CHAPTER 14 Intraoperative Neurophysiologic Monitoring of the Spine
Intraoperative Monitoring of the Spinal Cord
Somatosensory-evoked potential (SEP) monitoring has been used for many years to monitor spinal function intraoperatively during various surgeries involving the spine, such as corrective surgery for scoliosis or other congenital deformities and removal of intraspinal tumors or arteriovenous malformations. SEP monitoring has been shown to reduce the incidence of neurologic damage in large studies of experienced monitoring teams.1 SEPs monitor only sensory transmission through the dorsal column pathways, however; SEPs do not provide a direct measure of motor function. In addition, the dorsal columns receive their blood supply from the posterior spinal arteries, whereas the anterior spinal arteries supply the motor pathways. Ischemic damage to the spinal cord from the anterior spinal artery may be undetectable with SEP monitoring.2,3
MEPs are a more direct technique that evaluates the motor pathway. Monitoring has been previously performed by relying on stimulation of the spinal cord directly.4 Spinal cord stimulation can be done with the use of epidural electrodes inserted after a laminectomy has been performed or by percutaneous intraspinous needle electrodes. The epidural electrodes are invasive and often require placement by a skilled anesthesiologist. Percutaneous intraspinous needles are difficult to place accurately and may not achieve adequate or consistent stimulation of the spinal cord. In addition, there is the question of whether MEPs generated through spinal cord stimulation arise solely from propagation through the motor pathway or if multiple pathways are involved in their generation.5,6 There are reports of MEP monitoring in which spinal cord stimulation resulted in no significant intraoperative changes despite postoperative neurologic motor deficits (so-called false-negative result).7 It is now believed that motor cortex stimulation with transcranial electrical stimulation provides a more reliable method for monitoring of the motor pathways. This technique is now routinely used in spinal cord monitoring together with SEPs.
Somatosensory-Evoked Potential Monitoring
The use of SEPs in intraoperative monitoring of complex spine surgeries began in the early 1970s.8 Although SEP monitoring evaluates primarily the integrity of the posterior columns, it is often used to give an overall assessment of the spinal cord based on the assumption that many intraoperative mechanisms of injury would affect the spinal cord diffusely. An example of such an injury is spine distraction during scoliosis surgery. In addition, ischemic injury may initially result in a more diffuse dysfunction of the spinal cord that could be detected by SEPs (Fig. 14–1). SEP responses are thought to pass through large fiber somatosensory pathways of the dorsal column and possibly through the anterior spinothalamic tract, and this may be another reason why anterior spinal artery ischemia could be detected using this technique.
Generators of Somatosensory-Evoked Potential Responses
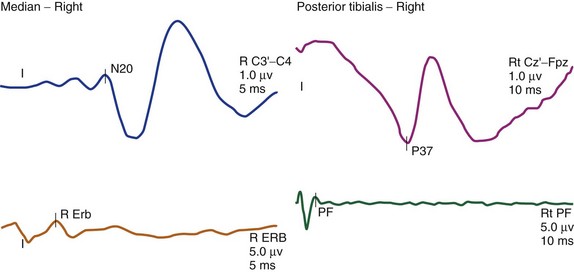
The cortical response for the lower extremity is called the P37 potential. The generator of this response arises from the primary somatosensory cortex of the leg, which is located in the mesial parietal cortex. The cortical response for the upper extremity, which is generated from the primary somatosensory cortex of the hand, is called the N20 potential (Fig. 14–2). Two important characteristics of these waveforms are (1) amplitude, which is recorded in microvolts and determined by either a baseline-to-peak or a peak-to-trough measure of the waveform, and (2) latency, which is recorded in milliseconds and is the time interval from the stimulus to the occurrence of the potential. An amplitude change from the initial baseline measure to a decrease of more than 50% is often considered a significant change in SEP amplitudes.9
Significant latency changes in SEP monitoring consist of a 10% prolongation beyond the baseline latency value (Table 14–1).10 Although these deviations from the baseline measures are thought to be significant, they should be interpreted with caution, taking into account various factors, including the evolution of the changes (e.g., a trend toward worsening is an ominous sign) and other intraoperative factors such as length of the surgery, type of anesthetic agent, and temperature effects. Also, significant latency and amplitude changes can occur in isolation. It is quite common to see a significant amplitude change without any associated latency changes. The most significant change is a complete loss of the cortical potential.
TABLE 14–1 Significant Changes in Different Monitoring Modalities
| Type of Study | Significant Changes | Highly Significant Changes |
|---|---|---|
| Somatosensory-evoked potentials | Amplitude <50%; latency >10% | Complete loss of amplitude |
| Motor-evoked potentials | Increase threshold voltage >50-100 V | Complete loss of amplitude |
| Pedicle screw stimulation | Current intensity <7-10 mA |
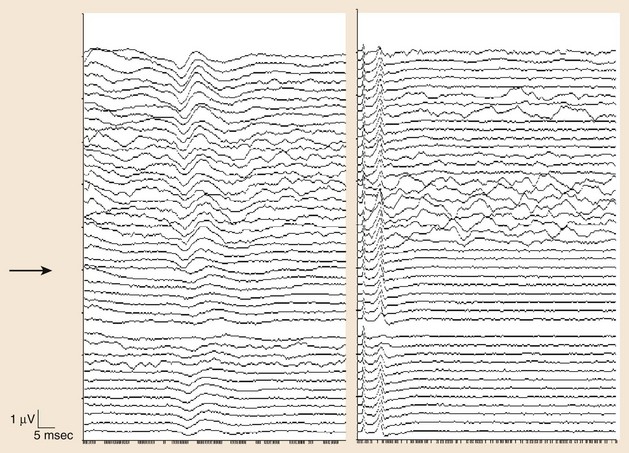
Another measurement made in posterior tibial or peroneal nerve SEP monitoring is the popliteal fossa potential. This is a nerve action potential that is recorded as the impulses pass within the popliteal fossa in the peripheral nervous system. The reason for this measurement is to ensure that an adequate stimulus has been applied. If there is an absence of the popliteal fossa response in addition to an absence of the leg cortical (P37) response, it suggests that the changes seen are not a result of a lesion at the level of the spinal cord. In this case, the change may be technical (e.g., the stimulating needles may have dislodged) or may be seen when the leg is ischemic (e.g., with femoral artery catheterization during thoracoabdominal aneurysm surgery or with direct compression of the peripheral nerve) (Fig. 14–3).
The other posterior tibial stimulation SEP response that can be monitored (besides the P37 response) is the P31/N34 complex, often termed the subcortical response because the generator for this response is at the level of medulla and midbrain. The benefit in recording these responses is that they are relatively more resistant to the effects of anesthesia compared with the cortical P37 response (see Fig. 14–3). The same is true for the subcortical potentials from median nerve stimulation (P14/N18) potential. In pediatric cases, the subcortical potentials may also be better formed and more easily monitored than cortical responses. Some of this effect may be the result of the variation of myelination in the younger age groups and the more significant effects of anesthetics on these patients. These differences from the adult morphology can persist into the early teenage years. Other factors affecting the responses include core body temperature changes. The core body temperature commonly decreases more than 1° C. The cooling affects the limbs more than the core body temperature, which can result in slowing of conduction.
Motor-Evoked Potential Monitoring
Noninvasive stimulation of the brain using TCES was first reported in 1980.11 Single-pulse TCES was subsequently used in monitoring motor pathways.12–17 Because of effects of general anesthesia, single-pulse stimulation was found to be less effective in reliably recording MEPs.18–22 With the introduction of the multipulse technique for motor pathway monitoring, reliable and robust MEP recording can now be obtained in most patients using some specific general anesthesia protocols.23–27 The use of the multipulse technique requires that neuromuscular blockade not be used during this part of the monitoring. Occasionally, the use of partial neuromuscular blockade may still allow for TCES monitoring.28 This method reportedly achieves more reliable stimulation of the motor cortex intraoperatively and is more resilient to the effects of general anesthesia.
The method of MEP monitoring has been revolutionized by the use of multipulse TCES. Previous methods for MEP recording used a variety of stimulation and recording techniques. Spinally elicited neurogenic responses were used in the past and were putatively stated to be a result of activation of the motor pathways in the spinal cord. More recent evidence has suggested that these spinally elicited neurogenic responses are generated through activation of the sensory pathways and retrograde activation of alpha motor neurons. In a collision experiment using stimulation of the spinal cord followed by stimulation of the posterior tibial nerve at various interstimulus intervals, the neurogenic responses were abolished, suggesting that the potentials were colliding in the spinal cord.6
At the beginning of TCES-MEP monitoring, threshold voltages for each side of the body and MEP amplitudes are calculated.23 The motor cortex on the side of the brain receiving the anodal stimulus is typically the first region to activate at the lowest stimulus threshold. The initial current used is typically 100 V, with a train of stimuli delivered to the cortex. After the stimulation, an MEP response is monitored in the muscles contralateral to the side receiving the anodal stimulus. If no response is seen, the voltage is increased typically by 50-V increments, and the process is repeated until an MEP response is seen in all the muscles contralateral to the anodal stimulus. This voltage is called the threshold voltage for that side.
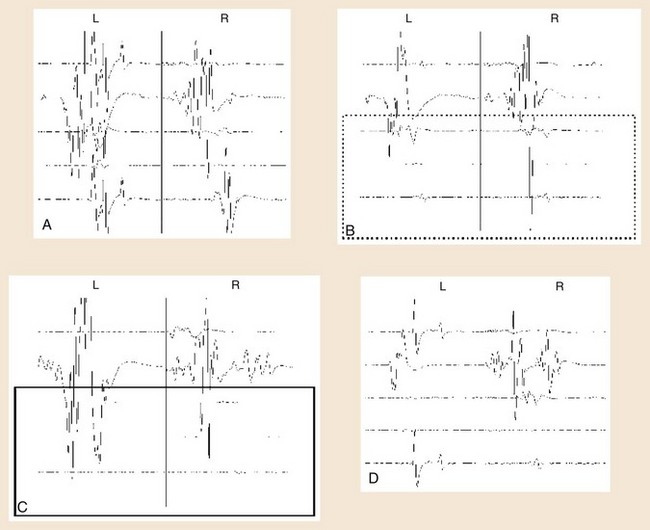
Determining significant changes during the course of surgery typically is most reliable if there is an absolute loss of myogenic responses to stimulation (Fig. 14–4). Some authors have also suggested that reduction of MEP amplitude to 25% of baseline amplitude values is predictive of motor pathway impairments.29 Significant changes can also be determined by the change of voltage required to obtain MEPs of greater than 50 V beyond baseline thresholds used in obtaining MEPs at the beginning of monitoring (see Table 14–1).30
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree