Evaluation of Bone Lesions Around the Hip
Eric A. Silverstein
Key Points
Introduction
The hip is a common location for many soft tissue and osseous neoplasms in addition to tumor simulators. It is the role of the orthopaedic surgeon to recognize and be aware of these lesions in order to treat them appropriately or refer these patients in a timely fashion. The spectrum of disease varies from latent benign to overly aggressive malignancies.
This chapter will focus on the epidemiology and clinical evaluation of neoplasms arising around the hip. An overview of common lesions and their treatment is presented. Chapter 50 will cover specific treatment techniques for benign tumors. Chapter 51 will cover malignant tumors, and Ch. 52 will discuss the treatment of metastatic disease in detail.
Epidemiology of Hip Lesions
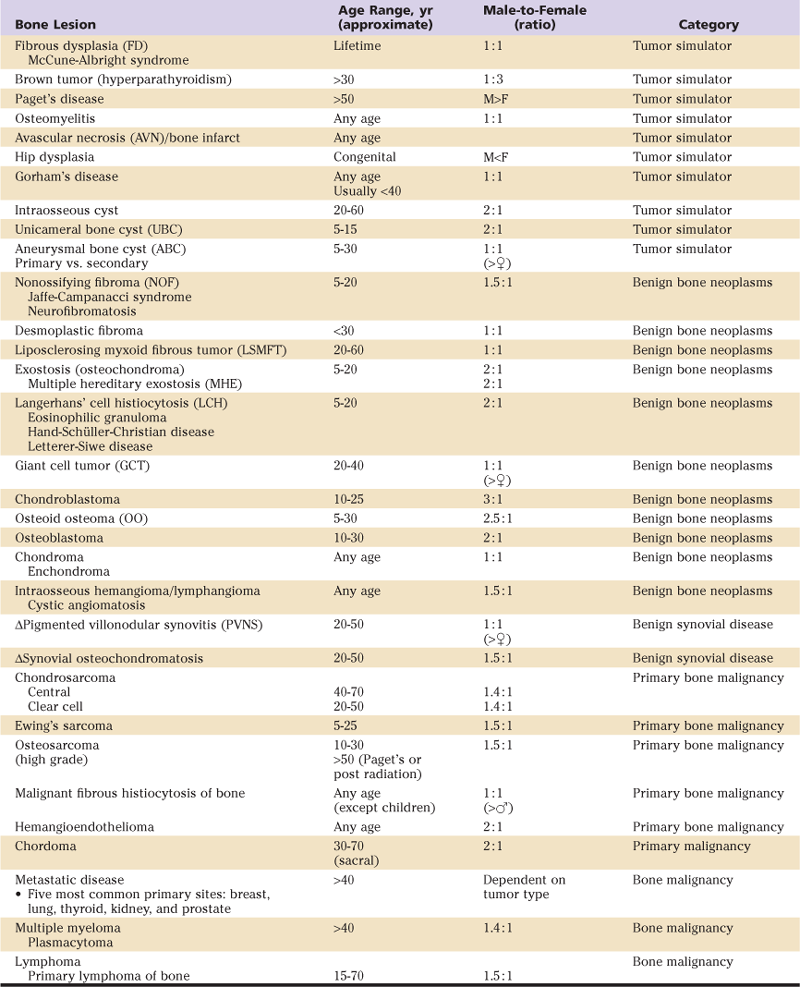
A variety of lesions are found in and around the hip. The age range is by no means absolute; however, certain neoplasms, such as Ewing’s sarcoma, are rarely seen beyond the age of 25. Most bone tumors have a predilection for males, with few exceptions. The plethora of hip lesions include broad categories such as tumor simulators (infection, congenital and endocrine disorders, genetic sequelae, and those unexplained), benign and malignant primary bone tumors, synovial-based disease, and metastatic/bone marrow–based disease. Table 49-1 summarizes most of the lesions found around the hip.1–8
Table 49-1
Most Common Lesions Around the Hip
Δ, Synovial-based disease (not bone).
Clinical Evaluation
The history is key in approaching a patient with a suspected tumor about the hip. The patient’s presenting age is an important factor in narrowing down the differential diagnosis. Although any tumor can theoretically occur at any age, there are characteristic age distributions to benign and malignant tumors which will focus the attention of the clinician toward the most likely candidates. These are outlined in Table 49-1.
Obviously, a personal or family history of malignancy or other tumorous condition of bone is relevant. A genetic basis is understood for a number of bone tumors and suspected for many for which it has not yet been ascertained. Particularly in any patient with a history of prior malignancy, the presence of a new tumor about the hip must prompt suspicion for metastatic disease.
The patient’s presenting pain is an essential aspect of the evaluation. Lesions may be diagnosed incidentally, they may present with mechanical pain, or they may present with true night pain. The last is pain which is present characteristically at night such that it will actually awaken patients from a sound sleep. Lesions which are found incidentally are most likely be to benign; those which present with mechanical pain are concerning for impending pathologic fracture. Patients presenting with true night pain will raise a high suspicion for malignancy.
The presence of a mass is also an important aspect of the history. The hip and pelvis region are deep-seated structures, so any mass which is detected is usually large and advanced by the time it is clinically apparent. Soft tissue tumors will often present with a painless mass even when malignant and quite large. Fortunately, only a tiny fraction of soft tissues masses presenting in adults represent malignancy.
Clinical Examination
The standard orthopedic examination of the hip joint is performed in any patient presenting with a suspected tumor. Examination is best expressed in degrees of motion as well as percentage comparison of the opposite side. Any maneuvers which elicit pain during the examination should be noted for further evaluation. In addition to a standard orthopedic joint evaluation, a full oncologic examination should be performed. This includes an assessment of any masses, evaluation for lymphadenopathy, and any skin changes which may be present in the region of the tumor. Masses should be investigated for the presence of any Tinel’s sign, mobility, tenderness, or pulsation. The presence of hepatosplenomegaly or other findings suggestive of disseminated disease are noted. As well, skin manifestations of any systemic disease or genetic syndrome (for example: café au lait spots suggestive of neurofibromatosis) are noted.
Patients with a large mass or destructive lesion will often have compromise of neurologic function in the limb. Careful testing of sciatic nerve function is critical. Although pulses may be palpable, in the setting of a large mass, the ankle brachial index may be decreased due to compression of vascular structures. Deep venous thromboses are common in patients presenting with large malignant masses around the hip and the limb should be assessed for swelling or other stigmata of this. It is helpful to measure the circumference of the limb compared to the uninvolved side at defined points.
Imaging
Characteristics that make an osseous lesion aggressive and concerning include an associated soft tissue mass, periosteal elevation, permeative appearance, large size and rapid growth. Plain film radiographs provide the first assessment of any patients presenting with a potential tumor about the hip. Radiographs have the advantage of defining any bony lesions as lytic, blastic, or having a mixed characteristic. They help determine whether a mass is producing osteoid or has matrix calcification. Additionally, even in patients with soft tissues masses alone, radiographs can define soft tissue calcifications which can be helpful in the evaluation process.
Radiographs provide a rapid assessment of present or impending fracture. However, it is important to remember that up to a third of bony mineral must be lost before a lesion is apparent of plain film radiographs. In imaging lesions around the pelvis, inlet and outlet as well as Judet views are helpful in evaluating periacetabular or iliac wing lesions. Proximal femoral lesions are adequately evaluated by AP and lateral radiographs of the hip. It is important to obtain full length radiographs of the entire femur to evaluate for the presence of any skip or distal lesions which are not immediately apparent on plain films of the hip itself.
Magnetic resonance imaging provides the most sensitive evaluation of tumoral processes in the bone marrow and the greatest definition of soft tissues masses. In obtaining an MRI scan, images before and after administration of gadolinium contrast are always obtained in evaluating potential tumors about the hip. At least one T1 weighted sequence which evaluates the entire extent of the bone is used to evaluate for skip lesions. Most commonly this is a coronal T1 weighted sequence of the femur. T1 weighted sequences provide a sensitive evaluation for marrow replacing processes to fully define the extent of lesions. Contrast enhancement patterns help separate cystic from solid masses in the evaluation of these patients.
Computed tomography is the most sensitive method to define bony architecture around the hip and provides a rapid assessment of whether any matrix or osteoid production is present. CT can be combined with angiography to define any vascular encasement by tumor. It also provides an efficient mechanism to evaluate for an impending pathologic fracture when plain film radiographs may not clearly demonstrate loss of bony trabeculae or cortical compromise.
In addition to its role for local imaging of the tumor, CT of the chest, abdomen, and pelvis is most commonly utilized as a part of staging studies for patients with suspected or proven malignancy. Although CT of the chest is the most defined study for staging of sarcomas, our practice is to obtain CT images of the abdomen and pelvis for any patient who has a malignancy extending at or above the level of the inguinal ligament to identify any regional metastases which may be missed by CT of the chest alone.
Technetium bone scan is used to define whether a process is active or latent and is a component of staging of most bony malignancies. Although false negatives can be seen (such as in the case of multiple myeloma or in very aggressive lytic tumors), the bone scan is a reliable method for evaluation of most bony processes around the hip and pelvis. In those cases in which it proves unreliable, a bone survey can be used.
The role of positron emission tomography (PET) scanning is evolving at this time. PET is established for the evaluation of patients with aggressive carcinomas among several other malignancies. As it relies upon metabolic uptake by tumor cells, it is less reliable in evaluation of patients with low or intermediate grade malignancies. The role of PET in evaluating patients with high-grade sarcomas is currently under study. PET scans are usually combined with a low resolution CT scan for co-registration of anatomic abnormalities.
Not all imaging modalities are necessary in all patients. Many benign lesions have characteristic appearances on plain radiographs and no further local imaging is necessary. The most common imaging protocol to define suspected lesions around the hip combines plain film radiographs with contrast enhanced MR scans. This combination of imaging is usually adequate to form a differential diagnosis, evaluate the likelihood of malignancy and plan an appropriate oncologic biopsy. Further imaging will be described in the other chapters in this section pursuant to individual lesions.
Biopsy
Biopsy confirmation of suspected histology is usually required. The hazards of a poorly planned biopsy have been well established by Dr. Mankin in a series of studies.9–10 Poorly planned biopsies will frequently alter the surgical options available for patients and may preclude limb salvage in the setting of a sarcoma diagnosis. Additionally, in adults presenting with suspected metastatic disease, proper staging studies will usually yield the diagnosis without the need for biopsy or may identify a safer alternative site to sample. Dr. Rougraff and colleagues established a protocol of history, routine laboratory studies, bone scan, and CT of the chest, abdomen, and pelvis as identifying the site of primary disease in 85 percent of patients.11
The key principle to musculoskeletal biopsy is that the biopsy tract must be able to be excised with the definitive resection if the tumor proves to be a primary sarcoma.12 For this reason there is the strong recommendation that a musculoskeletal biopsy either be performed or directed by a surgeon who is prepared carry out the definitive resection should a sarcoma be diagnosed. Most biopsies around the hip and pelvis can be performed either through a direct lateral approach for lesions of the proximal femur or along the line of the iliac crest for lesions of the pelvis. This follows the utilitarian incision to the hip as described by Enneking. Biopsies performed along this line are readily excised at the time of surgery.
A tension always exists between performing an open versus a closed biopsy. Open biopsies have the advantage of obtaining a large volume of tissue for histopathologic analysis, particularly if advanced cytogenic or other tests are necessary. They are generally considered the gold standard for diagnosis. However, open biopsies are not without risk; when compared to percutaneous biopsy they expose a larger area of tissue to contamination which will require ultimate excision should a malignancy be proven.
Percutaneous biopsy varies from fine needles (which yield very limited samples) to large bore core needles which can provide samples with preserved architecture. Large bore core needle biopsies directed by CT or magnetic resonance imaging can be used to access areas of the tumor which are likely to be most representative while minimizing areas of soft tissue contamination.13–14 Percutaneous core needle biopsies must be carefully planned with the performing radiologist as well as pathologist to ensure that representative tissue is obtained in a sufficient quantity to allow a diagnosis while maintaining a safe trajectory. We will often tattoo the site of the biopsy with a small drop of methylene blue to allow it to be identified later for subsequent surgical resection. The ultimate decision for closed versus open biopsy is tailored to the clinical situation and influenced by institutional practice patterns and resources. Cultures are sent in addition to histology studies.
Should an open biopsy be necessary, it is critical that the most direct route to the tumor be used for sampling. Classic anatomic planes are deliberately avoided during these procedures to minimize contamination of multiple compartments. Rather, the approach usually goes directly to the tumor through the edge of an anatomic compartment which can be excised en bloc with the tumor as a more classic plane of approach is used. Major neurovascular structures are deliberately avoided during the course of the biopsy so that they will not be subject to contamination when the tumor is sampled. Very careful hemostasis is practiced and a drain is almost always left during an open biopsy of the proximal femur or pelvis to minimize the resulting hematoma and potential zone of contamination. Nonabsorbable Ethibond sutures are used at closing the fascia following the biopsy so that they can be identified at the time of surgery to be certain that the area is excised fully.
Pathophysiology, Clinical Features, Radiographic Appearance, Differential Diagnosis, Treatment, and Prognosis
Tumor Simulators
This category encompasses tumor-like lesions (“pseudotumors”) that generally are reactive or hyperplastic tissue responses. This definition does carry some ambiguity, and semantics may play a role. Therefore, certain lesions, such as aneurysmal bone cyst (ABC) and unicameral bone cyst (UBC), may be considered a tumor simulator or a benign bone tumor.
Fibrous Dysplasia (FD)
Fibrous dysplasia is a benign intramedullary fibro-osseous dysplastic lesion first described by Lichtenstein in 1938 with an origin linked to an activating mutation in the gene that encodes the α-subunit of stimulatory G protein (Gsα). This results in developmental failure in the remodeling of primitive bone to mature lamellar bone. The consequence is biomechanically inferior bone that is non–stress oriented and prone to fracture. The process can be monostotic or polyostotic, and the most extreme presentation is McCune-Albright syndrome, which consists of polyostotic disease with café au lait spots and hyperfunction of multiple endocrine glands. Monostotic disease is fairly common and is not hereditary. Most lesions are asymptomatic and are found incidentally. However, a fairly common clinical presentation consists of bone pain, deformity, and fatigue/pathologic fracture. Wide variability in radiographic features is seen on plain radiographs. The classic pattern is a “ground-glass” appearance. However, cortical thinning, expansile remodeling, endosteal scalloping, and mixed radiolucency and radiodensity are common. Other findings may include coxa vara, shepherd’s crook deformity, and protrusio acetabuli (Fig. 49-1). A computed tomography (CT) scan is the best imaging modality, but magnetic resonance imaging (MRI) is often helpful, especially in cases of cystic formation. A radionuclide bone scan will frequently be hot. Histopathology normally demonstrates a bland-spindle cell stroma with embedded trabeculae of woven (immature) bone with no osteoblastic rimming. This is often referred to as an “alphabet soup” appearance.
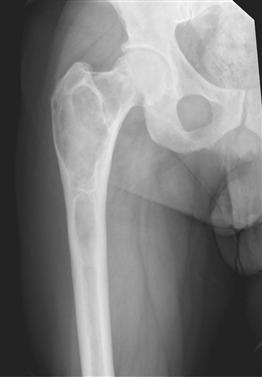
Figure 49-1 Anterior-posterior (AP) radiograph of the hip demonstrating a “ground glass” appearance with mild varus remodeling characteristic of fibrous dysplasia.
The diagnosis of fibrous dysplasia is often made on clinical and imaging presentation only. However, with atypical lesions, a biopsy is prudent. The differential diagnosis includes simple bone cysts, osteofibrous dysplasia, nonossifying fibroma, chondroma, low-grade intramedullary osteosarcoma, and rarely Paget’s disease. The clinical course is variable. Adults are often more symptomatic than children. There is no cure. Surgery is performed to prevent pathologic fracture, to correct limb alignment, and to decrease pain likely to result from fatigue fractures. The best options include the use of hardware or cortical allografts because the process (particularly if still active) can rapidly resorb most bone grafts. Bisphosphonates have shown efficacy in decreasing bone pain in patients with active symptomatic polyostotic disease. Cases of sarcoma developing within areas of fibrous dysplasia have been reported rarely.1–3,5–8,15–17
“Brown Tumor”
These pseudotumors are rare sequelae of primary hyperparathyroidism that has a delay in diagnosis or remains untreated. Primary hyperparathyroidism is caused by an adenoma of a single parathyroid gland in approximately 80% of cases, less likely both glands, hyperplasia, or cancer. Uncontrolled increased secretion of parathyroid hormone (PTH) causes an increase in gastrointestinal tract calcium absorption, renal tubular reabsorption of calcium in the kidney, and osteoclastic destruction of bone. This normally results in hypercalcemia and can present with symptoms such as lethargy, confusion, impaired mentation, depression, muscular weakness, loss of appetite, anorexia, nausea and vomiting, constipation, polydypsia/polyuria, nephrolithiasis, peptic ulcer, pancreatitis, and heart problems. This process can also cause profound bone changes. A “brown tumor” is one such consequence that is evident on x-ray as lytic areas, sometimes with expansile remodeling of the cortex and pathologic fractures. These may simulate metastatic bone disease. The diagnosis usually can be established by blood tests showing hypercalcemia, hypophosphatemia, and an elevated PTH, assuming normal renal function. A biopsy of these lesions shows variable results, including hyperplastic tissue that is fibrous, fibro-osteoid, and giant cell rich; however, lesions can mimic a giant cell tumor, a reparative giant cell granuloma, or an aneurysmal bone cyst. Treatment consists of surgical excision of the parathyroid adenoma. Bone tumors will normally resolve; however, in cases of pathologic fracture or impending fracture, they generally are treated with internal fixation.2-6,18
Unicameral Bone Cyst (UBC) (Simple Bone Cyst)
These lesions are common in skeletally immature patients, particularly males. They represent a centrally located cystic lesion of bone that originates in the metaphysis and normally migrates toward the diaphysis with skeletal growth. They are filled with fluid similar to serum. Most are asymptomatic until presentation with a pathologic fracture. X-rays typically demonstrate a central radiolucency in the metadiaphyseal region with significant attenuation and possible mild expansile remodeling. Bone scans normally show little to no uptake unless a fracture is present. CT and MRI show fluid content within the lesion, but a fluid-fluid level characteristic of an aneurysmal bone cyst will not be seen. Bone septation is often seen in varying amounts from ridges along the inner surface of the cortex. The characteristic “fallen leaf” sign may be seen in the presence of a pathologic fracture, which represents a fragment of cortex that sinks into the fluid of the cyst. Histopathology shows a thin membrane walling the cyst that is made of loose fibrous tissue with cells that may resemble endothelial or synovial cells. Hemosiderin, giant cells, and woven bone intermixed are not uncommon. Biopsies usually are not indicated unless atypical. The differential diagnosis includes aneurysmal bone cyst and fibrous dysplasia. Treatment is provided for those at risk of fracture, particularly those with active cysts (adjacent to physis, single cavity with a very thin cortex), one or more recent fractures, and weight-bearing bones. Observation is reasonable for inactive cysts and those at low risk of fracture. Surgical treatment options are still debatable. Many use an aspiration and injection technique with corticosteroids or a combination of autogenous bone marrow and demineralized bone matrix. However, open curettage and bone grafting with or without internal fixation still has utility, especially in lesions around the hip, where structural stability is critical.2–4,17,19–23
Aneurysmal Bone Cyst (ABC)
These lesions are considered hyperplastic pseudotumors, which normally are seen in patients younger than 30 years of age. They are thought to be the result of a reactive reparative process that is hemorrhagic. They can be primary or secondary (to a large number of tumors). ABC generally presents as an enlarging, occasionally painful mass. Growth can vary greatly and at times it can be explosive. Malignant transformation has not been documented, and rarely spontaneous involution is seen. Radiographically, lesions are radiolucent with expansile remodeling or a “blow-out” appearance within the metaphysis. They can propagate to the epiphysis and diaphysis and often are eccentric with an “eggshell-thin” outer cortex. Occasionally, they are subperiosteal and can appear septated. Bone scans show predominantly peripheral uptake. CT and MRI often show the characteristic fluid-fluid level representing the separation of blood components. MRI with or without intravenous gadolinium demonstrates rim enhancement consistent with cystic properties. Grossly, an ABC often has a paper-thin reactive shell of bone that has a bluish tinge from old blood in the cavity. Histopathology is consistent with bland stromal cells lining the cavity of bone with multinucleated giant cells. Giant cells often cluster around the edges of the vascular lakes (“blood lakes”). Hemosiderin is usually present. Principal lesions in the differential diagnosis include simple bone cyst, giant cell tumor, and telangiectatic osteosarcoma. Treatment consists of extensive curettage with local adjuvant and reconstruction in most cases. However, with severe bone loss, en bloc resection may be needed. Recurrence rates vary from 20% to 40% without adjuvant, but can exceed 50% in the most aggressive form. External beam radiation can be used for difficult or dangerous surgical regions (e.g., sacrum or spine) and is fairly effective at low doses. Embolization is rarely used in cases not amenable to surgery or radiation and has unpredictable results.*
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








