Chapter 4 Hip Resurfacing
Preoperative Radiographic Evaluation and Templating
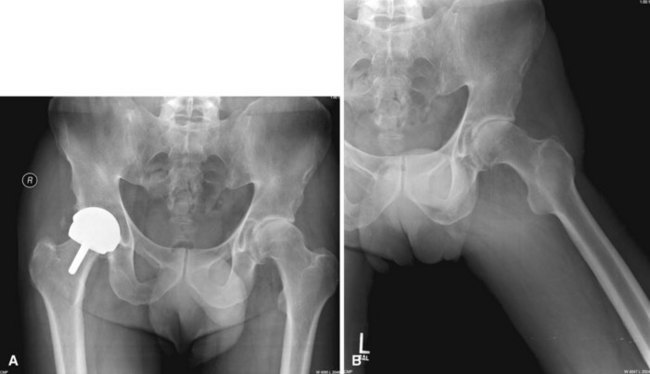
As with all arthroplasty hip surgery, preoperative templating, radiographic evaluation, and planning are critical to success. If a patient’s anatomy is distorted by previous surgery, injury, or deformity, he may not be a candidate for hip resurfacing. The femoral head and neck bone quality should be normal, and there should not be more than 25% to 30% of the head involved with avascular necrosis or cyst formation as seen on radiographs. If the femoral neck is enlarged by remodeling, there may not be a clear delineation between the head and neck, with the head being larger than the neck (Fig. 4-1). If the neck and head are of the same width, especially along the superior neck as seen on an anteroposterior radiograph, then removing bone from the head will risk notching the femoral neck and thus risk neck fracture.
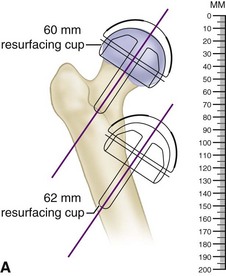
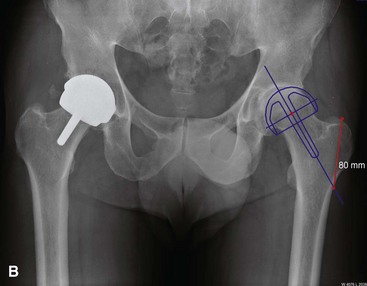
The first step in templating is to measure the size of the femoral component. A template is laid over a radiograph of the proximal femur. The width of the opening of the femoral component should be wider than the femoral neck by 2 to 4 mm total. If not, the next larger template should be used. Then, the center post of the implant is aligned over the center of the femoral neck on radiograph. The line from the top of the greater trochanter to where the line on the template intersects the lateral cortex is measured and documented (Fig. 4-2A). This distance will be used when measuring the valgus angle of the implant intraoperatively (Fig. 4-2B).
Technique
Hip Resurfacing Posterolateral Position
 Position the patient in the lateral position with the affected hip up. Stabilize the pelvis with a pelvic clamp or pegboard, with the pelvis oriented straight up and down. If the pelvis is leaning forward, the acetabular component may be placed in retroversion; and if it is leaning backward, the acetabular component may be placed in excessive anteversion.
Position the patient in the lateral position with the affected hip up. Stabilize the pelvis with a pelvic clamp or pegboard, with the pelvis oriented straight up and down. If the pelvis is leaning forward, the acetabular component may be placed in retroversion; and if it is leaning backward, the acetabular component may be placed in excessive anteversion.
Approach and Exposure
 To resurface the hip, extensive exposure is necessary to allow the acetabulum to be visible and later on in the procedure to keep the femoral head visible over its entire surface. Therefore, steps must be taken to achieve exposure not commonly used in total hip replacement surgery. Obviously, the femoral head is removed during a total hip replacement, which greatly aids in exposure.
To resurface the hip, extensive exposure is necessary to allow the acetabulum to be visible and later on in the procedure to keep the femoral head visible over its entire surface. Therefore, steps must be taken to achieve exposure not commonly used in total hip replacement surgery. Obviously, the femoral head is removed during a total hip replacement, which greatly aids in exposure.
 Make a curved skin incision over the greater trochanter, angling the proximal portion posteriorly, pointing toward the posterior superior iliac spine (Fig. 4-3A). Carry the incision over the center of the greater trochanter and then distally over the shaft of the femur to end over the attachment of the gluteus maximus on the linea aspera.
Make a curved skin incision over the greater trochanter, angling the proximal portion posteriorly, pointing toward the posterior superior iliac spine (Fig. 4-3A). Carry the incision over the center of the greater trochanter and then distally over the shaft of the femur to end over the attachment of the gluteus maximus on the linea aspera.
 Divide the subcutaneous tissue in a single plane over the fascia of the gluteus maximus proximally and the fascia of the iliotibial band distally. Make a longitudinal incision over the middle to posterior third of the fascia over the greater trochanter and extend it distally over the femoral shaft. Extend the proximal end of the incision through the thin fascia over the gluteus maximus in the same direction as the skin incision. Bluntly split the fibers of the gluteus maximus muscle, taking care to find and cauterize any bleeding.
Divide the subcutaneous tissue in a single plane over the fascia of the gluteus maximus proximally and the fascia of the iliotibial band distally. Make a longitudinal incision over the middle to posterior third of the fascia over the greater trochanter and extend it distally over the femoral shaft. Extend the proximal end of the incision through the thin fascia over the gluteus maximus in the same direction as the skin incision. Bluntly split the fibers of the gluteus maximus muscle, taking care to find and cauterize any bleeding.
 Release the tendinous attachment of the gluteus maximus from the linea aspera to maximally internally rotate the femur to provide satisfactory exposure of the proximal femur and femoral head. If the gluteus maximus is not released, the sciatic nerve may be at risk of compression at the time of preparation of the femoral head. Place a hemostat under the gluteus maximus tendon as the tendon is divided to avoid injuring branches of the medial femoral circumflex artery and the first perforating artery. Leave a centimeter of tendon attached to the linea aspera and femoral shaft for later repair.
Release the tendinous attachment of the gluteus maximus from the linea aspera to maximally internally rotate the femur to provide satisfactory exposure of the proximal femur and femoral head. If the gluteus maximus is not released, the sciatic nerve may be at risk of compression at the time of preparation of the femoral head. Place a hemostat under the gluteus maximus tendon as the tendon is divided to avoid injuring branches of the medial femoral circumflex artery and the first perforating artery. Leave a centimeter of tendon attached to the linea aspera and femoral shaft for later repair.
 Widely spread the fascial plane just divided using a Charnley or self-retaining retractor. The posterior greater trochanter and gluteus medius should be easily seen. Remove the trochanteric bursa.
Widely spread the fascial plane just divided using a Charnley or self-retaining retractor. The posterior greater trochanter and gluteus medius should be easily seen. Remove the trochanteric bursa.
 Retract the gluteus medius muscle and tendon anteriorly. A hooked instrument such as a Hibbs retractor is useful. Under the gluteus medius is the piriformis, which is exposed. Tag the piriformis tendon with suture, and then release it from the femur. Under and anterior to the piriformis tendon are the muscle fibers of the gluteus minimus. With an elevator, raise the gluteus medius off the capsule of the hip completely. The entire capsule of the hip should be exposed superiorly. Use of a narrow cobra retractor is helpful to see this area when it is placed under the gluteus minimus and medius.
Retract the gluteus medius muscle and tendon anteriorly. A hooked instrument such as a Hibbs retractor is useful. Under the gluteus medius is the piriformis, which is exposed. Tag the piriformis tendon with suture, and then release it from the femur. Under and anterior to the piriformis tendon are the muscle fibers of the gluteus minimus. With an elevator, raise the gluteus medius off the capsule of the hip completely. The entire capsule of the hip should be exposed superiorly. Use of a narrow cobra retractor is helpful to see this area when it is placed under the gluteus minimus and medius.
 Expose the plane distally between the capsule and the short external rotator muscles. Release the short external rotator muscles off the femur including the quadratus femoris distally. Coagulate the vessels in this area.
Expose the plane distally between the capsule and the short external rotator muscles. Release the short external rotator muscles off the femur including the quadratus femoris distally. Coagulate the vessels in this area.
 The capsule of the hip is now completely exposed posteriorly, superiorly, and inferiorly. The lesser trochanter also is visible. With the hip in internal rotation, make an incision in the capsule circumferentially, leaving at least a centimeter of capsule still attached to the femoral neck. This centimeter of capsule is later used to repair the capsule back as well as to provide protection to the intraosseous vessels needed to maintain vascularity of the femoral neck.
The capsule of the hip is now completely exposed posteriorly, superiorly, and inferiorly. The lesser trochanter also is visible. With the hip in internal rotation, make an incision in the capsule circumferentially, leaving at least a centimeter of capsule still attached to the femoral neck. This centimeter of capsule is later used to repair the capsule back as well as to provide protection to the intraosseous vessels needed to maintain vascularity of the femoral neck.
 Make two radial incisions in the posterior capsule to create a posterior capsular flap. This is helpful for retraction and later repair (Fig. 4-3B).
Make two radial incisions in the posterior capsule to create a posterior capsular flap. This is helpful for retraction and later repair (Fig. 4-3B).
 Dislocate the femoral head and perform a complete anterior capsulotomy with sharp scissors. The inferior portion of the capsule is seen by extending and internally rotating the femur. The psoas tendon is exposed at the lesser trochanter, and the capsule is isolated just in front of the psoas tendon. While maintaining the scissors just posterior to the psoas tendon, incise the capsule from inferior to superior (Fig. 4-3C). Maintain the femur in internal rotation and apply anterior traction with a bone hook on the lesser trochanter.
Dislocate the femoral head and perform a complete anterior capsulotomy with sharp scissors. The inferior portion of the capsule is seen by extending and internally rotating the femur. The psoas tendon is exposed at the lesser trochanter, and the capsule is isolated just in front of the psoas tendon. While maintaining the scissors just posterior to the psoas tendon, incise the capsule from inferior to superior (Fig. 4-3C). Maintain the femur in internal rotation and apply anterior traction with a bone hook on the lesser trochanter.
 Perform the proximal end of the capsulotomy by flexing the femur 90 degrees and maintaining a narrow cobra retractor under the gluteus muscles. Incise the capsule with sharp scissors while internally rotating the femur to beyond 100 degrees. If a complete capsulotomy is not performed, exposure of the femur is compromised.
Perform the proximal end of the capsulotomy by flexing the femur 90 degrees and maintaining a narrow cobra retractor under the gluteus muscles. Incise the capsule with sharp scissors while internally rotating the femur to beyond 100 degrees. If a complete capsulotomy is not performed, exposure of the femur is compromised.
 Measure the femoral neck from superior to inferior, its longest dimension (Fig. 4-3D). The Birmingham Hip comes with heads in 2-mm increments. The measurement tool should loosely fit over the femoral neck to avoid undersizing the femoral component, which could cause notching of the femoral neck. Femoral neck notches may weaken the neck and predispose it to early postoperative fracture. If there is any doubt, choose the next larger size of the femoral head component.
Measure the femoral neck from superior to inferior, its longest dimension (Fig. 4-3D). The Birmingham Hip comes with heads in 2-mm increments. The measurement tool should loosely fit over the femoral neck to avoid undersizing the femoral component, which could cause notching of the femoral neck. Femoral neck notches may weaken the neck and predispose it to early postoperative fracture. If there is any doubt, choose the next larger size of the femoral head component.
 Once the size of the femoral component is known, the acetabular component size also is known because the acetabular component is matched with components either 6 or 8 mm larger than the femoral component. So, if the femoral head measures 52 mm, the acetabular component will need to be either 58 or 60 mm. That means (in this case) the acetabulum will need to be reamed to 57 or 59 mm, respectively.
Once the size of the femoral component is known, the acetabular component size also is known because the acetabular component is matched with components either 6 or 8 mm larger than the femoral component. So, if the femoral head measures 52 mm, the acetabular component will need to be either 58 or 60 mm. That means (in this case) the acetabulum will need to be reamed to 57 or 59 mm, respectively.
 The key to exposure of the acetabulum is to dislocate the femoral head out of the way anteriorly and superiorly. Create an anterosuperior pouch large enough for the femoral head under the gluteus muscles and above the ilium. This is done by sharply dissecting the soft tissues off the bone of the ilium, including the capsule and tendons of the rectus femoris from the superior acetabular lip and the anterior inferior iliac spine.
The key to exposure of the acetabulum is to dislocate the femoral head out of the way anteriorly and superiorly. Create an anterosuperior pouch large enough for the femoral head under the gluteus muscles and above the ilium. This is done by sharply dissecting the soft tissues off the bone of the ilium, including the capsule and tendons of the rectus femoris from the superior acetabular lip and the anterior inferior iliac spine.
 Once the pouch has been created, dislocate the femoral head into the pouch under the gluteus muscles and retract it with a sharp, narrow Hohmann retractor driven into the ilium superior to the acetabulum and resting on the femoral neck (Fig. 4-3E). Additional pins may be driven into the ilium and ischium to help with the acetabular exposure. A retractor also is placed inferiorly to expose the transverse acetabular ligament. Sharply excise the labrum.
Once the pouch has been created, dislocate the femoral head into the pouch under the gluteus muscles and retract it with a sharp, narrow Hohmann retractor driven into the ilium superior to the acetabulum and resting on the femoral neck (Fig. 4-3E). Additional pins may be driven into the ilium and ischium to help with the acetabular exposure. A retractor also is placed inferiorly to expose the transverse acetabular ligament. Sharply excise the labrum.
 Ream the acetabulum medially through the cotyloid notch of the acetabulum to the medial wall. Take care not to ream through the medial wall. Once medialized, the reamers are used to increase the bony acetabulum to the desired size. The acetabulum usually is underreamed by 1 mm from the desired component size. Use an acetabular trial to assess the potential component’s stability. The trial components in the Birmingham Hip Resurfacing System are 1 mm smaller than their stated size to provide for tighter fitting of the actual component. Impact the trial into the acetabulum with a mallet, and excise osteophytes for unobstructed cup insertion (Fig. 4-3F and G
Ream the acetabulum medially through the cotyloid notch of the acetabulum to the medial wall. Take care not to ream through the medial wall. Once medialized, the reamers are used to increase the bony acetabulum to the desired size. The acetabulum usually is underreamed by 1 mm from the desired component size. Use an acetabular trial to assess the potential component’s stability. The trial components in the Birmingham Hip Resurfacing System are 1 mm smaller than their stated size to provide for tighter fitting of the actual component. Impact the trial into the acetabulum with a mallet, and excise osteophytes for unobstructed cup insertion (Fig. 4-3F and G
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree