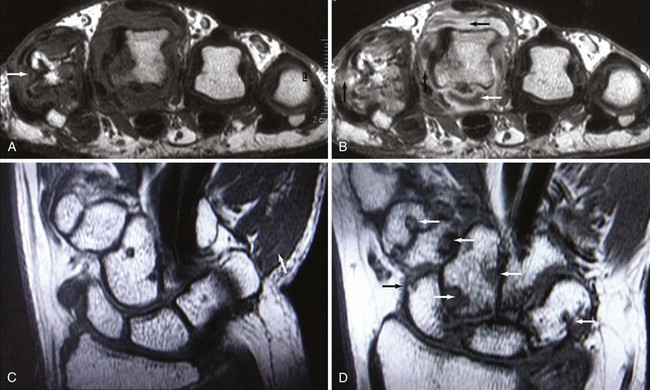
T1-weighted (T1w) imaging sequences are favored by relatively short imaging times, good anatomic detail, and the ability to visualize tissues with high perfusion and permeability, including the inflamed synovium, after intravenous contrast (paramagnetic gadolinium compound [Gd]) injection. Fat and Gd-enhanced tissues have high signal intensity on T1w images (Figure 58-14). T2-weighted (T2w) images depict both fat and fluid/edematous tissues with high signal intensity. They are used together with T1w images in degenerative spine disease, while in inflammatory conditions they are particularly useful when fat saturation (FS) techniques (Figure 58-15), in which the signal from fat is suppressed, are applied. This enhances detection of edematous tissue/fluid located in areas with fatty tissue (e.g., bone marrow edema). FS requires a homogeneous field and high magnetic field strength, which are not available on low-field (<1.0 T) MRI units. The only fat-suppressed sequence possible on low-field MRI, the short tau inversion recovery (STIR) technique, which is based on relaxation time differences, can provide information on bone marrow edema,60 but with less detail.
Some E-MRI units may provide information on synovitis and bone destruction that is not markedly inferior to what is obtained by standard sequences on high-field units,60,61 but different machines may perform very differently, and for some units, smaller field of view, longer imaging times, and lack of certain imaging techniques (particularly FS at low field) should be considered.62
Most MRI studies of the sacroiliac joint have used only one imaging plane—semi-coronal (i.e., parallel with the axis of the sacral bone). A supplementary T1w FS sequence may improve the evaluation of erosions,63 and sequences designed for cartilage evaluation (e.g., three-dimensional [3D] gradient echo sequences) may be added.64 For maximal sensitivity to changes in the ligamentous portion of the sacroiliac joints, imaging in the semi-axial plane is required.63 This may be recommended when MRI is used for diagnostic purposes, although it is probably not essential when used as an outcome measure in trials. On some indications (e.g., suspected disk herniation), MRI should include axial images; however, MRI of the spine in SpA generally involves only sagittal images, but these should extend sufficiently laterally to include the frequently involved facet and costovertebral and costotransversal joints.65 Bone marrow abnormalities in both sacroiliac joints and spine are detected almost equally well with STIR and contrast-enhanced T1w FS sequences in patients with SpA, so contrast injection generally is not needed.66,67 In contrast, injection of contrast increases sensitivity for synovitis in peripheral joints.68 For evaluation of structural (sometimes referred to as chronic) changes, such as bone erosion, new bone formation, and fat infiltration, T1-weighted images are mandatory. A supplementary T1w FS sequence may improve the evaluation of erosions.63 In peripheral and axial joints, specific sequences designed for optimal cartilage evaluation (e.g., 3D gradient echo sequences) can be applied.64
Rheumatoid Arthritis
Most MRI studies in RA have investigated knee, wrist, and finger joints. Although the knee joint is an excellent model joint for methodological studies, the clinical value of MRI is mainly dependent on its power to evaluate wrists, hands, and feet, which is also the primary focus of this section. Reports on other peripheral joints are few and are not essentially different. Definitions of key pathologies in RA and PsA are provided in Table 58-1.
Table 58-1 Magnetic Resonance Imaging Definitions of Inflammatory and Structural Lesions in Rheumatoid Arthritis, Psoriatic Arthritis, and Axial Spondyloarthritis
1 OMERACT MRI in Inflammatory Arthritis Task Force recommendations for MRI definitions of important pathologies in rheumatoid arthritis and peripheral psoriatic arthritis.86,121
2 Canada-Denmark MRI Working Group and MORPHO Group recommendations for MRI definitions of important pathologies in spine and sacroiliac joints in axial spondyloarthritis.10,104,105
From OMERACT MRI in Inflammatory Arthritis Task Force,86,121 Canada-Denmark MRI Working Group, and MORPHO Group.104,105,107
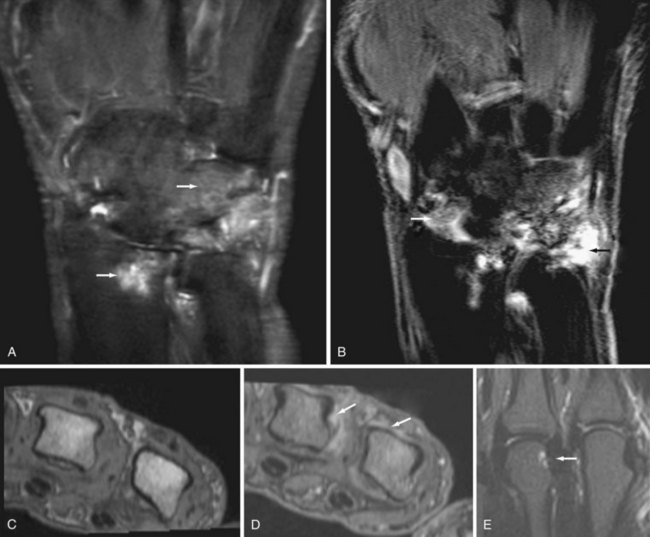
MRI allows assessment of all structures involved in RA (i.e., synovial membrane, intra-articular and extra-articular fluid collections, cartilage, bone, ligaments, tendons, and tendon sheaths) (see Figure 58-14). MRI and histopathologic and miniarthroscopic signs of synovial inflammation are closely correlated.69,70 MRI bone marrow edema (see Figure 58-14) represents inflammatory infiltrates in the bone marrow (i.e., osteitis), as has been demonstrated by comparison with histologic samples obtained at surgery in patients with RA.71,72 Whereas erosion reflects bone damage that has already occurred, bone marrow edema appears to represent the link between joint inflammation and bone destruction. A high level of agreement for detection of bone erosion in RA wrists and MCP joints (concordance at 77% to 90% of sites) between MRI and CT, the gold standard reference for detection of bony destruction, documents that MRI erosions signify true bone damage.48,73
In the cervical spine, the primary imaging modality is CR, but MRI can provide detailed information on bone and soft tissue abnormalities, which can serve as a valuable supplement to findings revealed by radiographic evaluation74–80 (see Figure 58-2C). MRI is able to directly visualize pannus tissue (e.g., around the odontoid process). Cord compression on MRI seems a better predictor of deterioration than initial clinical and plain radiographic features, supporting MRI as the imaging method of choice for evaluation of spinal cord involvement in RA.81
Use in Diagnosis, Monitoring, and Prognostication
Diagnosis
Two recent large follow-up studies of undifferentiated arthritis have documented an independent predictive value of MRI in the diagnosis of RA.82,83 The presence of bone edema (see Figure 58-15) had a positive predictive value of 86.1% for subsequent development of RA,82 and a prediction model, including clinical hand arthritis, morning stiffness, positive rheumatoid factor (RF) and MRI, and bone edema score in MTP and wrist joints correctly identified the development of RA or non-RA in 82% of patients.83
According to recent ACR/EULAR 2010 criteria for RA,6 classification as definite RA is based on the presence of definitive clinical synovitis (swelling at clinical examination) in one or more joints, absence of an alternative diagnosis that better explains the synovitis, and achievement of a total score of 6 or greater (of a possible 10) from the individual scores in four domains. In the joint involvement domain, which can provide up to 5 points of the 6 needed for an RA diagnosis, MRI and US synovitis count. In other words, MRI and US can be used to determine joint involvement in the ACR/EULAR 2010 criteria for RA.6,84,85
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree