CHAPTER 99 Revision Spine Surgery
The number of elective spinal surgeries performed yearly continues to increase at an accelerating pace.1 Although advancements in diagnostics, implant design, and biologic adjuncts have made these operations more predictable in outcome, a significant number of patients report persistent or recurrent symptoms. These patients represent a major challenge to spine care providers. In general, the odds for success continue to fall with each additional surgical undertaking. From a statistical standpoint, the second operation has a 50% chance of success, and after the second operation, patients are more likely to be made worse than better.2 The risk of reoperation holds true independent of the index diagnosis, procedure performed, and comorbidities. As such, in the classic words of Waddell and colleagues, “caution and restraint are required when contemplating repeat back surgery.”3
Patient Evaluation
The patient’s main symptoms before the index surgery should be elicited, as well their index diagnosis and details of their operative procedure. It is helpful to obtain the medical records and imaging studies that were performed before the previous surgical procedure to compare with the current situation. In some cases, after such careful analysis an incorrect initial diagnosis may be identified.4
The time frame over which the new or recurrent symptoms developed may suggest their underlying etiology. Failure to achieve any pain-free interval after the primary surgery has been associated with incorrect preoperative diagnosis, wrong-level surgery, and inadequate decompression, particularly in the lateral recess or foraminal region.5–7 Early onset of new radicular pain after surgery may suggest nerve root injury. If pedicle screws or interbody fusion devices have been placed, their position should be evaluated with computed tomography (CT).4 In the intermediate postoperative period (1 to 6 months), mild new or recurrent pain may develop as patients progress through rehabilitation. Symptoms that develop suddenly after a specific inciting event may be from recurrent disc herniation or hardware failure. Recurrent symptoms of gradual onset in the intermediate time frame are often related to the development of scar tissue such as epidural fibrosis or adhesive arachnoiditis.2 Symptomatic pseudarthroses may also begin to manifest themselves toward the end of the 6-month postoperative period. Other causes of late postoperative pain include recurrent disc herniations or stenosis and the development of adjacent level pathology. For the surgeon who may evaluate patients initially treated elsewhere, this timeline regarding the recurrence of symptoms is crucial to establishing the proper diagnosis.
A frequent cause of failed spine surgery is poor patient selection, which unfortunately is often more apparent in retrospect. Several authors have reported that psychologic factors documented on the Minnesota Multiphasic Personality Inventory (MMPI) can be related to poor outcomes after spinal surgery.8–10 Elevation of hypochondriasis, hysteria, and depression scales are most predictive. Poor results are also more likely in patients who exhibit abnormal pain behavior, receive worker’s compensation, or are involved in litigation.11,12 The influence of these factors on outcome, however, is poorly understood. The combination of physical, psychosocial, and economic problems with which patients with FBSS present make individualized treatment plans necessary.
Another regrettably common cause of persistent symptoms is performance of the wrong surgical procedure. Wrong-level discectomy is the most common avoidable error in spine surgery. The North American Spine Society has advocated intraoperative radiographic localization in almost all cases to help prevent this misfortune.7 Another example of an improper procedure is to decompress only what appears to be the most stenotic level and leave other areas, particularly in the lateral recess or foraminal region, unaddressed. Likewise, failure to identify the pain generator before performing an interbody fusion for discogenic pain can lead to disappointing results. Many patients will have one or two significantly abnormal discs on magnetic resonance evaluation but other areas with comparably mild changes, which may be surprisingly concordant at discography. In our experience, such patients are advised against surgery.
Leg Pain Versus Back Pain
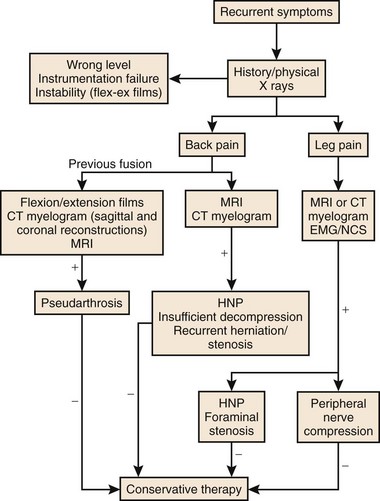
As with primary lumbar degenerative syndromes, differentiating the degree of axial back pain from radicular or neurogenic claudicatory symptoms is paramount to formulating an effective treatment plan. Although the diagnostic tests that follow will help identify the pathoanatomy, all of the findings should be kept in context with the patient’s main complaint. An algorithm based on the patient’s main complaint is presented in Figure 99–1.
Physical Examination
The physical examination of patients with FBSS is similar to the initial examination for nonoperated patients. Posture, alignment, and gait should be observed. Range of motion, tenderness, and nerve root tension signs should be elicited and a thorough neurologic examination should be performed. Although the physical examination findings of this condition may be nonspecific, a thorough examination may be helpful to exclude other conditions that may have similar presentations. In patients with leg pain, thorough examination of the hip and knee and assessment of distal pulses may reveal other potential causes of their symptoms. Nonorganic physical findings should be recorded, as described by Waddell.13
Diagnostic Testing
Plain Radiography
Plain radiographs of the lumbar spine should include biplanar standing films. A coned-down lateral of the lumbosacral junction and an anteroposterior Ferguson view may be particularly helpful in evaluating patients who have had previous surgery at the L5-S1 level. Patients with pain after a discectomy should be evaluated for iatrogenic pars fracture. Endplate rarefaction suggests a postoperative discitis. Previously placed spinal implants should be scrutinized for malposition, loosening, subsidence, or breakage. In addition to evaluation of the disc height, endplate characteristics, and alignment, anterolisthesis or retrolisthesis at the adjacent levels should be noted (Figs. 99–2A and 99–2B). Flexion-extension radiographs should be scrutinized for motion involving segments where fusion was attempted because gross motion detectable in this manner typically indicates pseudarthrosis. These films should also be evaluated for dynamic instability at levels adjacent to the previous surgery. The overall sagittal balance should be assessed on standing 36-inch radiographs. A flatback deformity from poorly contoured constructs or adjacent segment degeneration (ASD) is best evaluated with this technique.
Advanced Imaging
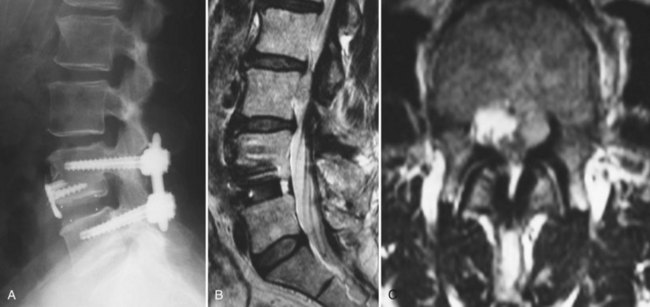
CT with fine-cuts and sagittal/coronal reconstructions can help delineate fusion status.14 The adjacent levels should be carefully evaluated for hypertrophic facet arthropathy, spondylolysis, and pedicle stress fractures. Magnetic resonance imaging (MRI) (Fig. 99–3) with and without gadolinium enhancement is the most sensitive test for evaluating neurologic compression at both the previously operated site and the adjacent levels.15 Comparison of the enhanced and nonenhanced sequences can help differentiate epidural scar (enhancing) from recurrent disc herniations and hypertrophic ligamentum flavum (nonenhancing). With the advent of MRI, CT myelography is less commonly required but is still helpful in certain situations. Patients with contraindications to MRI, pronounced scoliosis, known stainless steel implants, or patients with substantial imaging artifact degrading magnetic resonance images can be better evaluated with CT myelography.
Diagnostic Injections
Selective nerve root injections have been shown to have both therapeutic and diagnostic efficacy in the management of patients with lumbar radicular pain.16,17 Controlled studies documenting their predictive value in patients with failed back surgery syndromes are lacking. The authors have found them useful in situations where MRI findings are equivocal and in situations where multilevel pathology is present. The use of provocative discography is controversial, and its role in the evaluation of previously operated discs is not universally accepted.18 Some reports indicate that discography may predict surgical outcome for patients undergoing anterior lumbar interbody fusion for persistent pain after successful posterior fusion.19,20 The role of discography in evaluating degenerated adjacent discs as pain generators is also unclear. Although this has been suggested as a useful test in determining whether a degenerated segment below a long thoracolumbar fusion is a significant pain generator,21 the role of provocative discography in the more common setting of adjacent segment disease cranial to more limited lumbar procedures continues to be poorly defined.
Nonoperative Treatment
Nonoperative treatment will typically be involved to varying degrees in many patients with FBSS because measures such as physical therapy and pain medications are part of the usual postoperative regimen for most index lumbar procedures. When persistent complaints have triggered the workup outlined earlier, a diagnosis can be rendered in many cases. Several conditions may be amenable to nonoperative treatment (Box 99–1). The degree to which a patient’s symptoms can be attributed to epidural scar formation is unclear. Epidural scar is an incidental finding in many patients who do not develop symptoms, and most feel that the scarring will inevitably return to some degree after revision, making the indication for repeat surgical intervention unclear. As new agents to help prevent proliferative epidural scarring are developed, the overall management of such patients may continue to evolve.
Nonoperative treatment may have a potential role in several other conditions. Fractures involving noninstrumented fusion masses, pedicles within the fused segments, or compression fractures cephalad to a fusion may be treated with bracing in the absence of frank instability. Postoperative discitis may be treated with biopsy, immobilization, and directed antibiotic therapy, but infection after instrumented fusions generally merits surgical intervention (see Chapter 98). Sacroiliac symptoms are rarely treated with arthrodesis, and medical/physical therapy plus fluoroscopically guided injections are favored. Leg symptoms due to peripheral neuropathy should be managed medically, and leg symptoms due to peripheral nerve compression may be amenable to bracing or specialized local care. Leg symptoms due to recurrent disc herniation or stenosis can be treated initially with physical therapy and nonsteroidals. Epidural injections may be offered as a treatment option; however, their effectiveness in the postoperative patient is less predictable. Transforaminal and caudal injections have some reported therapeutic success in uncontrolled studies.22,23 Some have theorized that the presence of postoperative scar tissue interferes with dispersion of the injectate and leads to diminished effectiveness. Patients with diffuse degenerative disease and axial symptoms are typically advised to pursue intensive multidisciplinary rehabilitation combining physical training with cognitive behavioral interventions, vocational planning, and consultation with a specialist in chronic pain medical management (covered separately in Chapters 104–107).
Spinal cord stimulation has been used for more than 30 years to treat chronic pain including FBSS. A thorough review is conducted in the chapter on interventional procedures for chronic pain. As one can imagine, it has been difficult to conduct well-designed trials to compare spinal cord stimulation to revision surgery. In a review published in 2004 of 583 studies of spinal cord stimulation, the only class I randomized controlled trial did not include patients with FBSS.24 A subsequent randomized controlled trial compared reoperation and spinal cord stimulation for patients with FBSS. Using at least 50% pain relief as a criterion for success, the authors reported that spinal cord stimulation was significantly more successful than reoperation.25 The authors typically consider implantable spinal cord stimulators an option for patients with back and leg pain after spinal fusion when solid arthrodesis is apparent by imaging studies and all other potential causes of persistent pain have been excluded. A trial stimulator is typically done first and converted to a permanent device if the patient demonstrates a favorable response.
Surgical Treatment—Back Pain Predominant
Pseudarthroses and Failed Instrumentation
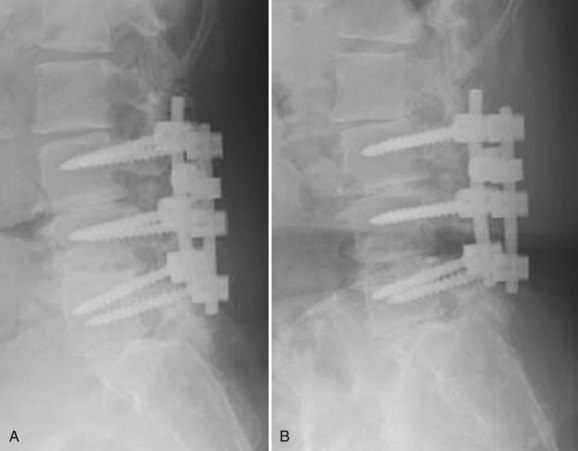
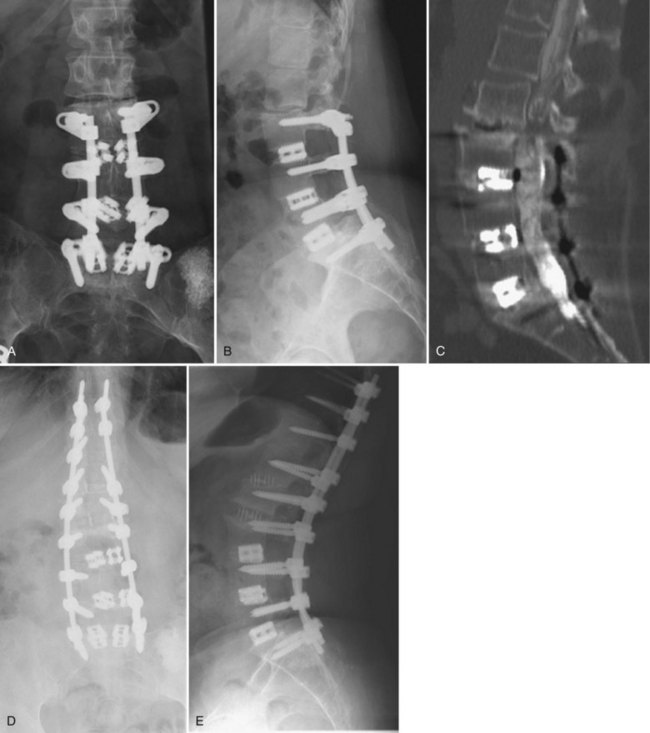
Although pseudarthroses are not always symptomatic, they do account for a considerable proportion of cases of FBSS. Likewise, instrumentation failure typically occurs in the setting of a pseudarthrosis; however, broken implants have been reported in cases of solid fusion. When either of these conditions is felt to be the cause of a patient’s FBSS, any revision surgery should be carefully planned and executed. There is no commonly accepted treatment algorithm for surgical management of pseudarthrosis. The surgical approach must be individualized on the basis of the location and etiology of the nonunion, the absence or presence of failing implants requiring revision (Fig. 99–4A to E), the medical status of the patient (including any previous abdominal surgeries), and the surgeon’s comfort level with specific techniques.
Nonunions after a posterior fusion that have adequate bone stock can often be managed by a revision posterior approach.26 This should involve decortication of the pseudarthrosis and posterolateral fusion with instrumentation. At exploration of the fusion, the rods and screws are typically removed first. If the screw lengths and diameters are not known from the previous operative note, these should be determined from direct inspection and measurement. The posterolateral fusion masses are then exposed, and any fibrous tissue associated with pseudarthrosis is débrided. If any question remains regarding the gross stability of the fused segments, we typically cannulate the previous pedicle screw holes with blunt probes or appropriate-sized curettes and attempt to elicit segmental motion by cantilevering the instruments. Use of a gouge to decorticate the previous fusion masses helps preserve any remaining bone stock. Because removing and replacing a pedicle screw results in a 34% decrease in pullout strength, some attempts must be made to improve purchase. If the anatomy permits, increasing the diameter of the screw by at least 1 mm or increasing the length by 5 to 10 mm can help improve fixation. Using a different pedicle screw system is beneficial because the screws will have a different thread pitch. In severely osteoporotic bone, polymethylmethacrylate (PMMA) can augment fixation strength. Achieving fusion can be challenging in such revision scenarios. As such, local bone is preferentially augmented by morsellized autograft harvested from the iliac crest. Allograft and demineralized bone matrix are used as extenders rather than graft substitutes. If both iliac crests have been previously used, other strategies for enhancing fusion should be considered. Options include electrical stimulation, bone marrow concentrates, or bone morphogenetic protein, although few studies have been performed to delineate their role in this circumstance.
When breakage of a pedicle screw is encountered, spanning that level with the addition of a crosslink is an option. If the affected level is at the end of the construct, extension of the fusion can be considered. If it is felt that reinstrumentation of the level with the broken screw is necessary, then screw removal is necessary. Commercially available screw removal sets are available, but most techniques involve removal of additional bone, so augmentation may be necessary. Careful patient selection is necessary to identify those patients who will benefit from a posterior or posterolateral strategy rather than a more involved procedure. West and colleagues26 reported a 35% pseudarthrosis rate and 47% clinical failure rate in patients with preexisting pseudarthrosis in whom posterior fusion and pedicle screw fixation were performed.
In cases of failed posterolateral fusion with inadequate bone stock (such as after a wide decompression), the interbody region may provide the best area of vascularized host surface for revision grafting. Excessive epidural scarring makes entry into the interbody space through virgin areas more desirable. Anterior lumbar interbody fusion (ALIF) permits placement of structural allografts or cages with large footprints. Using an anterior-posterior approach in this situation has been associated with the best fusion rates.27 For salvage of a single-level pseudarthrosis where decompression is not necessary, anterior lumbar interbody fusion followed by percutaneous pedicle screw fixation has recently been described, with radiographic fusion reported in 52 of 54 cases.28
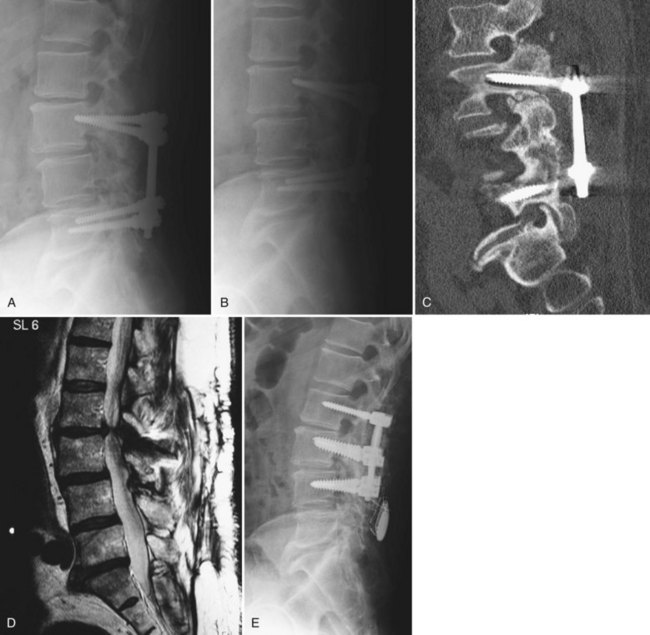
In cases where an anterior approach is problematic, a transforaminal interbody fusion (TLIF) is a good option29,30 because the entry site to the interbody space may be less involved by postoperative scar. Studies examining this technique specifically for the management of pseudarthrosis, however, are lacking. Another recently described technique, the extreme lateral interbody fusion (XLIF), offers an additional approach to the interbody space without a formal anterior exposure (Fig. 99–5A to E). This involves development of the plane between the posterolateral margin of the paraspinals and the peritoneal sac, followed by splitting the fibers of the psoas muscle. This technique requires high-quality intraoperative neurophysiologic monitoring of the nerve roots of the lumbar plexus31 and intraoperative fluoroscopy. Although early reports of the technique have demonstrated encouraging results with low complication rates,32 data on this technique for revision arthrodeses are currently lacking.
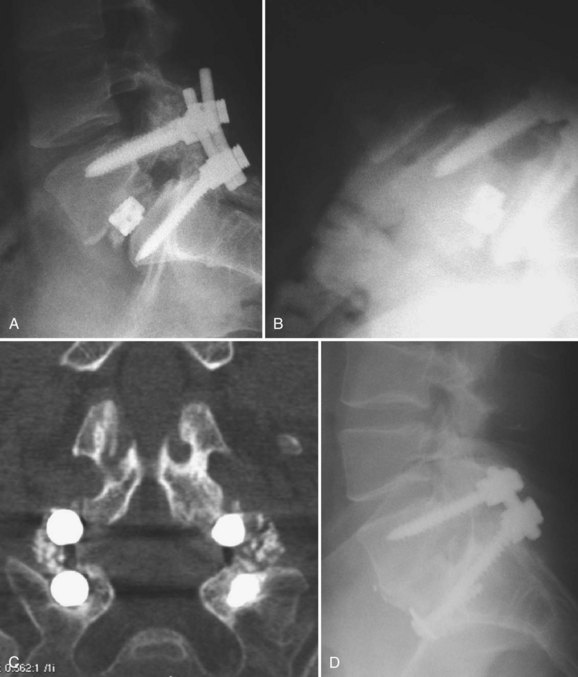
Revision of pseudarthroses after attempted posterior lumbar interbody fusion (PLIF) or TLIF can be challenging. The authors prefer to remove the cage from an anterior approach to the disc space (Fig. 99–6A to D). Osteotomes are used to create planes adjacent to the cage and assist removal. The choice of implant to then use for the ALIF is based on the defect left after removal of the cage. Whether a revision posterior fusion is required is then based on the integrity of the previously placed posterior implants and the overall segmental stability.
Anterior nonunions and failed lumbar disc arthroplasty present unique challenges. If the failed ALIF or arthroplasty device is not grossly unstable, a posterolateral fusion with instrumentation may suffice.33, 34 A recent article reported that removal of the prosthesis and circumferential fusion yielded slightly better results than posterior fusion at 1-year follow-up.35 The authors, however, acknowledged that significant risks are inherent with removal of the arthroplasty device, so further studies are necessary to clarify management of this scenario. If the patient had a 360-degree fusion and the posterior fusion is solid, an isolated revision ALIF can be performed. The entire construct should be revised in cases of failed 360-degree fusions with both anterior and posterior pseudarthroses and unstable implants. Revision anterior lumbar procedures are technically demanding. Vascular injuries are common, and an experienced approach or vascular surgeon should be considered.36 Strategies to help navigate the hazards of the revision anterior approach have been recommended. A previous left retroperitoneal approach to L5-S1 may be best revised through a right retroperitoneal or transperitoneal approach. A previous left retroperitoneal approach to L4-5 may be best revised through a transperitoneal or transpsoas approach.37 Because retroperitoneal scarring may obscure the anatomy, other measures such as preoperative ureteral stenting and intraoperative cannulation of the iliac veins with balloon catheters can be considered.
Painful Hardware
A small subgroup of patients may report recurrent pain despite intact implants, a solid fusion, and no other obvious pain generators. Implant removal in such cases remains controversial, particularly because the mechanism as to how the implants generate pain is not well understood. Some authors have recommended injection of local anesthetic around the hardware as a diagnostic step.38 For patients who report temporary relief of their pain with the injection, removal of the implants can be considered, provided that the patient clearly understands that such an intervention may not have a predictable outcome.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree