CHAPTER 98 Postoperative Spinal Infections
Incidence/Epidemiology
Postoperative wound infections are the most common complication following spinal surgery. The incidence documented in the literature has historically been quite variable with reported ranges from 0.5% to 20%.1–7 This discrepancy is in part to significant variation in case complexity, use of instrumentation, and surgical approach in the reported cases. In general, increasing the complexity and invasiveness of the surgery correlates with a higher incidence of infection.
Historically, lower-risk spinal surgeries include those that do not require instrumentation. Discectomy and laminectomy have reported incidences of infection less than 3%. With the addition of instrumentation, the incidence of postoperative infection increases to greater than 12% in some studies.8–18 Specifically, lumbar discectomy has had a reported incidence of 0.7%, and using a microscope for the procedure increases the incidence to 1.4%. In the United States, the National Nosocomial Infections Surveillance (NNIS) System, a Centers for Disease Control and Prevention (CDC) orchestrated voluntary performance-measurement system, has reported a 1.25% rate of surgical site infection following laminectomy and a 2.1% rate following laminectomy with noninstrumented fusion.19
Consistently throughout the literature, cases that require more extensive soft tissue dissection, longer operative time, greater blood loss, more significant soft tissue devitalization, or the creation of dead space have an increased infection rate. One study comparing infection rates in patients undergoing discectomy alone and those undergoing discectomy and fusion showed infection rates of 1% versus 6%, respectively. In other reports fusion without instrumentation has been associated with an infection rate ranging from 0.4% to 4.3%.20 The use of devitalized bone graft material in fusions results in an infection rate from 1% to 5%.
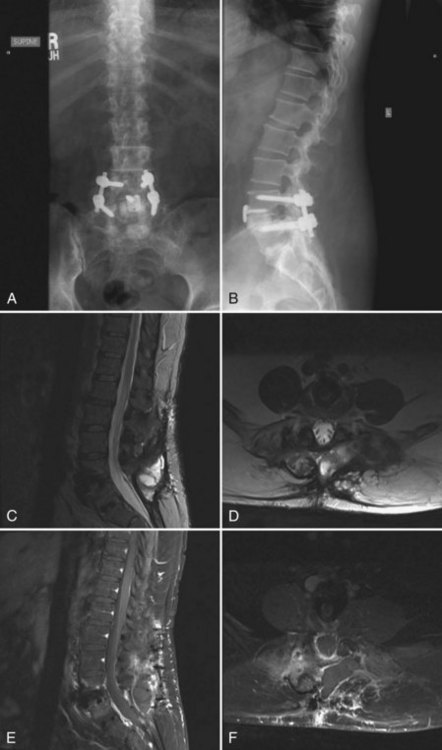
As the use of instrumentation has become more commonplace, attention must be paid to a possible associated increased infection risk. Colonization of implanted devices occurs in upwards of 50% of patients, although most do not display clinical symptoms of infection. Although implants rarely act as the initial source of infection, they may become a nidus for inoculation and subclinical growth of infectious organisms. The implant provides an avascular surface on which bacteria can create a glycocalyx, which serves as a barrier to the host immune response and antibiotic treatment. In addition, micromotion can create metallosis and subsequent granulomas, which may become a potential site for bacterial colonization. Other theories postulate that local soft tissue inflammation and postoperative seromas may serve as a potential cause for the increased infection risk seen with instrumented fusions (Fig. 98–1). Due to these unique risk factors, instrumented fusions have reported postoperative infection rates of up to 20%8,21 with dramatic variation in the reported literature. Moe reported a postoperative infection rate of 7% in cases with Harrington instrumentation.22 More recent literature reports infection rates in elective instrumented surgical cases between 2.8% and 6%.8,23,24 Many authors feel that the actual infection risk with the use of spinal instrumentation is between 5% and 6%.16–18,23,25
Spinal trauma patients represent a unique population that has an increased risk for developing postoperative infections. The significant soft tissue devitalization and devascularization caused by the traumatic event results in local hypoxia leading to tissue necrosis, edema, acidosis, and hematoma formation. This combination results in a media optimal for bacterial proliferation isolated from the host defenses.26 Systemically, the patient sustaining major trauma shows a hyperinflammatory state with alterations in the normally tightly controlled homeostasis of pro- and anti-inflammatory cytokine levels. The resultant imbalance leads to a state of immunosuppression that is thought to increase susceptibility to infection. In addition, comorbid factors such as age, medical conditions, poor nutritional status, and body habitus cannot be controlled in the same manners that they are in elective surgery. The presence of complete neurologic injury significantly increases the risk of postoperative infection. In the largest clinical series investigating 256 surgically treated traumatic spinal injuries, the rate of postoperative wound infections was 9.4% compared with an infection rate of 3.7% seen in patients undergoing elective spinal surgery during the same time period at the same hospital.27 Similar reviews have found postoperative infection rates in spinal trauma patients ranging from 9% to 15%,8,28,29 which is greater than the previously discussed average infection rate seen in elective spinal surgeries.
Anterior spinal procedures appear to be less susceptible to infection than posterior procedures. Infection rates following anterior cervical spinal surgery have been reported in the literature to be as low as 0% to 1%.30–32 Anterior thoracic and lumbar surgery also display significantly less infection risk than their posterior counterparts, with rates 50% lower than those occurring after posterior surgery. The infection rates for anterior approaches are likely decreased by multiple factors including better vascularity of the spinal column and decreased dead space creation. Furthermore, the incidence of infection in combined anterior and posterior procedures does not appear to be greater than posterior surgery alone.27
Risk Factors
Obese patients are also considered at high risk for developing postoperative infections.33–35 Overweight patients often require more extensive dissection through poorly vascularized adipose tissue. The resulting tissue devitalization and fat necrosis result in an environment favoring bacterial growth and proliferation. In addition, the increased operative time and blood loss necessary with obese patients increase their risk of infection. Obesity in itself is a risk factor for malnutrition, diabetes, and other medical comorbidities, further contributing to a poor healing environment with diminished immunogenic potential.
Nonmodifiable risk factors that may increase the susceptibility to infection must also be evaluated before surgery. Thorough assessment of a patient’s medical history during the preoperative evaluation may reveal systemic comorbidities that should be identified and optimized before surgery. In all patients, preoperative infections, whether in the spine or elsewhere, should be addressed and treated before undergoing elective surgery.10,23,30,36 Conditions such as rheumatoid arthritis, acquired immunodeficiency syndrome (AIDS), adrenocortical insufficiency, long-term corticosteroid use, and malignancy may pose significant risk for developing postoperative infections.37–39 A thorough discussion of potential complications associated with these confounding medical conditions is important during preoperative counseling. Medical optimization of these conditions may limit potential postoperative complications. Although age is not considered an independent risk factor, older patients are more likely to have comorbidities associated with an increased risk of postoperative infection.
Microbiology
Three potential sources are hypothesized to be responsible for postoperative infections: (1) direct inoculation during the operative procedure, (2) contamination during the early postoperative period, and (3) hematogenous seeding.21,40–43 Of these, direct inoculation during the surgery is the most common, making aseptic technique and the appropriate use of prophylactic antibiotics of paramount importance.
Gram-positive cocci are the most common pathogens responsible for acute postoperative infections. The most commonly reported organism in the literature is Staphylococcus aureus, which causes greater than 50% of the infections in some reports.23,31,34 Other common gram-positive species that cause postsurgical infections include Staphylococcus epidermidis and β-hemolytic streptococci. Common gram-negative organisms cultured from infected surgical sites include Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Enterobacter cloacae, Bacteroides, and Proteus species.
Infections that present greater than 1 year after surgery are generally caused by low-virulence organisms such as coagulase-negative Staphylococcus, Propionibacterium acnes, and diphtheroids.30,41 These organisms can be present as normal skin flora, and it is hypothesized that prolonged surgical wound drainage and inflammation may result in these infections. These low-virulent organisms are usually rapidly cleared by the host immune response with appropriate treatment and generally do not result in a clinical sepsis. In a retrospective review of postoperative infections presenting more than 1 year after surgery, 10 of 11 patients with cultures incubated for longer than 1 week grew low-virulence skin organisms.44
Hematogenous spread can also cause surgical site infections. These blood-borne infections are usually due to highly virulent organisms including gram negative bacteria. These infections are often associated with systemic illness and sometimes have grave consequences such as multisystem organ failure. Due to repeated cannulization of the venous system, intravenous drug users have a higher incidence of gram-negative infections, as do patients who have prolonged hospital admissions.34
Diagnostic Modalities
Laboratory Testing
ESR elevates following spinal surgery and may not normalize until several weeks postoperatively. Variations in this time period to normalization have been reported. In one study, ESR rarely elevated to levels greater than 25 mm/hour and returned to baseline levels by the third postoperative week.45 Another study showed that ESR elevation was prolonged and lasted up to 6 weeks postoperatively.46 Peak ESR levels have been shown to correlate with the degree of invasiveness of the surgery, with more extensive surgeries causing higher ESR elevations than less invasive procedures.47
As with ESR, CRP values rise sharply during the initial postoperative period. Unlike ESR, however, CRP decreases to baseline levels more rapidly. CRP levels generally peak on the third day postoperatively and return to baseline within 10 to 14 days. This rapid normalization makes CRP a more sensitive indicator of infection and a more useful diagnostic tool when determining the presence of infection, especially in the acute and subacute postoperative period.46–50 Elevated ESR or CRP outside of this postoperative period can indicate a developing infection and can be used to monitor the efficacy of treatment.
The precise and accurate identification of the culprit organism is a critical step in the treatment of a postoperative spinal infection. Cultures obtained from the superficial wound are often contaminated with skin flora and can confuse the diagnostic workup. Some authors suggest early aspiration of a suspicious wound in order to attempt to isolate the infectious organism.21 If there is no fluctuant mass to aspirate, as is often the case, computed tomography (CT) or fluoroscopic guidance can be used to accurately obtain a deep culture from the affected area. Frequently, fine-needle aspiration of the affected region does not provide ample tissue for an accurate diagnosis. We prefer to obtain a core biopsy specimen in order to ensure that an adequate sample is provided to the laboratory. Blood cultures can reveal the responsible organism if taken in a septic individual before the initiation of antibiotics. If the blood cultures are positive and provide identification of an organism, it can be presumed that the same organism is the cause of the spinal infection and a biopsy of the spinal infection site is not necessary. The most accurate cultures are those obtained during the surgical débridement before the administration of antibiotics. In many cases, however, such a surgical intervention is not necessary and these surgical cultures are not obtained.
Imaging
Plain radiographs are often the first imaging modality obtained during workup of a suspected infection. Findings on plain radiographs frequently can be quite subtle, and up to 4 weeks are often required to pass before radiographs show evidence of infection.50 Inspection of the instrumentation for loosening or adjacent bony lysis may be clues of an infection. In cases of postoperative discitis, disc space narrowing is the first radiographic finding. This change generally occurs 4 to 6 weeks postoperatively. Early infectious disc space changes, however, may be difficult to distinguish from degenerative changes. Paravertebral soft tissue swelling may indicate the presence of an abscess, especially in the retropharyngeal space or paraspinal musculature. More significant radiographic findings such as reactive bone formation, endplate destruction, osteolysis, and deformity indicate a more significant infectious process and usually require at least 2 months to develop.
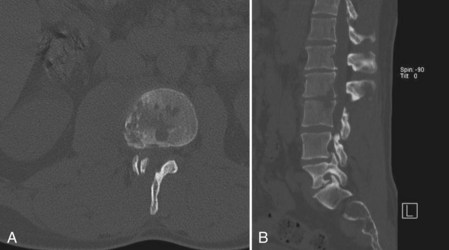
CT provides a more detailed view of spinal anatomy and may allow for earlier detection of postoperative infections than plain radiographs. Endplate changes, bony lysis, and/or soft tissue fluid collections can indicate early infection (Fig. 98–2). As the infection progresses, more significant bony and intervertebral disc destruction may be seen. Implant-related artifact may distort the detail and limit the usefulness of CT scans in patients with spinal instrumentation. CT-guided biopsies can also be used to provide an aspirate for culture or tissue biopsy from infected soft tissue or bone as noted earlier.
Nuclear medicine studies are sometimes used to supplement other radiographic methods when working up a postoperative infection. Unlike magnetic resonance imaging (MRI) and CT, nuclear medicine studies are not limited by the implant-associated artifacts. Bone scans are often nonspecific and may show generalized uptake around the surgical site in a postoperative spinal infection.50 Although gallium-67 and technetium-99m scans provide early evidence of postoperative infections, their diagnostic value is somewhat compromised relative to studies evaluating the appendicular skeleton. Diagnosis of early postoperative disc space infection is better achieved with gallium-67 relative to technetium-99.51,52 Indium 111-labeled WBC scans will often have limited usefulness because of their poor specificity, particularly in the early postoperative period. Technetium-labeled ciprofloxacin, when combined with single photon emission computed tomography (SPECT), has been shown to have improved sensitivity and specificity over other nuclear medicine modalities, particularly if performed more than 6 months after surgery.53
MRI is the most important imaging modality when evaluating postoperative spinal infections. Because of its high contrast resolution, MRI with and without intravenous gadolinium contrast is the most effective imaging technique available. Relative to other imaging modalities, MRI is both highly sensitive (93%) and specific (96%) when evaluating spinal infections.54–56 As with other imaging techniques, it may be difficult to distinguish nonpathologic postoperative changes from infections on MRI scans obtained in the early postoperative period. Thus accuracy and reliability of the study is dependent on the elapsed time from the date of surgery, the level of clinical suspicion, and correlation with other diagnostic tools. Spinal instrumentation, particularly when composed of stainless steel, can cause significant MRI artifact and severely limit the diagnostic utility of the study.
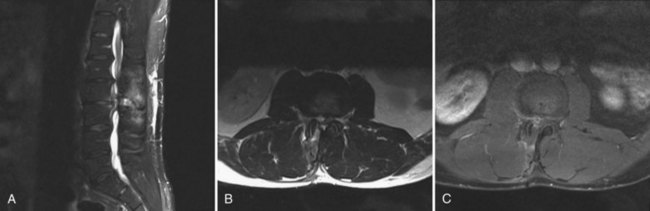
MRI can identify, with high sensitivity and specificity, postoperative osteomyelitis, discitis, and epidural abscesses. An epidural abscess will display a T1 isointense fluid collection with potential obliteration of the otherwise well-defined neural elements, and the T2-weighted images show significant increased intensity. Abscesses will display ring enhancement on T1 images following the addition of IV gadolinium (Fig. 98–3). Osteomyelitis appears as areas of vertebral body and disc space hypointensity on T1-weighted images and hyperintensity on T2 images. In addition, there is a loss of definition between the vertebral bodies and the intervertebral disc space.55–57
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree