CHAPTER 84 Spinal Cord Injury Rehabilitation
SCI is defined as an injury to the spinal cord that partially or completely interrupts the three main functions of the cord: motor, sensory, and reflex activities. Before World War I, SCI inevitably resulted in early death. Egyptian physicians long ago labeled SCI as an ailment not to be treated at all because they feared the pharaoh would kill them if they let a patient die under their care.1 However, advances in treatment that began during World War II and have progressed since have allowed many persons with SCI to live much longer. Today in many cases, patients with SCI can expect to live an almost normal lifespan. As a result, the numbers of persons alive today with SCI in the United States has increased to more than 200,000.2
Epidemiology
In 1970 the first federally funded Model Spinal Cord Injury Care Systems (MSCICS) were developed. Currently there are 14 such centers (there were 18 before funding was cut) as designated by the National Institute on Disability and Rehabilitation Research (NIDDR). Each center represents a comprehensive interdisciplinary service delivery integrating all aspects of care for the SCI patient from the initial injury to lifelong follow-up. The model centers are based on the principles set forth by Sir Ludwig Guttman, a British physician who pioneered advances in the care of SCI patients during World War II1:
The 14 MSCICS centers and 8 previously designated centers have been contributing clinical information to a national database following a jointly agreed-upon protocol. They have been able to report results on the basis of an estimated 13% of all SCI injuries in the United States occurring since 1973. The database contains information on about 25,000 spinal cord injured persons.3 The end result is a wealth of information regarding injury trends from 1973 to1998 and some recent updates that include information through the year 2004. The data not only allow physicians to know the current status of SCI patients but also give physicians the ability to spot trends in the demographic data. These changes in the demographics of SCI have allowed rehabilitation programs and centers to “change with the times” to reduce the cost and ultimately improve the quality of medical rehabilitative care for their patients with SCI.4
Incidence, Race, Age, and Gender
On the basis of information from the national database, there are approximately 11,000 new cases (40 per million in the U.S. population) each year. However, this number is based on older data from the 1970s. The prevalence is estimated to be 225,000 to 296,000 persons and growing.3 The recently updated national SCI Statistical Center statistics put the prevalence at 255,702.
Between 1973 and 2004, 66% of patients were Caucasian, 21% were African American, and 9.7% were Hispanic.3
Cervical injuries resulting in tetraplegia occur in 54% of SCI patients. Lumbar and thoracic injuries causing paraplegia comprise the rest (45%). A few (1%) were discharged as “normal” or indeterminate. Since 1990, the numbers of patients with complete SCI has been higher than incomplete injuries (56% vs. 44%).3 Since 2000, however, the most frequent injury has been incomplete tetraplegia (34.7), followed by incomplete paraplegia (23.0), complete tetraplegia (18.5), and complete paraplegia (18.5).
SCI affects young adults most often. Population-based data show the highest incidence, nearly 51.6% of injuries, occurs in the 16- to 30-year age group.3 However, the most recent trends suggest that SCI is occurring with increasing frequency in older persons. The result is that the fastest growing population of new SCI patients is older than 60 years of age. Since 1990, the incidence of SCI in persons older than 60 has increased to 12% from 4.5% in the 1970s.3 The average age at SCI has increased from 28.7 to 38.9 as of 2007. Additionally, while SCI is an injury that occurs primarily in males (81.6% since 1973), some centers are reporting increasing numbers of females suffering SCI.
Life Expectance
Life expectancies for persons with SCI that were so low many years ago continue to steadily increase. Mortality rates, however, while improving, continue to be highest during the first year after injury. Complete injuries still have lower ultimate survival rates than incomplete injuries. The degree of neurologic impairment, as measured by the Frankel Grade or the American Spinal Injury Association (ASIA) Impairment Scale, and level of injury were significantly correlated with mortality risk. For example, the average life expectancy of a 20-year-old who suffers complete paraplegia is 66 years. This compares with 61 years for a C5 to C8 SCI, 57 years for a C1 to C4 SCI, and 43 years for a ventilator-dependent patient. This compares with the normal life expectancy in the general population of 77 years and in a patient with incomplete SCI of 72 years.5 The factors that can increase life expectancy are favorable circumstances for good health, community integration, and higher income.6
Mortality
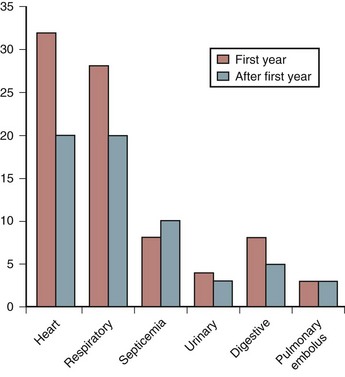
Many years ago, the leading cause of death among persons with SCI was renal failure. Pulmonary embolus (PE) usually in the first few weeks after injury, and most often associated with a deep vein thrombosis (DVT), was another frequent cause of death. Advances in urologic management, DVT and PE prevention, and early mobilization have reduced the number of early deaths. Today, renal failure rarely occurs in the acute SCI setting. Because more SCI patients live not only through their first year after injury but also survive to old age, cardiovascular disease has become a more frequent cause of death (Fig. 84–1). Nash and colleagues7 demonstrated that SCI is an independent risk factor for developing multiple risks for cardiovascular disease such as increased cholesterol. Respiratory illness, particularly pneumonia, is now the leading cause of death among older SCI patients with cervical injuries.8 The cause of death in persons with paraplegia is more varied. In addition to heart disease, cancer, suicide, and septicemia are the leading causes.
Etiology
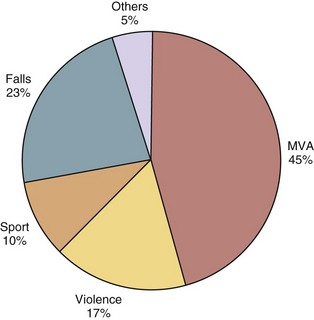
The causes of spinal cord injury are typically divided into traumatic and nontraumatic. The top five causes of traumatic SCI in males are auto accidents, falls, gunshot wounds, diving, and motorcycle accidents.3 The top three remain the same in women, but the final two are medical causes and diving3 (Fig. 84–2).
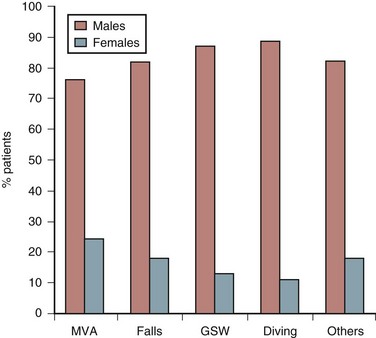
FIGURE 84–2 Etiology of spinal cord injury by gender of patients.
(Adapted with permission from Jackson AB, Dijkers M, DeVivo MJ, Poczatek RB: A demographic profile of new traumatic spinal cord injuries: Change and stability over 30 years. Arch Phys Med Rehab 85:1740-1748, 2004.)
The major trend is that violent injuries have decreased from 20% in the 1990s to 9.8% in 2000s.3 Geographic variations occur depending on the state or city. In the older SCI patients, in addition to falls, nontraumatic causes of SCI such as cancer, vascular injuries, and infection make up a larger proportion of injuries.
Etiology is also reported in terms of major groups. Figure 84–3 illustrates the frequency that each of these groups causes SCI.
Classification of Spinal Cord Injury
Two assessment tools are used in the evaluation of patients after SCI.9 One is the neurologic classification of SCI using the ASIA impairment scale. The other is the functional score, or Functional Independence Measure (FIM) score.
The ASIA impairment scale measures the degree of completeness of injury using categories from A to E (Table 84–1). Complete paralysis is defined as the absence of sensory and motor function in the lowest sacral segments and incomplete paralysis as preservation of sensation below the level of the injury including the lowest sacral segments. Sacral sparing is defined as voluntary anal contraction or presence of dull touch and pinprick sensation in the rectal and perianal area.
TABLE 84–1 American Spinal Injury Association (ASIA) Impairment Scale
| Scale Grade | Description |
|---|---|
| A | Complete—No motor or sensory function is preserved in the sacral segments S4-5. |
| B | Incomplete—Sensory but not motor function is preserved below the neurologic level and includes the sacral segments S4-5. |
| C | Incomplete—Motor function is preserved below the neurologic level, and more than half of key muscles below the neurologic level have a muscle grade less than 3. |
| D | Incomplete—Motor function is preserved below the neurologic level, and at least half the key muscles below the neurologic level have a muscle grade of 3 or more. |
| E | Normal—Motor and sensory function is normal. |
FIM score is a widely accepted functional assessment tool and is currently used as an indicator of function in patients with SCI. FIM is an 18-item scale. There are two categories of items, motor and cognitive. Individual items in each category score from 1 to 7, from “total assistance” to “complete independence.” There are 13 items of motor scoring important in determination of self-care, sphincter control, and mobility (Box 84–1). Scores falling below 6 indicate that the patient requires another person for assistance. Many rehabilitation units now use FIM scores to measure patient outcomes. Quality assurance units use these data from groups of patients to draw inferences on the effectiveness of the rehabilitation unit.
Clinical Syndromes
In addition to classifying patients on the basis of their individual motor and sensory injury levels, the following specific clinical syndromes have been identified in the literature: central cord, anterior cord, posterior cord, Brown-Séquard, conus medullaris, and cauda equina syndromes.10,11
Central Cord Syndrome
The most common type of incomplete SCI in the elderly, central cord syndrome also carries with it a relatively favorable prognosis for recovery. The mechanism of injury is usually hyperextension of an already stenotic cervical canal producing central hematomyelia.12 Because the lumbar and sacral motor tracts are located in the periphery of the white matter, the syndrome is characterized by motor weakness of the upper extremities greater than the lower extremities and sacral sparing. Recovery occurs more favorably in younger patients. Between 87% and 97% of patients younger than 50 will eventually ambulate, versus 31% to 41% of those older than 50. Hand intrinsic function is last to recover, usually incompletely in all ages.
Anterior Cord Syndrome
This is an injury to the anterior two thirds of the spinal cord including both gray and white matter. The posterior columns are preserved. The mechanism of injury is either due to thoracic fractures with retropulsion of disc or bone fragments or lesions of the anterior spinal artery. The resulting SCI is most commonly complete paralysis with spasticity. In the lower extremities deep pressure and proprioception are preserved. However, the prognosis for motor recovery is relatively poor.13
Brown-Séquard Syndrome
This injury occurs with a literal or functional hemisection of the spinal cord. The most common cause is a bullet or knife wound. The classic presentation is ipsilateral loss of motor function, ipsilateral loss of dorsal column sensation, and contralateral loss of pain and temperature below the level of the lesion. Overall, these patients have the greatest potential for functional outcome and ambulation (75% to 90%).14 Bowel and bladder continence is regained in 82% and 89%, respectively.
Therapy Approaches
On the rehabilitation unit, the SCI patient begins a new journey. The basic skills of mobility, self-care, and bladder/bowel functions are relearned regardless of the severity of the injury—sometimes quickly but other times painfully slowly. Patients try to achieve independence in each area, although sometimes only modified or partial independence is possible. The level of neurologic injury best predicts the level of independence and mobility each patient can achieve,15 even though individual differences occur. SCI patients use a significant time in rehabilitation to review a variety of handbooks and computer programs to become educated on their injury. Most patients also receive counseling for emotional and psychologic adjustment, discover initial vocational options for their future, and participate in recreational and leisure activities that ease their reintegration into society. A subset of SCI patients who suffered concurrent head trauma receives cognitive testing and retraining. Another subset of SCI patients who remain on ventilators receives breathing and communication training. Finally, each patient is discharged to the most independent living situation possible. Usually they can only go home after multiple modifications are built and after a myriad of adaptive equipment is prescribed.
Because the tasks of rehabilitation can be so daunting and involve so much of the “whole person” with all organ systems including the psychosocial system affected, only a dedicated multidisciplinary team can accomplish a successful comprehensive rehabilitation. Each member of the multiple rehabilitation disciplines functions together as a team to optimize each patient’s outcome.16 Historically the leader of the team has been the rehabilitation physician, or physiatrist, who would create the rehabilitation goals, lead weekly team meetings, and decide when the program is complete. More recently, most rehabilitation programs have shifted to a more patient-centered approach with the interdisciplinary team working toward goals set forth together.17 All disciplines focus their treatments on the patient, so teamwork and treatment overlap are encouraged. With this approach the patient, and often his or her family, is intimately involved in deciding the direction of the rehabilitation and the date of discharge. The individual disciplines are listed in Table 84–2.18
TABLE 84–2 Interdisciplinary Team
| Discipline | Role |
|---|---|
| Psychiatrist | Medical care, coordination of rehabilitation, team leader |
| Rehabilitation nurse | Daily care, medication, education, reinforce skills, transfers |
| Physical therapist | Gross motor skills, transfers, mobility (wheelchair, standing, ambulation) |
| Occupational therapist | Fine motor skills, activities of daily living (e.g., feeding, dressing, bathing) |
| Recreational therapist | Leisure activities, community reintegration, assistive technology use |
| Social worker | Obtain community resources; assist with disposition planning; communicate with insurance companies |
| Speech therapist | Assess cognitive status; provide communication, swallowing, and cognitive training |
| Respiratory therapist | Manage patients on respirator; provide respiratory treatments |
| Prosthetist/orthotist | Design and fabricate upper/lower extremity braces and prosthesis; fit spine braces |
| Vocational counselor | Test skills and interest; recommend training and assist with placement |
| Vendor | Wheelchair procurement and fitting, adaptive equipment |
Recent advances in rehabilitation techniques have led to both increased functional outcomes and decreased lengths of stay. One of the most influential advances has been in surgical stabilization. Historically, large bulky cervical braces including halos dominated rehabilitation units. Now, better internal fixation has allowed patients to come onto the rehabilitation units almost immediately after surgery and usually with only a hard collar.19 The less intrusive bracing allows patients to progress more quickly in their mobility training.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree