CHAPTER 79 Late Decompression of Patients with Spinal Cord Injury
Nonoperative care was the earliest standard treatment modality for patients with spinal cord injury. This approach was superseded for a time by delayed surgery on the basis of data that suggested nonoperative treatment, or early surgery, did not reliably improve neurologic outcome and may be associated with increased risks of complications.1–17 With improvements in imaging techniques18,19 and our understanding of the primary and secondary mechanisms of spinal cord injury,12,17,20–27 there is increasingly greater emphasis on early pharmacologic and surgical intervention.28–44
According to the National Acute Spinal Cord Injury Study (NASCIS) III, methylprednisolone may improve outcome if given as a loading dose of 30 mg/kg over 15 minutes and continued at 5.4 mg/kg/hr for 24 hours in acute nonpenetrating spinal cord injuries presenting at less than 3 hours and for 48 hours in injuries presenting at 3 to 8 hours.28,45,46 Preliminary data from further prospective studies are largely negative but are under way. Rabinowitz and colleagues47 reported surgical decompression is more effective for improving neurologic recover in beagle dogs than methylprednisolone alone.
Despite the lack of a clear definition of “early” surgery, the current consensus is that early decompression and stabilization of traumatic spinal cord injuries with cord compression provides an improved physiologic environment for neurologic recovery. Earlier concerns of increased complications with early surgery are unsupported. Surgical decompression should therefore be done as soon as the patient is medically stable. This more aggressive approach may improve neurologic recovery in some patients and decrease patient mortality and morbidity from pulmonary, skin, and gastrointestinal complications, as well as length of hospitalization and time to mobilization and rehabilitation in most patients.18,31–40,43,48–50 The Spine Trauma Study Group has defined early intervention as within 24 hours of the injury.51 Chen and colleagues52 found no statistical difference in outcomes between traumatic central cord syndrome (TCCS) patients treated operatively less than 4 days and greater than 4 days from injury. Clearly, timing of surgical intervention remains controversial.
As a result of improved acute care and rehabilitation, more patients with spinal cord injury are living longer and more productive lives; however, with increased longevity after traumatic spinal cord injuries, some patients with incomplete spinal cord lesions will present late with chronic cord compression and a neurologic status that has plateaued or is deteriorating. Experimental53,54 and clinical14,39,55–67 evidence suggests that these patients may receive improved cord function from decompression even as late as 9 years after spinal cord injury.15 Patients with complete cord lesions may also benefit from root recovery after late decompression.* Other late complications of traumatic spinal cord injury such as spasticity,70 syringomyelia,71–74 and Charcot arthropathy75–79 of the spine may cause neurologic deterioration or pain that may also benefit from late decompression.
Experimental Studies
An acute traumatic injury to the spinal cord may leave varying amounts of physiologically viable tissue in an incomplete lesion that may progressively deteriorate due to secondary injury.44,53,54,80–83 The amount of viable tissue that remains and the ability of the tissue to function partly depend on the primary mechanism of the injury such as the characteristics of the initial force, as well as on the secondary mechanisms of injury caused by the combination of elevated levels of intracellular calcium, endogenous opiates, and inflammatory mediators, lipid peroxidation, edema, glutamatergic excitotoxicity, and mechanical factors producing continued compression and cord ischemia.†
Several experimental models have been developed to determine how much injury the cord can tolerate and how long it can be compressed anteriorly and still recover neurologic function.‡ Allen,22 in 1911, developed a standardized weight-drop model to produce graded spinal cord lesions in vivo. Pathologic evaluation of the injured cords revealed central hemorrhage and progressive necrosis that was thought to be the underlying mechanism behind neurologic deterioration in these dogs. Some dogs regained the ability to ambulate after myelotomy and evacuation of the hemorrhagic tissue. Other animal experiments have subsequently been done using the weight-drop method, and others have used dynamic loading with a piston device or a graded constriction method using clips, constriction bands, or balloons.* Collectively, these experiments suggest that surgical decompression, preferably early but also late, may result in varying degrees of neurologic recovery.
†References 20, 21, 25, 27, 28, 46, 58, 82–85.
‡References 22, 39, 44, 53, 54, 58, 80, 81, 86–97.
The duration of compression and timing of decompression are not the only factors affecting the potential for improved neurologic function in these animal studies. Other factors such as force of the initial compression, amount of cord displacement, and the kinetic energy of the applied load affect the severity of the injury. Dolan and colleagues90 and Guha and colleagues92 used modified aneurysmal clips to constrict the T1 cord of rats with graded forces over different time periods. Clinical neurologic recovery varied exponentially according to the force of injury and linearly according to the duration of compression. They concluded that the major determinant of recovery was the initial force of injury. Although earlier decompression produced significantly more neurologic recovery, later decompression has the potential to salvage physiologically viable tissue that survived the initial injury and subsequent compression. Sommerson and Stokes98 showed that compression injuries with less than 1-mm deformation of the spinal cord in rodents result in linear displacement of the cord relative to the applied force. This linear relationship becomes nonlinear if the cord displacement exceeds 1 mm. The result of this study suggests the possibility of a similar relationship between a traumatic compressive lesion and neurologic deterioration. Late decompression may, therefore, prevent further neurologic deficit owing to increasing cord deformation from progressive deformity or instability. Kobrine and colleagues88,89 compressed the T6 spinal cord level in monkeys using an inflatable extradural balloon at different compression rates. Monkeys who had slow balloon inflation had return of function based on spinal cord evoked potentials even after several minutes of compression.88 In contrast, monkeys that had rapid balloon inflation and acute cord compression showed return of response in 1 hour if compression lasted for 1 minute but no recovery up to 1 hour if compression lasted for 3, 5, 7, or 15 minutes.89 Although this study can be criticized for not checking responses beyond 1 hour, the authors concluded that recovery of spinal cord function depended on the rapidity and length of time of compression. Therefore lower energy injuries or progressive deficits due to cord compression after an acute injury may have potential for neurologic recovery even with late decompression.
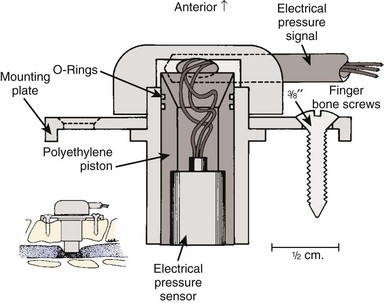
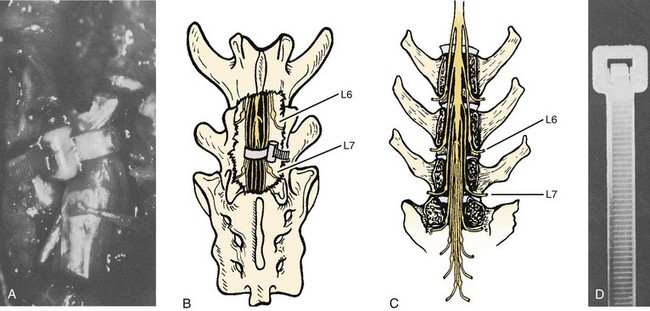
Bohlman and colleagues53,54 developed an animal model to demonstrate that late decompression of a traumatic incomplete spinal cord injury that occurred after either cord contusion or compression may result in neurologic recovery. Incomplete paralysis was produced in eight beagles by remote control of a pressure transducer (Figs. 79-1 to 79-4) and maintained for 3 to 8 weeks until recovery plateaued. After cord compression was relieved, all animals recovered completely by 10 weeks after injury. Recovery was faster in the five with complete decompression versus the three with lesser degree of ongoing compression from the transducer. Microscopic examination was normal in two dogs, central gray necrosis in two, peripheral demyelinization in two, and lacerations in three. The weight-drop method was used to contuse the cord in seven dogs using 257 to 722 gm/cm of energy. Variable levels of quadriparesis occurred, and all dogs recovered by 6 weeks after injury. Thirteen dogs developed central cord syndromes (forepaw paralysis greater than hindpaw) with either method. Central gray matter damage was the most common finding. The gray matter seemed more sensitive to deforming forces of external degeneration of the ascending sensory posterior columns, whereas others showed damage of the anterior and lateral corticospinal tracts, indicating descending wallerian degeneration. However, histopathologic damage of the spinal cord did not always correspond to the degree of clinical paralysis in these dogs. For example, there was normal histology in three dogs, two with quadriplegia for 1 to 6 weeks, and one with grade 3 paralysis. Because these dogs recovered neurologic function after contusion and compression, it appeared that deformation of the cord produced a physiologic block but the cord remained viable. In the contusion model, greater contusion forces caused proportionately increased neurologic damage and recovery time, whereas in the compression model anterior decompression allowed for faster and more complete recovery.
Clinical Studies
Although there are likely significant differences between the mechanical and biologic properties of the spinal cord in human versus animals, the animal studies suggest that the characteristics of the initial force and the amount and duration of cord compression alter neurologic recovery. Persistent compression may cause progressive mechanical injury and diminished blood flow to the cord in the region of the anterior spinal artery, thus preventing improvement with nonoperative treatment. Early decompression of incomplete cord injury restores blood flow and provides faster and more complete recovery, but late decompression can achieve varying degrees of the same result in cases of residual compression. Decompression in the region of the lesion in a patient with an incomplete spinal cord injury may provide functional recovery of motor roots and the spinal cord at that level and below. Patients with complete cord injuries are not expected to improve distal neurologic function from late decompression, but they may recover nerve roots near or above the level of injury and prevent ascending nerve root loss. The majority of clinical studies that have tried to determine the optimum timing of surgery for traumatic spinal cord injury are retrospective16,31–40,43,48,49 with only a few prospective studies.16,17,40,43,99 These studies have inconsistent definition of early versus late surgery that may have contributed to conflicting findings in which some studies found a benefit from early decompression and stabilization, whereas others found no advantage. However, if 4 weeks is considered late, there are studies, albeit retrospective, that have shown reversal of neurologic deficits in incomplete lesions.*
Cervical Spine Lesions
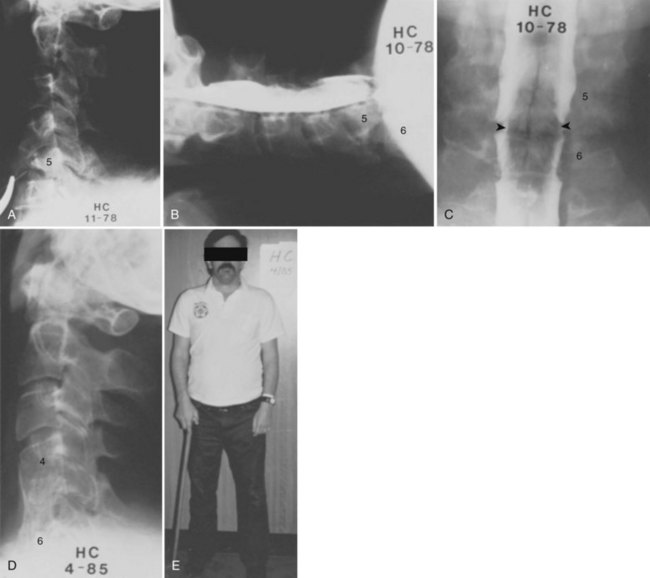
Bohlman and colleagues15,55 demonstrated that later anterior decompression can result in reversible neurologic function in patients with incomplete cord injuries (Fig. 79–5). In one study, 58 patients with incomplete cord injuries underwent late anterior cervical decompression and fusion with iliac crest autograft an average of 13 months (range, 1 month to 9 years) from injury. Twenty-nine patients who could not walk preoperatively became functional ambulators, and six patients who could walk before surgery had improvement in the ability to walk. Thirty-nine patients had improvement in motor root function in the upper extremities. Improvement was less in the patients who had decompression more than 12 months after injury.
In a similar study by Benzel and Larson,61 41 of 51 (81%) patients with incomplete myelopathies and whose neurologic function had plateaued had improved spinal cord function after decompression and fusion as late as 115 and 173 days in 2 patients. Among eight patients with motor complete lesions preoperatively with no useful function, six regained useful function, with four able to walk with assistance. The return of neurologic function in these patients began promptly after decompression, suggesting a cause-and-effect relationship. These studies suggest that late anterior decompression and stabilization can result in reversible neurologic deficits of both upper and lower extremity roots and distal cord function in the lower extremities.
Reduction of pain and restoration of even a single functional nerve root level may greatly improve the quality of life in complete quadriplegic patients. Return of sensory perception improves the chances of preventing pressure sores, and arm functional recovery of one or more levels may allow independent ambulation. Several studies38,39,55–57,61,62,69 have shown that these are realistic goals even when decompression is done as late as after 21 years.62 Patients with complete cord injuries have far less predictable and dramatic recovery. The mechanisms of recovery are poorly understood, but it appears that the injury falls along a continuum of an incomplete lesion where spinal cord fibers are anatomically but not functionally intact. Dimitrijevic101 proposed the term discomplete quadriplegia to describe this concept. Multiple factors including persistent compression, instability, and progressive deformity may diminish the healing environment of the cord and cause secondary injury. Nevertheless, decompression appears to have a limited but positive effect on recovering function of remaining viable motor roots and cord tissue.
Anderson and Bohlman69 performed anterior cervical decompression and fusion with iliac crest autograft in 51 patients with complete motor quadriplegia in an attempt to gain improvements of the motor roots in the upper extremities and thereby to improve function. Five patients died in less than 1 year. The average time from injury to decompression was 15 months (range, 1 month to 8 years), and 46 patients were followed for an average of 5 years (range, 2 to 13 years). Neurologic improvement of at least two new functional motor root levels occurred in 7 patients, and 18 patients had one-level recovery. Six patients had increased motor strength. Therefore anterior decompression resulted in neurologic improvement in 67% of the patients. Results were better in patients younger than age 53 years, those who had decompression less than 18 months after injury, and those who had neurologic levels cephalad to the site of skeletal injury rather than those with functioning motor roots at or caudad to the level of injury.
Benzel and Larson61 reported on 35 patients with complete quadriplegia who also underwent late surgery to improve neurologic function. Twenty-five patients underwent anterior decompression with or without posterior fusion and foraminotomy, whereas 10 had posterior fusion alone. Recovery of nerve root function only occurred in the patients who had anterior decompression (15 of 35 patients). No patient with both motor and sensory complete lesions recovered long tract function. However, of the 11 patients with motor injuries and sensory perception, 5 improved significantly and 2 were able to walk. It is highly unusual to have return of long tract motor function in patients with motor complete injuries, but it highlights the potential for late decompression to allow the cord maximal opportunity to recover neurologic function.
Thoracolumbar and Cauda Equina Lesions
Benzel and Larson61 described the results of anterior decompression and fusion via the lateral extracavity approach in 105 patients with thoracic and lumbar spinal cord injuries. There was significant improvement in the neurologic status of patients with incomplete lesions. Four of 10 patients with motor complete injuries with some sensory perception improved neurologically, with one able to ambulate. The anterior approach was deemed superior to a nonoperative or posterior stabilization approach for anterior compressive lesions.
Similarly, Maiman and colleagues56 reported the cases of 20 patients with incomplete thoracolumbar spinal cord injuries who underwent late anterior decompression via a lateral extracavity approach. Operations were done on average at 23 months and as late as 5 years after the initial injury or prior surgery. Thirteen patients had a mass in the canal, and seven patients had kyphotic deformities. Seventeen patients had substantial neurologic improvement. All but three of the nonambulatory patients with canal masses and the seven patients with kyphosis became ambulatory. The authors concluded that anterior decompression with appropriate stabilization affords the best opportunity for neurologic improvement in thoracolumbar spinal cord injuries.
Injuries to the cauda equina and conus medullaris with ongoing compression may also benefit from late decompression. The peripheral nerve roots of the cauda equina are fairly resilient, and conus medullaris injuries have significant potential for neurologic recovery, especially bladder control after decompression.57,59 Bradford and McBride64 studied 59 patients with incomplete deficits after thoracolumbar and cauda equina injuries. Eighteen patients who had anterior decompression were treated an average of 15.8 months (range, 1 to 79 months) after their injury. Anterior decompression was shown to be superior to posterior decompression, with 88% versus 64% neurologic improvement, respectively.
Although laminectomy is rarely indicated in spinal cord injuries, Ramamurthi and colleagues66 observed improvement in bladder function in 14 of 27 patients and motor function in 8 of 27 patients who had decompressive laminectomy between 6 and 12 weeks after conus and cauda equina injuries. Compression was due to displaced lamina, subarachnoid adhesions after traumatic fractures involving T12 to L4.