Chapter 85 Diabetic Foot
Risk Factors for Foot Problems in Diabetic Patients
Peripheral Neuropathy
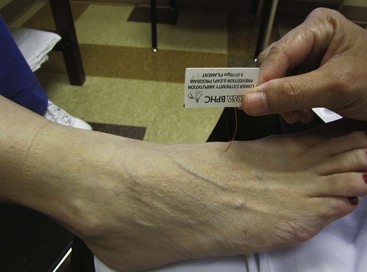
Peripheral neuropathy can be diagnosed by physical examination and can be confirmed with electromyography/nerve conduction studies. Sixty to 70 percent of patients with diabetes have some neural manifestation, and almost 30% of patients with diabetes who are 40 years or older have loss of sensation in the feet. Loss of protective sensation can be determined by use of a 5.07-mm Semmes-Weinstein filament and is thought to be the threshold at which complications such as neuropathic ulcers and Charcot arthropathy occur (Fig. 85-1).
Peripheral Vascular Disease
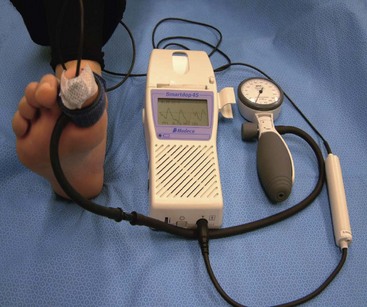
The ankle-brachial index can be unreliable for the diagnosis of peripheral arterial disease in diabetic patients, because calcification of arteries can lead to elevated results, masking the severity of the disease. Toe pressure or TcpO2 is thought to be more reliable in diabetic patients (Fig. 85-2). Angiography is the gold standard for diagnosing arterial lesions that may require intervention; however, it may be complicated by the fact that many diabetics have renal disease, which can be worsened by the intravenous contrast medium used in such studies.
Diabetic Ulcers and Infections
Pathophysiology
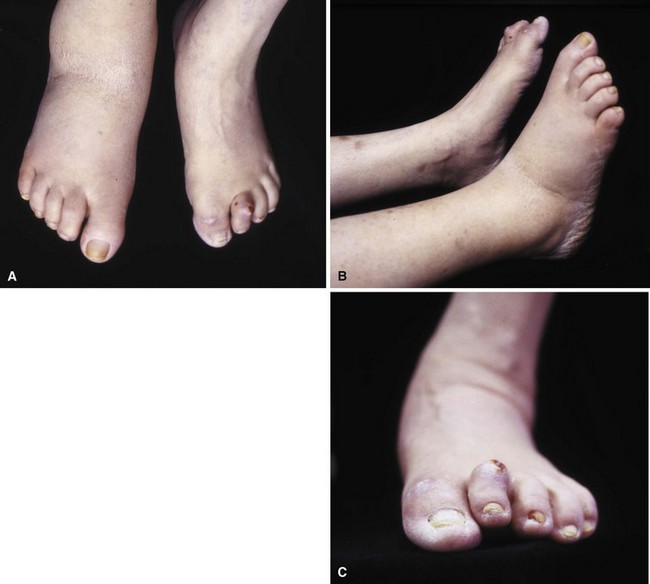
Patients who develop ulcers usually have a combination of peripheral neuropathy, deformity or joint contracture, increased plantar pressure, and peripheral arterial disease. Because of the loss of sensation, patients do not realize that undue pressure is placing their skin at risk. Motor neuropathy can lead to deformity such as claw toes; the bony prominences from the deformity make the skin more vulnerable to breakdown (Fig. 85-3). Achilles contracture can lead to increased forefoot pressures, which increases the chances of forefoot ulceration. There are known structural changes in the Achilles tendon in diabetic patients, including disorganization of the tendon fibers and calcification within the tendon. These changes are more prevalent in older patients and can explain the increased stiffness that is known to occur in the Achilles tendons in diabetics. Peripheral arterial disease also places the skin at risk and leads to impairment of healing once ulceration does occur. The presence of peripheral arterial disease increases the risk of ulceration ninefold.
Classification
The Wagner classification (Box 85-1) is the most commonly used for grading foot ulcers. The University of Texas San Antonio classification system adds the criteria of infection and ischemia and correlates more closely with prognosis because increased severity (higher grade) is associated with longer healing times and amputation.
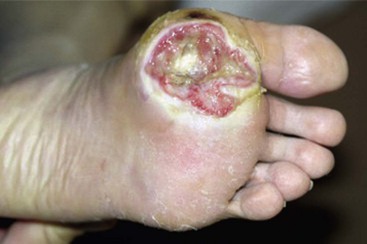
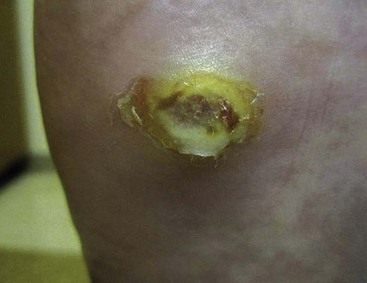
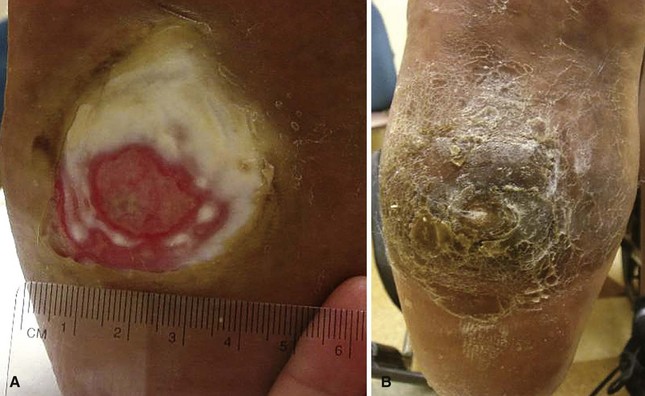
FIGURE 85-5 Deep (Wagner grade 2) ulcer with exposed tendon and joint capsule.
(From Brodsky JW: The diabetic foot. In Coughlin MJ, Mann RA, Saltzman CL, editors: Surgery of the foot and ankle, ed 8, Philadelphia, 2007, Elsevier.)
Treatment
Nonoperative Treatment
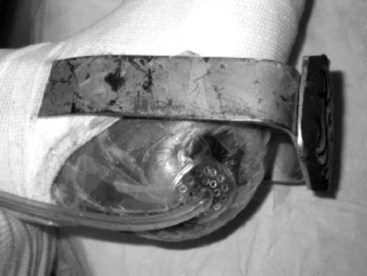
Total contact casting is the standard of care, because it reduces plantar loads better than a well-molded shoe cast and, by extrapolation, better than shoes with custom insoles (Fig. 85-7). Complications may arise from the use of total contact casts, but most are minor and reversible new areas of ulcerations. The risk for complications of total contact casting is lower after deformity-correcting surgery, as well as when the patient is non–weight bearing. Trepman et al. described a method for total contact casting (Fig. 85-8). In a patient who cannot be kept non–weight bearing, the addition of a metal stirrup that extends beyond the foot-plate of the cast takes pressure off the plantar surface of the foot and transmits it to the shank of the cast (Fig. 85-9). Healing rates after total contact casting can be high; however, the recurrence rate also can be high unless severe deformities are surgically corrected.
Removable diabetic boots (Fig. 85-10) have been shown to be as efficacious as total contact casting in some studies; however, in one study, whereas the boot demonstrated better forefoot unloading than a total contact cast, healing rates were better in the cast, presumably secondary to lack of patient compliance with the boot. Wrapping the diabetic boot to make it less removable does lead to healing rates that are higher than boots that are not wrapped, again suggesting that patient compliance is an issue with the removable boot.
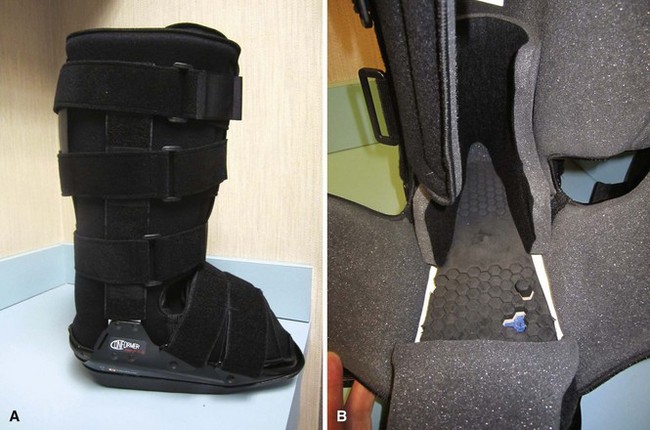
FIGURE 85-10 Removable diabetic boot can be used as alternative to total contact cast. A, Exterior. B, Interior.
Operative Treatment
According to guidelines based on a systematic review by the International Working Group on the Diabetic Foot, the indications for urgent surgical intervention include necrotizing infections and gangrene or deep abscesses (Fig. 85-11). Less urgent surgery may be required if there is a substantially compromised soft tissue envelope, loss of mechanical function of the foot, or bone involvement that is limb threatening or if the patient prefers to avoid prolonged antibiotic therapy. Surgical débridement of osteomyelitis is not always required.
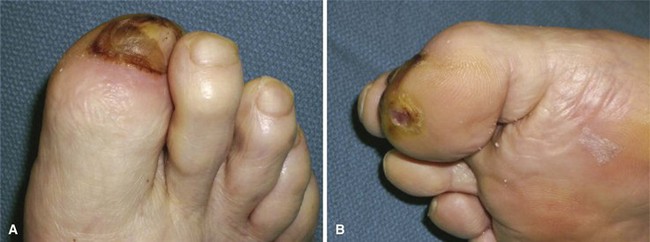
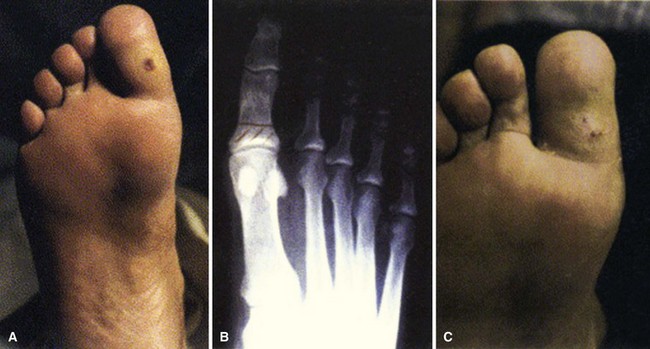
FIGURE 85-11 Cellulitis, abscess, and osteomyelitis arising from ulcer over fifth toe caused by tight shoe.
(From Brodsky JW: The diabetic foot. In Coughlin MJ, Mann RA, Saltzman CL, editors: Surgery of the foot and ankle, ed 8, Philadelphia, 2007, Elsevier.)
For severe infections with abscess formation, incision and drainage with thorough débridement may be required. In cases of chronic osteomyelitis that has failed to respond to intravenous antibiotics, surgical débridement may be necessary. Infected bone should be completely excised; however, every effort should be made to preserve as much bone as possible. For osteomyelitis in the toes, amputation may be required. Preservation of part of the proximal phalanx may help keep the adjacent toes from drifting (Fig. 85-12).
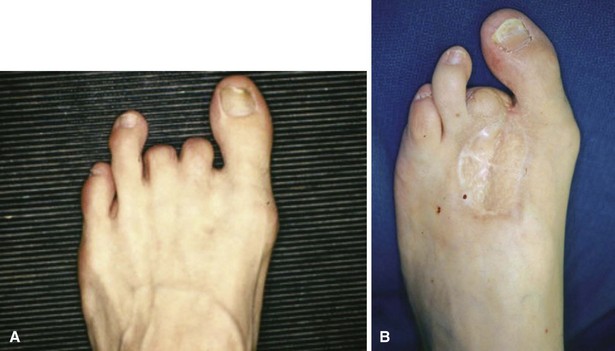
FIGURE 85-12 A and B, Drift of toes to fill defect created by amputation of toe.
(From Brodsky JW: The diabetic foot. In Coughlin MJ, Mann RA, Saltzman CL, editors: Surgery of the foot and ankle, ed 8, Philadelphia, 2007, Elsevier.)
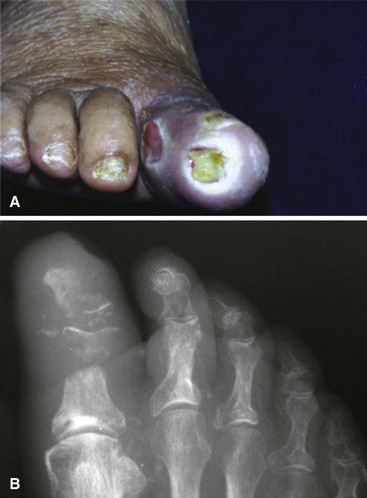
Osteomyelitis of the metatarsal heads is relatively common (Fig. 85-13) because many ulcers occur in this area.Metatarsal head resection may be indicated. If multiple metatarsal heads are involved, resection of all lesser metatarsal heads or transmetatarsal amputation may be required. Ray resection may be needed if the osteomyelitis involves more than the metatarsal head. Midfoot osteomyelitis can be treated with exostectomy if stability can be preserved. Hindfoot infections occasionally can be treated with amputation at that level, but a below-knee amputation often is more functional. Partial calcanectomy may be attempted if osteomyelitis affects the calcaneus secondary to a heel ulcer and can avoid a below-knee amputation.
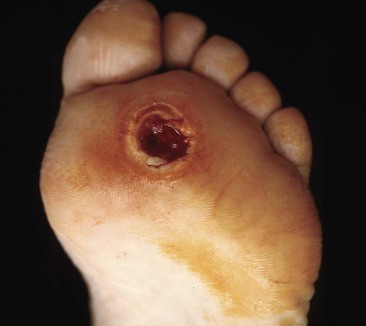
FIGURE 85-13 Grade 3 ulcer under second metatarsal head extends into metatarsal head with presumptive contiguous osteomyelitis.
Modified resection arthroplasty after débridement of infected tissue can avoid toe amputation in patients with chronically infected ulcers and claw toe deformities (Fig. 85-14), and toe flexor tenotomies can lead to healing of ulcers at the tip of the toe if the claw toe deformity is flexible and wounds are Wagner grade 1, 2, or 3. For chronic infected ulcers under the first metatarsal head, ray resection may be avoided with resection of the first metatarsophalangeal joint and pin stabilization.
Charcot Arthropathy
Pathophysiology
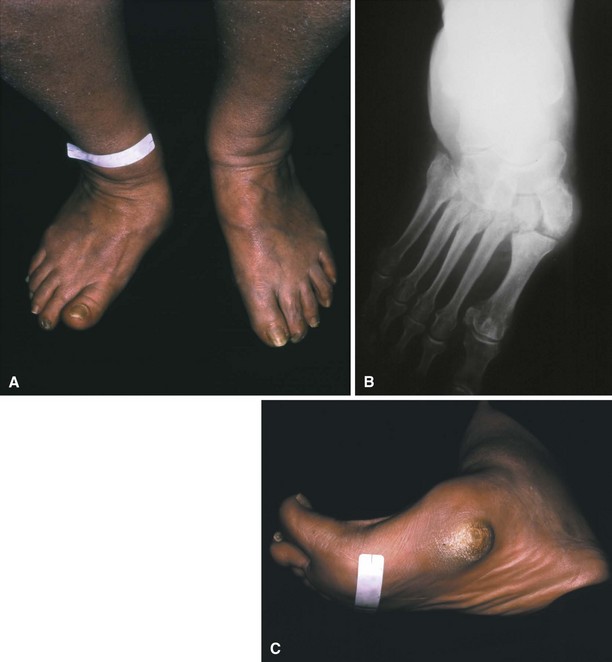
The most plausible explanation for the development of neuropathic arthropathy in a diabetic foot is loss of autonomic control of the vasculature. Resting blood flow in patients with diabetic neuropathy may be five times normal values. Arteriovenous shunting also has been documented in these feet. This high flow rate results in osteopenia, and, combined with somatosensory loss of pain and proprioception, multiple small mechanical insults unrecognized by the patient and occurring in osteopenic bone set the stage for bony dissolution and loss of structural integrity, followed by a collapse deformity (Fig. 85-15). Even with minor trauma to the foot (sprains, contusions, minor fractures), neuropathic skeletal changes may develop in a patient with diabetes mellitus.
Evaluation
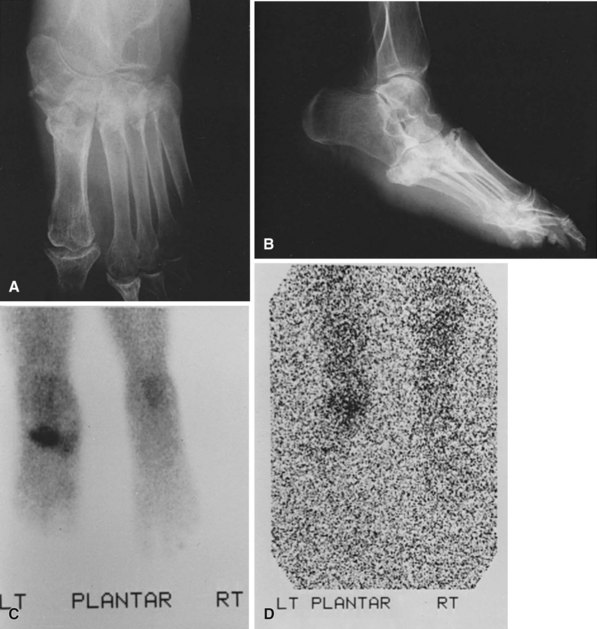
The diagnostic dilemma occurs when Charcot arthropathy is accompanied by ulceration and possible infection. Imaging studies such as plain radiographs, CT scans, and bone scans can be positive in both Charcot arthropathy and osteomyelitis (Fig. 85-16). White blood cell scans can be helpful, especially when combined with sulfur colloid bone marrow imaging. If infection is present, the tagged white cells will accumulate at the site but the sulfur colloid scan will be negative because bone marrow activity will be depressed by the infection. It does, however, take about 1 week for the sulfur colloid scan to be negative after the onset of infection. Positron emission tomography can be very sensitive and specific, but this test is not widely available. It also can be difficult to differentiate between Charcot arthropathy and infection on MRI; MRI characteristics of sinus tract, replacement of soft tissue fat, fluid collection, and extensive bone marrow abnormality may indicate infection in a patient with Charcot arthropathy. Thin rim enhancement of effusion, subchondral cysts, and intraarticular bodies suggest a lack of infection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree