Chapter 74 Compartment Syndromes and Volkmann Contracture
Anatomy
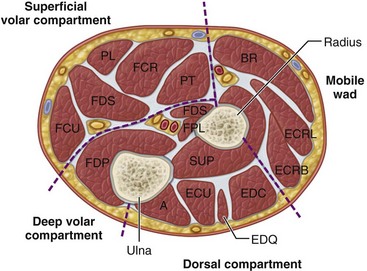
Four interconnected compartments of the forearm are recognized (Fig. 74-1): (1) the superficial volar compartment, (2) the deep volar compartment, (3) the dorsal compartment, (4) and the compartment containing the mobile wad of Henry (brachioradialis and extensor carpi radialis longus and brevis). The volar compartments are most commonly involved, but the dorsal and mobile wad compartments can be involved alone or in addition to the volar compartments. Clinically differentiating isolated or combined involvement of the deep and superficial volar compartments usually is difficult; however, the deep volar compartment (flexor digitorum profundus, flexor pollicis longus, and pronator quadratus) may be solely involved.
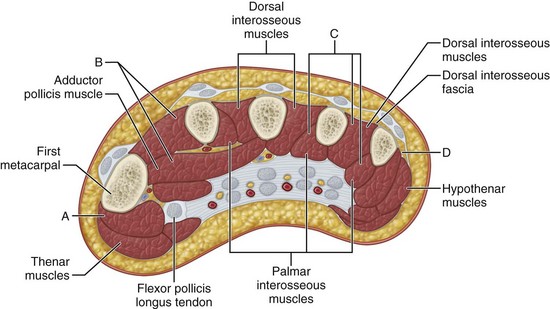
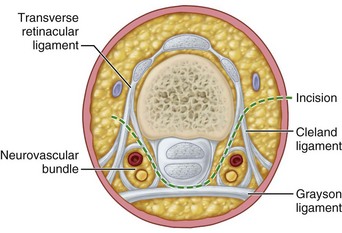
In the hand, each interosseous muscle is surrounded by a tough investing fascial layer, each making an individual compartment as shown by the injection dissections of Halpern and Mochizuki. The adductor pollicis muscle and the thenar and hypothenar muscles form three separate compartments (Fig. 74-2). The neurovascular bundles of each digit also are compartmentalized by fascial layers, making them vulnerable to excessive swelling (Fig. 74-3).
Etiology
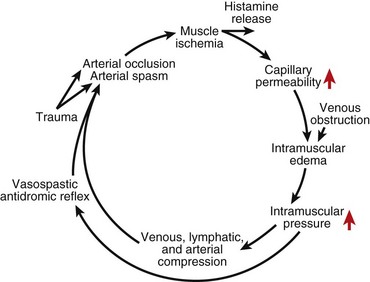
Any situation that causes a decrease in compartment size or increase in compartment pressure can initiate compartment syndrome. As the intracompartmental pressure increases, capillary blood perfusion is reduced to a level that cannot maintain tissue viability. The increase in interstitial pressure overcomes the intravascular pressure of the small vessels and capillaries, causing the walls to collapse and impeding local blood flow. In a canine model, muscle necrosis was shown to occur with a rise in pressure to within 20 mm below diastolic pressure. The local tissue ischemia leads to local edema, which increases intracompartmental pressure. This cycle of increasing muscle ischemia was depicted by Eaton and Green as shown in Figure 74-4.
Diagnosis
Measuring Compartment Pressures in the Forearm and Hand Using a Hand-Held Monitoring Device
Measuring Forearm Compartment Pressure
 Place the compartment to be measured at heart level.
Place the compartment to be measured at heart level.
 Use adequate local analgesia infiltrated into the skin only and avoiding underlying muscle and fascia to control discomfort and pressure spikes.
Use adequate local analgesia infiltrated into the skin only and avoiding underlying muscle and fascia to control discomfort and pressure spikes.
 To measure volar compartment pressure, insert the needle just ulnar to the palmaris longus, to a depth of 1 to 2 cm. Confirm proper needle depth by observing a rise in pressure during external compression of the volar forearm or passive extension of fingers.
To measure volar compartment pressure, insert the needle just ulnar to the palmaris longus, to a depth of 1 to 2 cm. Confirm proper needle depth by observing a rise in pressure during external compression of the volar forearm or passive extension of fingers.
 To measure the dorsal compartment, insert the needle just radial to the border of the ulna to a depth of 1 to 2 cm. Confirm placement by external compression of the dorsal compartment with passive flexion of the wrist.
To measure the dorsal compartment, insert the needle just radial to the border of the ulna to a depth of 1 to 2 cm. Confirm placement by external compression of the dorsal compartment with passive flexion of the wrist.
 To test the mobile wad, identify the radial most portion of the forearm and insert the needle perpendicular to the skin to a depth of 1 to 1.5 cm. A rise in pressure is identified by external pressure or passive flexion of the wrist.
To test the mobile wad, identify the radial most portion of the forearm and insert the needle perpendicular to the skin to a depth of 1 to 1.5 cm. A rise in pressure is identified by external pressure or passive flexion of the wrist.
Measuring hand compartment pressure
 Insert the needle perpendicular to the skin.
Insert the needle perpendicular to the skin.
 Evaluate the compartments individually. Pressure measurements are not obtained from the digits but at the site of maximal swelling of the thenar, hypothenar, and interosseous compartments.
Evaluate the compartments individually. Pressure measurements are not obtained from the digits but at the site of maximal swelling of the thenar, hypothenar, and interosseous compartments.
 If a single compartment pressure is elevated, release all compartments and the carpal tunnel.
If a single compartment pressure is elevated, release all compartments and the carpal tunnel.
 To measure the dorsal interosseous compartment pressure, insert the needle through the dorsal hand 1 cm proximal to the metacarpal head until it rests in the muscle belly. To judge the depth it is helpful to place identifiable marks on the needle at 1.0, 1.5, and 2.0 cm.
To measure the dorsal interosseous compartment pressure, insert the needle through the dorsal hand 1 cm proximal to the metacarpal head until it rests in the muscle belly. To judge the depth it is helpful to place identifiable marks on the needle at 1.0, 1.5, and 2.0 cm.
 To measure the adductor pollicis compartment pressure, insert the needle on the radial side of the second metacarpal in the substance of the thumb-index web space.
To measure the adductor pollicis compartment pressure, insert the needle on the radial side of the second metacarpal in the substance of the thumb-index web space.
 To measure the thenar and hypothenar spaces, insert the needle at the junction of the glabrous and nonglabrous skin over the maximal bulk of the muscle compartment. Advance the needle at least 5 mm below the enveloping fascia for pressure assessment.
To measure the thenar and hypothenar spaces, insert the needle at the junction of the glabrous and nonglabrous skin over the maximal bulk of the muscle compartment. Advance the needle at least 5 mm below the enveloping fascia for pressure assessment.
Management
Acute Compartment Syndrome of the Forearm
Forearm Fasciotomy and Arterial Exploration
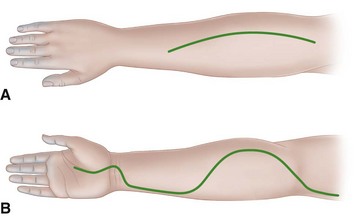
 For the volar fasciotomy, make a curvilinear incision similar to McConnell’s combined exposure of the median and ulnar nerve neurovascular bundles as described by Henry (Fig. 74-5). Make an anterior curvilinear incision medial to the biceps tendon, crossing the elbow flexion crease at an angle. Carry the incision distally into the palm to allow for a carpal tunnel release, but avoid crossing the wrist flexion crease at a right angle.
For the volar fasciotomy, make a curvilinear incision similar to McConnell’s combined exposure of the median and ulnar nerve neurovascular bundles as described by Henry (Fig. 74-5). Make an anterior curvilinear incision medial to the biceps tendon, crossing the elbow flexion crease at an angle. Carry the incision distally into the palm to allow for a carpal tunnel release, but avoid crossing the wrist flexion crease at a right angle.
 Divide the lacertus fibrosus proximally, and evacuate any hematoma.
Divide the lacertus fibrosus proximally, and evacuate any hematoma.
 In patients with suspected brachial artery injury, expose the brachial artery and determine whether there is a free blood flow. If the flow is unsatisfactory, remove the adventitia to expose any underlying clot, spasm, or intimal tear. Resect the adventitia if necessary, and anastomose or graft the artery.
In patients with suspected brachial artery injury, expose the brachial artery and determine whether there is a free blood flow. If the flow is unsatisfactory, remove the adventitia to expose any underlying clot, spasm, or intimal tear. Resect the adventitia if necessary, and anastomose or graft the artery.
 Release the superficial volar compartment throughout its length with open scissors, freeing the fascia over the superficial compartment muscles.
Release the superficial volar compartment throughout its length with open scissors, freeing the fascia over the superficial compartment muscles.
 Identify the flexor carpi ulnaris, and retract it with its underlying ulnar neurovascular bundle medially, and retract the flexor digitorum superficialis and median nerve laterally to expose the flexor digitorum profundus in its deep compartment. Check to see if its overlying fascia or epimysium is tight, and incise it longitudinally.
Identify the flexor carpi ulnaris, and retract it with its underlying ulnar neurovascular bundle medially, and retract the flexor digitorum superficialis and median nerve laterally to expose the flexor digitorum profundus in its deep compartment. Check to see if its overlying fascia or epimysium is tight, and incise it longitudinally.
 If the muscle is gray or dusky, the prognosis for recovery may be poor; however, the muscle may still be viable and should be allowed to perfuse.
If the muscle is gray or dusky, the prognosis for recovery may be poor; however, the muscle may still be viable and should be allowed to perfuse.
 Continue the dissection distally by incising the transverse carpal ligament along the ulnar border of the palmaris longus tendon and median nerve.
Continue the dissection distally by incising the transverse carpal ligament along the ulnar border of the palmaris longus tendon and median nerve.
 In cases of median nerve palsy or paresthesias, observe the median nerve along the entire zone of injury to ensure that it is not severed, contused, or entrapped between the ulnar and humeral head of the pronator teres. If it is, a partial pronator tenotomy is necessary.
In cases of median nerve palsy or paresthesias, observe the median nerve along the entire zone of injury to ensure that it is not severed, contused, or entrapped between the ulnar and humeral head of the pronator teres. If it is, a partial pronator tenotomy is necessary.
 In a patient with a supracondylar fracture, reduce the fracture, pin it with Kirschner wires, and control the bleeding.
In a patient with a supracondylar fracture, reduce the fracture, pin it with Kirschner wires, and control the bleeding.
 Do not close the skin at this time; anticipate secondary closure later.
Do not close the skin at this time; anticipate secondary closure later.
 If the median nerve is exposed within the distal forearm, suture the distal radial-based forearm flap loosely over the nerve.
If the median nerve is exposed within the distal forearm, suture the distal radial-based forearm flap loosely over the nerve.
 Check the dorsal compartments clinically, or repeat the pressure measurements. Usually, the volar fasciotomy decompresses the dorsal musculature sufficiently, but if involvement of the dorsal compartments is still suspected, release them also.
Check the dorsal compartments clinically, or repeat the pressure measurements. Usually, the volar fasciotomy decompresses the dorsal musculature sufficiently, but if involvement of the dorsal compartments is still suspected, release them also.
 Make the incision distal to the lateral epicondyle between the extensor digitorum communis and extensor carpi radialis brevis, extending approximately 10 cm distally. Gently undermine the subcutaneous tissue, and release the fascia overlying the mobile wad of Henry and the extensor retinaculum.
Make the incision distal to the lateral epicondyle between the extensor digitorum communis and extensor carpi radialis brevis, extending approximately 10 cm distally. Gently undermine the subcutaneous tissue, and release the fascia overlying the mobile wad of Henry and the extensor retinaculum.
 Apply a sterile moist dressing and a long-arm splint. The elbow should not be left flexed beyond 90 degrees.
Apply a sterile moist dressing and a long-arm splint. The elbow should not be left flexed beyond 90 degrees.