CHAPTER 88 Osteoporosis
Surgical Strategies
Osteoporosis is a systemic disease characterized by decreased bone mass and microarchitectural deterioration. The resulting decrease in bone mechanical strength typically manifests as fragility fractures, with about one half of osteoporotic fractures occurring in the spine. According to the National Osteoporosis Foundation, low bone mass currently affects more than 44 million Americans and frank osteoporosis affects 15% of postmenopausal white women and 35% of women older than 65 years of age. In addition, 50% of white women will sustain an osteoporotic fracture at some time during their lifetime. Men have a lower but still significant osteoporotic fracture risk, which peaks 10 years later than the risk in women.1
Osteoporotic Vertebral Compression Fractures
Osteoporotic vertebral compression fractures are a leading cause of disability and morbidity in the elderly.2–4 The consequences of these fractures include pain and, in many cases, progressive vertebral collapse with resultant spinal kyphosis. Osteoporotic vertebral compression fractures have been shown to adversely affect quality of life, physical function, mental health, and survival.5–7 These effects are related to the severity of the spinal deformity and are, in part, independent of pain.6,7
Nevertheless, the pain associated with acute vertebral compression fractures may be incapacitating. In a number of cases, the pain will subside over a period of weeks or months, although it is not uncommon for the pain to become chronic.7 Chronic pain after vertebral fracture most likely results from (1) incomplete vertebral healing with progressive bony collapse, (2) altered spine kinematics as a consequence of spinal deformity, or (3) the development of a pseudarthrosis at the involved vertebra. Chronic pain associated with vertebral compression fractures often leads to impaired quality of life and depression.6,7
Kyphotic deformity in the osteoporotic spine may also create a biomechanical environment favoring additional fractures. The kyphotic deformity anteriorly shifts the patient’s center of gravity, creating a longer moment arm acted on by the center of gravity. This results in greater flexion-bending moments around the apex of the kyphosis, which will promote further increases in kyphotic angulation and additional fractures.8,9 Clinical studies report that the risk of a new vertebral fracture in the first year after an incident vertebral compression fracture rises 5 to 25 times above baseline,10–12 with the vertebra adjacent to the previously fractured level at particular risk.13–14 Prevention of progressive kyphotic deformity or correction of existing deformity may therefore be important both in reducing the adjacent level fracture risk and in preventing the consequences of spinal kyphosis such as impaired pulmonary function.
Nonsurgical Treatment
Traditionally, acute osteoporotic vertebral compression fractures have been treated nonsurgically except in rare cases of fractures associated with neurologic compromise or advanced spinal instability. Spinal surgery in the osteoporotic patient is fraught with complications related to the patient’s advanced age and frequent comorbidities and due to the difficulties in securing fixation in osteoporotic bone. Thus the treatment of most patients with painful vertebral compression fractures traditionally includes bed rest, analgesic medications, bracing, antiosteoporotic drugs, or some combination thereof.15–18 Although these treatments appear to be reasonable, anti-inflammatory and narcotic medications are often poorly tolerated by the elderly and may predispose to confusion, increased risk for falling, and gastrointestinal side effects. Bed rest can lead to an overall physiologic deconditioning and acceleration of bone loss. In addition, bracing is typically poorly tolerated by older patients, is expensive, and may further restrict diaphragmatic excursion.
Vertebroplasty and Kyphoplasty
Orthopedic fracture care emphasizes restoring anatomy, correcting deformity, and preserving function. These goals have been largely ignored in the management of patients with osteoporotic spine fractures. Recently, minimally invasive procedures to address the pain and deformity associated with osteoporotic vertebral compression fractures have been developed. Vertebroplasty, involving the percutaneous fluoroscopically guided injection of polymethylmethacrylate (PMMA) directly into a fractured vertebral body, has been used to stabilize osteoporotic vertebral compression fractures. Substantial pain relief in a majority of patients treated with vertebroplasty has been reported.18–25 Kyphoplasty is a minimally invasive procedure that involves the percutaneous insertion of an inflatable bone tamp into a fractured vertebral body under fluoroscopic guidance. Inflation of the bone tamp will elevate the endplates, restoring the vertebral body back toward its original height while creating a cavity to be filled with bone void filler, most commonly PMMA. Results of kyphoplasty suggest significant pain relief, as well as the ability to improve height of the collapsed vertebral body and reduction of spinal kyphosis.15,25–32
When recommending vertebroplasty or kyphoplasty to treat a painful fracture, the vertebral compression fracture must be confirmed as the source of the patient’s back pain. This requires careful correlation of the patient’s history and clinical examination with radiographic documentation of an acute or nonhealed vertebral compression fracture. The physician should treat the symptomatic fracture(s) and should not indiscriminately treat multiple vertebral fractures seen on radiographic studies. Magnetic resonance imaging (MRI) is useful for detecting edema, which may indicate an acute vertebral fracture, and for helping to rule out malignancy or infection. Tumors resulting in vertebral compression fractures are usually associated with an ill-defined margin, enhancement, pedicle involvement, as well as a paravertebral soft tissue mass.33 Sagittal MRI with short tau inversion recovery (STIR) sequences highlights the marrow edema changes associated with acute vertebral compression fractures and is useful in determining the acuity of a vertebral compression fracture.
Indications and Contraindications
Vertebroplasty is designed primarily to relieve pain, and the procedure may be considered when a painful osteoporotic vertebral fracture does not respond to a reasonable period of conservative care. In addition to providing pain relief, kyphoplasty is designed to reduce the fractured vertebra so that intervening before fracture healing offers the best chance of achieving optimal fracture reduction.34 If significant kyphosis is already present at the time of presentation of an acute vertebral compression fracture, we consider kyphoplasty to improve sagittal alignment. In contrast, for patients with acute vertebral compression fractures and relatively minor degrees of vertebral collapse, an initial trial of nonsurgical care may be considered, during which serial radiographs are obtained. If the patient’s pain is incapacitating or does not respond to a period of nonsurgical care, kyphoplasty or vertebroplasty may be recommended. If progressive collapse of the vertebral body is observed during the trial of nonsurgical care, kyphoplasty is recommended. In a recent randomized controlled trial comparing kyphoplasty to nonoperative treatment, a significantly more rapid improvement in quality of life, function, mobility, and pain was observed in the kyphoplasty group.35
Techniques and Results
Vertebroplasty
Vertebroplasty may be performed in a radiology suite or operating room and is typically performed with the use of local anesthesia. The patient is positioned prone with the spine extended by chest and pelvic bolsters. Typically, an 11- to 13-gauge needle is advanced toward the center of the vertebral body using a transpedicular or extrapedicular approach and fluoroscopic guidance. If necessary, biopsy needles can be used to obtain samples before cement injection.36,37 PMMA, the bone cement most commonly used, is mixed with barium for fluoroscopic opacification. Whereas some physicians treat patients with intravenous antibiotics, some physicians add antibiotics to the cement mixture itself, especially when operating on immunocompromised patients.25,32,36,38 When the mixture attains the consistency of toothpaste, the cement is transferred to syringes or specially designed cement injection tools. Between 2 and 10 mL of cement is injected into the vertebral body under live, multidirectional fluoroscopy. Cement injection is stopped if extravertebral extravasation is detected. Ideally, the vertebral body is completely filled with cement, but pain relief has been reported when the anterior two thirds of the vertebra contains cement.32 The patient is not moved from the prone position until the cement has cured. Most patients rest supine under observation for at least 4 hours before discharge.
The mechanism of pain relief after vertebroplasty is not clear. One possible explanation is a mechanical immobilization of the fracture and support to the cortex by the cement.39 Another theory suggests that the heat produced during PMMA polymerization causes deafferentation of the fractured vertebra.
Research on the outcome for vertebroplasty has suggested that most patients experience partial or complete pain relief within 72 hours of the procedure.18–25,36,40,41 Overall, 60% to 100% of patients noted decreased pain after vertebroplasty with pain reduction maintained for months up to 10 years.21,24 In addition to decreased pain, improved functional levels and reduced analgesic medication requirements have been reported.24,36,42–45 Published studies have noted a low complication rate for vertebroplasty, with most complications resulting from extravertebral cement leakage causing spinal cord injury, nerve root compression, or pulmonary embolism.18,19,21–25,36,40,41
The limitations of the vertebroplasty technique relate to the inability of the procedure to correct spinal deformity and the risk of extravertebral cement extravasation during injection. In certain instances, some degree of postural fracture reduction is achieved with vertebroplasty.46–48 During vertebroplasty, the high-pressure injection of low viscosity cement directly into cancellous bone makes it difficult to control cement flow in the vertebral body. This creates an unpredictable risk of cement extravasation outside the vertebral body.32 In fact, extravertebral cement extravasation rates of up to 65% have been reported for vertebroplasty.40 Although a low risk of clinically relevant complications results from cement extravasation, cases of fatal pulmonary embolism and paralysis have been described.49–52
Kyphoplasty
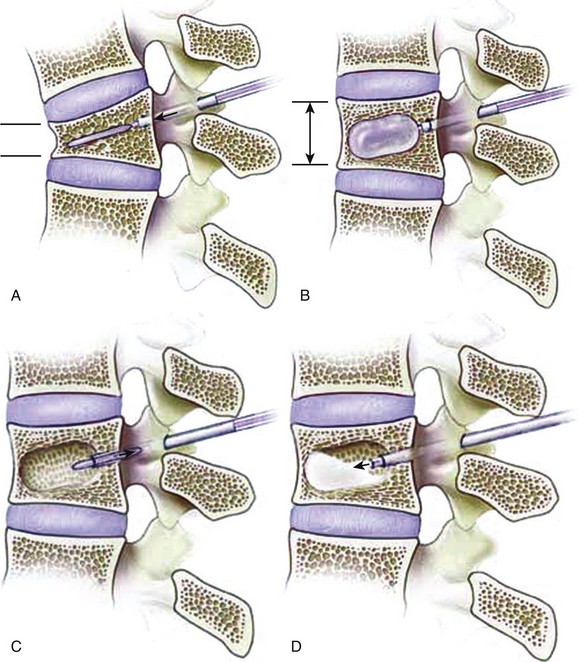
Kyphoplasty involves the percutaneous placement and expansion of an inflatable bone tamp in a fractured vertebral body. The tamp elevates the depressed vertebral body endplate(s), thereby restoring vertebral body height, and also creates a cavity within the vertebral body that is then filled with bone void filler (Fig. 88–1).15,25–32 The patient is positioned prone on a spinal frame. Fluoroscopy is used throughout the procedure, and we have found simultaneous, biplanar fluoroscopy to be advantageous. After needle positioning into the fractured vertebral body through a transpedicular or extrapedicular approach, a series of tools creates a working channel into the vertebral body. Once inserted through the cannula into the vertebral body, the balloon tamp is expanded using visual (fluoroscopy), volume, and pressure (digital manometer) controls. The inflation of the tamp continues until one of the following endpoints is reached: (1) fracture reduction is achieved, (2) the maximal balloon pressure or volume is reached, or (3) cortical wall contact occurs. The balloons are then deflated and removed. Thick cement can then be fed through the cannula under low pressure to fill the void created by the balloon tamp. The cement volume should approximate the volume of the intravertebral cavity. The patient is not moved from the prone position until the leftover cement has solidified.
Garfin and colleagues53 reported the initial multicenter experience with kyphoplasty to treat 2194 vertebral fractures in 1439 patients between 1998 and 2000. Ninety percent of patients reported significant pain relief within 2 weeks of the procedure.53 In this large series of kyphoplasty procedures, a 0.2% per fracture complication rate was observed. Lieberman and colleagues30 observed highly significant improvement in physical function, role physical, vitality, mental health, and social function scores of the SF-36 questionnaire after kyphoplasty. These authors reported five clinically insignificant cement leaks (8% overall).30 Phillips and colleagues34 reported on 29 patients treated with kyphoplasty. In this study, mean visual analog scale (VAS) pain scores decreased from 8.6 preoperatively to 2.6 one week postoperatively to 0.6 one year postoperatively. Cement leaks without apparent clinical consequence occurred in 6 of 61 vertebral levels with no cases of cement leakage into the spinal canal.34
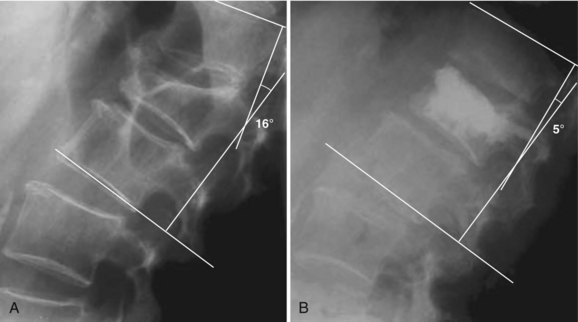
Kyphoplasty has the potential to improve spinal deformity by elevating the vertebral endplates before fixation. In an ex vivo study, Belkhoff and colleagues26 showed a 97% reversal of deformity with kyphoplasty compared with a 30% reversal with vertebroplasty. Lieberman and colleagues30 reported vertebral height restoration in 70% of 70 fractured vertebrae treated with kyphoplasty. In those patients in whom the vertebral fractures were reduced by kyphoplasty, vertebral height was increased by a mean of 46.8%. Wong and colleagues32 and Garfin and colleagues28 similarly noted increased vertebral body height after kyphoplasty. Theodorou and colleagues studied 15 patients (24 fractures) with osteoporotic vertebral compression fractures and reported a mean improvement in kyphosis of 62.4% ± 16.7%.31 Phillips and colleagues34 reported that, in their early experience with kyphoplasty, local sagittal alignment was improved by a mean of 8.8 degrees for all fractures and 14.2 degrees in those fractures that were reduced by at least 5 degrees (Fig. 88–2). Voggenreiter, in a series of 39 patients, reported a kyphosis reduction of 6.5 degrees with intraoperative prone positioning and a subsequent improvement of 3.4 degrees with inflation of the balloon tamp. However, he did note a subsequent loss of 3.1 degrees on upright standing films post operatively.47 Hulme and colleagues,54 in their systematic literature review, reported the average angular correction with kyphoplasty to be 6.6 degrees.
Three recent multistudy analyses have pooled the literature results and complications of kyphoplasty and vertebroplasty. In these analyses, clinical improvement was noted after both kyphoplasty and vertebroplasty.54,55 Kyphosis correction was noted more frequently in kyphoplasty than in vertebroplasty. Overall, complications were rare in both groups. Pooled results suggested the mortality and medical complications were comparable in both procedures.55,56 The rate of cement leakage, asymptomatic and symptomatic, was noted to be higher in vertebroplasty than in kyphoplasty in all three reviews. Lee and colleagues56 reported the symptomatic cement leak rate to be 1.48% in vertebroplasty and 0.04% in kyphoplasty (P < 0.05). These differences are likely due to subtle differences between the techniques. In vertebroplasty, the cement is usually less viscous and injected under a higher pressure and subsequently more difficult to control.
Instrumentation of the Osteoporotic Spine
Cervical Instrumentation
In the subaxial spine, anterior cervical instrumentation is frequently used to maintain alignment and improve fusion rates after anterior bone grafting. Few clinical reports specifically addressing anterior fixation in the osteoporotic spine have been published. Biomechanical studies of anterior cervical plating have shown a direct relationship between bone mineral density (BMD), screw insertion torque, and the axial force generated at the plate-screw junction.57 Because low BMD predisposed to plate pullout, Zink57 suggested that stable anterior cervical fixation with 3.5-mm screws cannot be obtained with a BMD below 150 mg/mL. The risk of anterior cervical plate failure in osteoporotic bone is reduced by increasing screw length and placing bicortical screws.57 The desire to increase stability with bicortical cervical screw fixation should be tempered by the neurologic risk associated with screw penetration of the posterior vertebral body cortex.
Anterior cervical plate-screw constructs spanning multiple levels have been reported to have a high rate of clinical failure, which usually resulted from graft-plate dislodgement.58 Biomechanical studies have confirmed that excessive screw-vertebra motion caused by fatigue at the lower end of a multiple-level corpectomy construct may explain the clinically observed failures at the caudal end of long anterior cervical plate constructs.59 This research also suggested that longer screws, larger diameter screws, and supplemental posterior fixation would potentially decrease screw-vertebra failure.59 These findings seem to be particularly relevant in osteoporotic bone where impaired bone quality would predispose to excessive screw-vertebra motion and failure. DiAngelo and colleagues60 have reported that although multiple-level cervical plating increases construct stiffness after corpectomy, the anterior plate results in the graft being excessively loaded in extension. Because these loads may exceed the adjacent endplate yield strength, pistoning and strut settling into the adjacent vertebral body may occur.60 This problem is accentuated in the osteoporotic spine, where the adjacent vertebral endplates are less able to resist graft settling. When using anterior instrumentation over multiple levels in the osteoporotic cervical spine, the surgeon should attempt to use as many points of fixation as the construct will allow. If the surgeon is concerned that anterior cervical screw purchase is compromised in osteoporotic bone, it may be advisable to proceed with segmental posterior cervical instrumentation to stabilize the anterior arthrodesis.
Dens fractures are the most common condition of the upper cervical spine requiring surgery in the elderly. This population is predisposed to dens fractures by osteoporosis of the dens coupled with the increased rigidity of the subaxial spine as a consequence of age-related degenerative spondylosis.61 Halo immobilization of these injuries has been recommended; however, this is poorly tolerated in the elderly and is associated with a high incidence of nonunion and complications.62,63 Dens fractures in this age group are frequently treated surgically using anterior or posterior approaches. For example, acceptable clinical results have been reported for the widely used technique of posterior C1-2 fusion using either wiring or screw fixation.64,65 Andersson and colleagues66 reported an unacceptable rate of anterior screw fixation failure and nonunion in eight elderly patients with dens fractures. In contrast, Berlemann and Schwarzenbach67 reported a low failure rate and successful healing of the fracture in 16 of 19 elderly patients with dens fractures treated with anterior screw fixation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree