Normal Hip Embryology and Development
Elizabeth Weber and Andrew W. Ritting
Key Points
Introduction
Normal hip development is the result of a carefully orchestrated series of prenatal events, which act in concert to form the acetabulum, the femoral head, intra-articular structures of the hip, and the blood supply to the hip joint. After birth, the hip joint remains immature and the intimate relationship between the femoral head and the acetabulum is crucial for appropriate postnatal development. This chapter will elucidate normal hip embryology and some of the postnatal pathology that can influence the ultimate shape and motion of the mature adult hip.
Intrauterine life is divided into three phases: blastocystic, embryonic, and fetal. The blastocyst phase refers to fertilization through the end of the second week post conception. Following blastocyst, the period of primary tissue differentiation is weeks 2 through 8. During this embryonic phase, the entire musculoskeletal system develops in the following ways: both upper and lower limb buds appear, along with the cartilaginous anlage of the osseous limbs; joints, longitudinal growth plates, and vascular supply to the bones are formed; and, ultimately, the complex articulated human mobility system is fully differentiated and formed. Vascular insults, confining pathology, and teratogens, which act during this phase, have the greatest likelihood of interfering with the development of healthy functional symmetric lower extremities. The fetal phase, from 8 weeks to birth, consists of refinement of the vascular supply, ossification of the diaphysis of long bones, and formation of intra-articular structures. The hip joint is fully mature at 35 weeks post conception. The complex genetic code and signaling pathways, which direct the development of the human hip, are mostly unknown.
The Embryonic Phase of Prenatal Development
At the end of the third week of development, the three primary germ layers—ectoderm, endoderm, and mesoderm—have formed and will give rise to all tissues in the developing embryo.1 After the formation of the notochord and neural tube, the mesodermal tissue alongside forms the paraxial mesoderm, which subsequently divides into 38 paired somites. These are the primordia of the future axial skeleton and all associated muscles.1 The mesoderm is also responsible for most of the structures in the appendicular skeleton. Cartilage, connective tissue, striated muscle, bone, and blood vessels all take their origins from the mesoderm. The appendicular skeleton begins at day 26 with upper limb buds, which appear as paired outpouchings along the lateral wall of the developing embryo. More caudal, at the level of the lumbar and first sacral elements,2 lower limb bud swellings appear at day 28. At this time, a cellular template for the future femur exists. Chondroblasts aggregate and condense, and through chondrification, the separate aggregates condense to form the femoral anlage.3 At the same time, a similar process is defining the future innominate.3
Development and refinement of the limbs are continuous during the embryonic period, with the upper extremities preceding the lower at all points. Between 37 and 40 days, foot plates are formed, and by 46 days, the digital rays are visible. The limbs continue to elongate ventrally toward each other. By 55 days, the toe webbing has disappeared and the toes are distinct.1 Within the lower limb bud, cells are dividing and differentiating into specific tissues. The bones, which will form the articulating hip joint, begin as a solid mass of chondroblasts. The first recognizable structure to appear is the cartilage model of the femoral diaphysis during the sixth week. Precartilage is present at the future site of the femoral head, which is indistinguishable at this time from the acetabulum. Undifferentiated mesenchymal cells called blastemal cells appear as the trochanteric projection.4 Blastemal cells will form apophyses, precartilage will form the covering at the articulations of long bones, and cartilage will form the anlagen for the osseous structures.2
During the seventh week, an interzone appears, which will distinguish the sides of the hip joint. Proximally, the acetabulum appears as a shallow depression that must deepen from 65 to 180 degrees during its development.2 Distally, the femoral head and the overlying articular cartilage are forming. In the middle, autolytic degeneration occurs by programmed cell death in this interzone to separate the two structures and form the actual joint space.5 This is the first time the hip joint could theoretically be “dislocated.” The hip is well protected from dislocating in all directions except for inferiorly because the transverse acetabular ligament is poorly defined.2 The beginnings of synovial tissue and the ligamentum teres are found at this time in the middle layer of the interzone. Blastemal cells at the periphery of the acetabulum condense to begin to form the acetabular labrum.4 At the end of the embryologic stage, the secondary center of ossification appears in the ilial portion of the pelvis. No secondary center of ossification is present in the femur, but the neck of the femur has elongated.
The Fetal Phase of Prenatal Development
From 8 weeks post conception until birth is the fetal phase. The vital organs and the appendicular skeleton are completely formed. This phase is focused on ossification, vascularization, and maturation. Ossification in the femur proceeds proximally and distally from the center. The labrum has coalesced as a triangular structure in coronal section. The lower limb bud begins to internally rotate so the knee joint moves from lateral to anterior.3 In the 11th week, the femur is characterized by a 2-mm spherical head and 5 to 10 degrees of anteversion, which will continue to increase to 45 degrees at 36 weeks’ gestation.6 The limb is characterized by knee flexion and hip flexion and adduction. By the 16th week, the muscles investing the hip joint and the hip capsule are formed. The joint capsule joins distally with the femoral perichondrium.2 Internally, the acetabular labrum, the ligamentum teres, and the transverse acetabular ligament are now formed. Mature hyaline cartilage covers the articulating surfaces of both the femoral head and the acetabulum. The femoral shaft is fully ossified, and the ossification center of the ischium is now evident.4 Muscles are formed and attached and can actively move the hip; the left leg comes to override the right.3 As the vascular supply matures, a distinction is made between the metaphyseal supply and the epiphyseal supply. Retinacular vessels enter the head and neck.2 At 20 weeks’ gestation, the spherical femoral head is 7 mm and anteversion is 25 to 30 degrees. At 32 weeks, the shaft ossification has reached the level of the greater trochanter. Both the ilium and the ischium are ossified4 (Table 14-1; Figs. 14-1 through 14-7).
Table 14-1
Prenatal Lower Extremity Development
| Time Post Conception | Embryonic Development | Hip Development |
| 28 days | Lower limb bud appears | |
| 37 days | Foot plates appear | |
| 46 days | Digital rays are visible | |
| 55 days | Toe webbing is gone | |
| 6th week | Cartilage model of femoral diaphysis appears | Trochanteric projection (blastemal cells) and nonarticulating hip joint (cartilage) |
| 7th week | Interzone appears, which separates the acetabulum from the femoral head | |
| 8th week | Secondary center of ossification in the ilium |
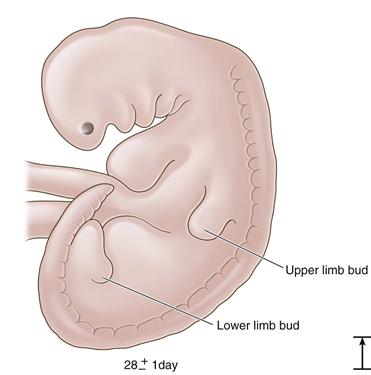
Figure 14-1 The 28-day embryo: the first appearance of the lower limb bud (length, 4 mm).
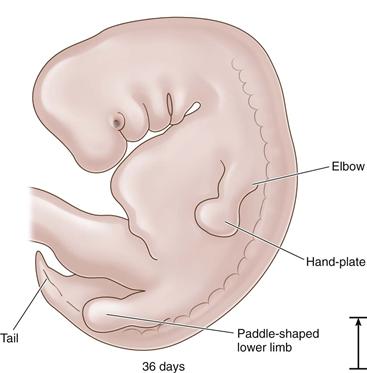
Figure 14-2 The 36-day embryo: paddle-shaped limb (length, 9 mm).
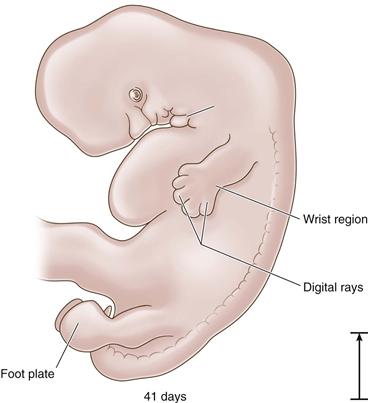
Figure 14-3 The 41-day embryo: foot plate appears (length, 10 mm).
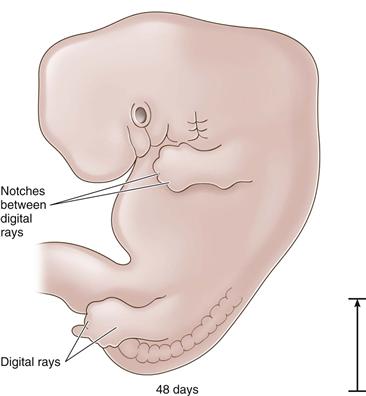
Figure 14-4 The 48-day embryo: digital rays are distinguishable (length, 15 mm).
Figure 14-5 The 52-day embryo: fan-shaped foot with webbed toes (length, 19 mm).
Figure 14-6 The 56-day embryo: toes are separated (length, 27 mm).
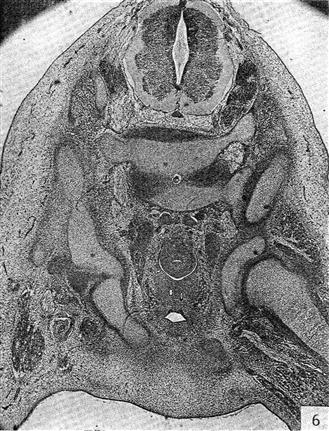
Figure 14-7 Human hip at the end of embryonic development. Note that the femoral head is completely distinct from the acetabulum. This is the first time the joint could conceivably dislocate. (From Strayer LM: The embryology of the human hip joint. Yale J Biol Med 16:13–26, 1943.)
Femoral Anteversion
Femoral anteversion increases from 5 degrees in the 11th week to 45 degrees at 36 weeks’ gestation. Postnatally, femoral anteversion begins to decrease as the result of muscle forces acting across the hip joint.5 Normal postnatal femoral anteversion is approximately 30 degrees at 1 year and 15 degrees at skeletal maturity.7
Neck Shaft Angle
The neck shaft angle is very valgus in early development, decreasing from 145 degrees in the 15th week to 130 degrees at 36 weeks’ gestation.6 Postnatally, with normal muscle forces acting across the hip joint, a further decrease in the neck shaft angle to approximately 127 degrees occurs at 18 months.8
Both femoral anteversion and the neck shaft angle are found to remain constant postnatally in cerebral palsy patients, suggesting that abnormal muscle forces inhibit normal postnatal development of the hip.9
Postnatal Development
Acetabulum
At birth, the acetabulum is immature, consisting of a cartilaginous ring surrounding the femoral head. At its depth, the ring is confluent with a Y-shaped triradiate cartilage. Eventually, with growth, the arms of the triradiate cartilage coalesce to form the medial, nonarticular acetabulum. Growth of the triradiate cartilage is interstitial. During this same period, the ring of cartilage investing the femoral head enlarges to become the load-bearing acetabulum, which grows at pace with the developing femoral head by apposition. The triradiate cartilage develops from three primary centers of ossification. The os acetabuli is the pubic contribution. It is the largest center of ossification and eventually will form the anterior wall of the mature acetabulum. The superior dome is formed from the iliac contribution, and the posterior wall is formed by the ischial contribution. The shape and depth of the adult acetabulum are heavily dependent on the dynamic interaction of the mature femoral head. A preponderance of acetabular shape and depth has been established by the appearance of ossification centers at 8 or 9 years of age. This is the watershed age, after which very little acetabular remodeling can be expected; it therefore defines the prognosis in many pediatric hip disorders. Ossification centers of the acetabulum fuse between 17 and 18 years of age.10
Femur
At birth, the ossified portion of the femur has progressed proximally to the level of the greater trochanter and the femoral neck. Three separate growth plates define the cartilaginous proximal femur: the longitudinal growth plate (LGP) of the femoral neck, the growth plate of the greater trochanter (TGP), and the connecting growth plate on the lateral neck called the femoral neck isthmus (FNI) (Fig. 14-8). The LGP and the TGP have divergent growth vectors that additively create longitudinal growth of the proximal femur along the axis of the femoral shaft. Alterations in growth of either of these physes result in angular deformities of the mature hip. Additionally acting on the LGP is contact pressure from the acetabulum, which forces spherical appositional growth within the socket. The mature shape of the femoral head and that of the acetabulum are inter-related and rely heavily on the dynamic and continuous reciprocal relationship of the round head in the round socket. Disruptions in this relationship result in failures of formation and imperfect adult hip joints. For example, if developmental hip dysplasia is unrecognized and untreated, the resultant acetabulum is shallow and steep from inconsistent pressure from the femoral head within the developing socket.
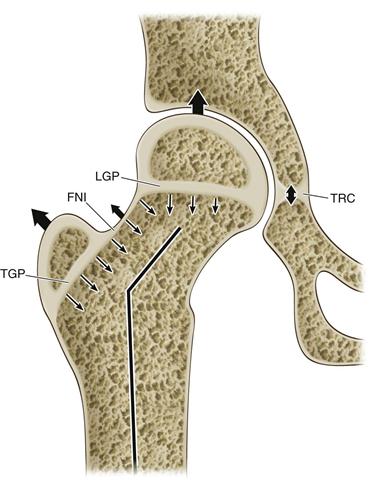
Figure 14-8 Growth plates at the hip joint. On the femoral side, the trochanteric growth plate (TGP) grows by traction, and the longitudinal growth plate (LGP) grows by apposition. The femoral neck isthmus (FNI) connects the two and allows widening of the femoral neck with growth. On the acetabular side, the triradiate cartilage (TRC) allows the socket to expand by interstitial growth.
Blood Supply
Femoral
The first evidence of vascularity to the femur occurs at 8 weeks post conception, as capillaries break into the cartilage model of the femur in the middle third of the diaphysis, at the site of the primary ossification center. This eventually will become the nutrient artery in the mature bone. By the 14th week, a ring of vessels has encircled the femoral neck to form primitive retinacular vessels. These vessels invade the cartilage model. This primitive femoral model of vascularity does not change appreciably from week 14 to skeletal maturity.11 The postnatal vascularity of the proximal femur can be broken down into three distinct supplies: an extracapsular ring of vessels, ascending cervical vessels, and intracapsular vessels.
The extracapsular ring comes directly from the femoral artery and travels around the femoral neck as the medial and lateral circumflex arteries. These vessels converge laterally in the piriformis fossa. They provide most of the blood supply to the proximal femur and greater trochanter.11 The vascular anastomosis in the piriformis fossa is particularly vulnerable to embarrassment during drill entry into the femoral canal, as well as to femoral neck fracture. During growth, the contribution to the overall supply of the proximal femur by the medial circumflex increases, as the lateral circumflex contribution becomes less significant. By the age of 10, a 50% decrease has occurred in the number of vessels along the medial and anterior aspect of the neck; this has important implications for vascular preservation during femoral neck osteotomy.
Ascending cervical branches from the extracapsular ring pierce the hip joint capsule and travel under the synovium along the neck inside the hip joint. These branches enter the bone and travel distally in the metaphysis; they either anastomose with the ascending diaphyseal vessels or travel lateral or medial to supply the greater trochanter or the femoral neck, respectively.
The intracapsular synovial ring of vessels ascend superficial to the perichondrial ring and supply the epiphysis. In this way, the physis serves as a barrier between the blood supplies of the primary and secondary centers of ossification until the two supplies coalesce at skeletal maturity.12 An orderly pattern of blood supply to the epiphysis occurs such that each penetrating vessel defines an autonomous vascular zone. Until this system matures, the femoral head is susceptible to patterned necrosis should any vascular territory suffer an occlusion.
Acetabular
The blood supply to the acetabular portion of the hip joint is completely defined before birth. The incidence of acetabular side avascular necrosis, either idiopathic or iatrogenic, is very low. From the internal iliac artery within the pelvis, the acetabular supply arises from three different branches, which arborize in such a way that they allow circumferential supply to the socket. The posterior division of the internal iliac terminates as the superior gluteal after giving off the iliolumbar and lateral sacral arteries. It supplies the superior portion of the acetabulum and the weight-bearing dome.13 Most of the inferior acetabulum is supplied by the posterior branch of the obturator artery. A small watershed area in the posterior acetabulum is redundantly supplied by the inferior gluteal artery. If the right acetabulum is seen as a clock face, superior and inferior gluteal arteries exit the pelvis at the greater sciatic notch at 10 o’clock. They diverge to supply an area superior and anterior (10-4 o’clock) and an area posterior (8-10 o’clock), respectively. The obturator artery exits the pelvis through the obturator foramen at 6 o’clock and supplies the acetabulum inferiorly (4-8 o’clock).14
Developmental Dysplasia of the Hip
Developmental dysplasia of the hip (DDH) is a disease process that affects primarily very young children and has the ability to interfere with early postnatal hip development. Its course is variable, ranging from full resolution with or without treatment to frank hip dislocation; therefore its true incidence is unknown.15 An intimate dynamic relationship between the spherical femoral head in the reciprocal acetabulum is crucial for appropriate postnatal development of a congruent hip joint. The primary disturbance in DDH is loss of femoral head congruency within the acetabulum, resulting in abnormal development of the femoral head and acetabular concavity.16 As many as 95% of cases of subluxation or frank dislocation can be successfully treated with conservative abduction devices such as the Pavlik harness.17 However, intra-articular pathology such as hypertrophied acetabular cartilage (neolimbus) or accessory centers of ossification may develop concurrently.18–20 If the young hip has been dislocated for any length of time, pressure against the ilium, superior to the actual acetabulum, may lead to the formation of a false acetabulum, the presence of which markedly increases the risk of future osteoarthritis.21 If persistent, these changes may require operative management, including closed reduction and casting, open reduction with or without femoral shortening, a variety of acetabuloplasties, and ultimately total hip arthroplasty. “Successful” treatment of DDH carries a risk of later avascular necrosis, which, depending on severity and on the age of the patient, may result in radiographic features that challenge the adult arthroplasty surgeon and may mimic changes seen in Legg-Calvé-Perthes disease.22
Initial radiographic findings in the infant with DDH include delayed presentation of the ossific nucleus, increased acetabular index for age, poorly formed teardrop or sourcil, a break in Shenton’s line, and the presence of an acetabular notch and a false acetabulum23 (Fig. 14-9). Often, the earliest radiographic finding in DDH with subluxation is increased sclerosis in the weight-bearing dome at the point of greatest contact. Osteoblastic activity is thought to be activated in response to increased forces born in this area, resulting in sclerosis.24 A definite association of dysplasia with roentgenographic degenerative joint disease has been noted, especially in females.10,15 Several other radiographic findings unique to DDH evolve over time. Children with dysplastic hips fall into a broad continuum of congruency at skeletal maturity. Moderate to severe arthritis is not seen in Severin I hips but is evident in 22% of Severin II, 35% of Severin III, and 76% of hips that are subluxated or dislocated at maturity25 (Table 14-2). These hips were thought to have valgus proximal femurs related to selective lateral growth arrest, which does not manifest radiographically until the adolescent growth spurt. 25 However, more critical review suggests that persistent femoral anteversion gives the radiographic appearance of increased valgus (>135 degrees), and that the true incidence of femoral valgus in DDH is no greater than that in the general population.22
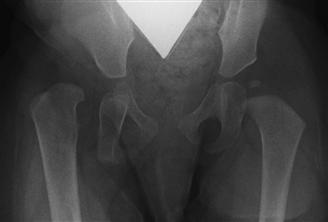
Figure 14-9 Anteroposterior (AP) radiograph of a dysplastic right hip. On the left, the acetabular sourcil is well defined and sclerotic. An ossific nucleus (secondary center of ossification at the femoral epiphysis) is present, along with a developing teardrop. Shenton’s line (a continuation of the curvature of the medial femoral neck as it relates to the inferior aspect of the superior pubic ramus) is intact. In the right, dysplastic hip, these features are absent.
Table 14-2
Severin Classification of Hip Dysplasia
| Class | Radiographic Appearance | Center Edge Angle, degrees |
| I | Normal | |
| IA | >19 (6-13 yo), >25 (>14 yo) | |
| IB | 15-19 (6-13 yo), 20-25 (>14 yo) | |
| II | Moderate femoral head; neck and acetabular deformity | |
| IIA | >19 (6-13 yo), >25 (>14 yo) | |
| IIB | >15-19 (6-13 yo), 20-25 (>14 yo) | |
| III | Dysplasia without subluxation | <15 (6-13 yo), <20 (>14 yo) |
| IVA | Moderate subluxation | >0 |
| IVB | Severe subluxation | <0 |
| V | Femoral head pseudoarticulation with acetabulum | |
| VI | Redislocation |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








