CHAPTER 65 Degenerative Spondylolisthesis
Degenerative spondylolisthesis (DS) was first described in 1930 by Junghanns, who coined the term pseudospondylolisthesis to describe the presence of forward slippage of a vertebral body in the presence of an intact neural arch.1 The clinical and pathologic features of this entity were further defined by MacNab, who described the condition as “spondylolisthesis with an intact neural arch.”2 The term degenerative spondylolisthesis was originally used by Newman and Stone3 and is the terminology most commonly used to describe the anterior slippage of one vertebral body on another in the presence of an intact neural arch. More recently, an etiology-based classification has been proposed that distinguishes between the pathologic processes that may cause spondylolisthesis.4 Under this classification, two broad types of spondylolisthesis have been defined: developmental and acquired. DS, as described initially by MacNab, and subsequently by Newman and Stone, is a subtype of the acquired form subsequently described by Marchetti and Bartolozzi.4,5 In that classification, DS may be either primary or secondary. Primary degenerative spondylolisthesis is typically seen in middle-aged women and usually presents with clinical spinal stenosis. Secondary degenerative spondylolisthesis occurs as a result of a predisposing factor such as adjacent segment degeneration and slip above a preexisting fusion.
Epidemiology and Biomechanics
DS is a condition of older people and rarely affects those younger than 40 years of age. It most commonly involves the L4-5 level, although other levels may be affected. It much less commonly involves the L5-S1 level, in contrast to isthmic spondylolisthesis, which most commonly occurs at L5-S1. Factors that have been reported to predispose to anterolisthesis at the lumbosacral junction include an L5 vertebral body that is less deeply seated within the pelvis, a more slender L5 transverse process, and increased sacral inclination, all of which are more common in women than men.6
DS is approximately four to five times more common in females than in males and is more common in black females than in white females.7 The female preponderance is thought to be due to greater ligamentous laxity and hormonal effects.8–10 A significantly increased expression of estrogen receptors has been found in facet joints of postmenopausal women having severe facet arthritis associated with DS compared with those with spinal stenosis only. It was not clear whether the higher expression of estrogen receptors aggravated the degenerative facet changes or was a causative factor for DS.9
In an epidemiologic survey of 4151 patients, the Copenhagen Osteoarthritis Study found the incidence of DS was 2.7% in males and 8.4% in females.11 The only factors associated with an increased risk of DS in women were elevated body mass index (BMI), increased age, and increased angle of lordosis. In men, only increased age was associated with a higher risk of degenerative spondylolisthesis. There was no association between DS and age at menopause, smoking, or occupational lifting exposure.
It is likely that the development of DS is multifactorial and dependent on anatomic factors such as intervertebral disc pathology, ligament laxity, posterior facet joint arthrosis, the amount of lumbar lordosis, hormonal factors, and prior pregnancy. Genetic factors are also a likely contributor to the development of spinal stenosis with DS in some patients. One study demonstrated that a type IX collagen gene polymorphism that introduced a tryptophan residue into the protein’s triple helix predisposed its carriers to development of spinal stenosis with DS.12
A cadaveric model investigating the relative contributions of disc integrity and anterior and posterior longitudinal ligament factors in the development of low-grade anterolisthesis found that integrity of the disc was more important than ligamentous factors, although disruption of both was necessary to produce significant destabilization.13 The effect of facet joint orientation has also been reported to be a potential factor in the development of DS. Specifically, more sagittally oriented L4-5 facet joints have been implicated as a cause of DS.14–16 In one study, individuals in whom both L4-5 facet joint angles were sagittally oriented more than 45 degrees were 25 times more likely to have DS than those with less than 45 degrees of facet angulation.14 Whether the sagittal orientation is developmental or acquired is unclear and is a matter of debate. One study reported that sagittal orientation is a result of facet joint remodeling associated with arthrosis rather than the cause of anterior subluxation.16
A retrospective, age- and sex-matched, case-control radiographic study of middle-aged women found that decreased anterior disc height and increased lumbar index (lumbar index = posterior vertebral body height/anterior vertebral body height) were two independent predictors of DS.17
Natural History
The natural history of DS, like many other spinal conditions, is not well characterized.18 A meta-analysis of the literature on DS between 1970 and 1993 found that only 25 of the 152 studies reviewed, representing 889 patients, satisfied their inclusion criteria.19 Only three of these studies, encompassing 278 patients, described the natural history of DS.20–22 Overall, 90 of these 278 patients (32%) achieved satisfactory results without treatment. The study by Matsunaga and colleagues21 represented the best of the three studies and was the only true natural history study. In that study 40 patients who received no treatment were followed from 5 to 14 years (mean, 8.25 years). Only 4 of 40 patients (10%) showed clinical deterioration over the course of the study, and all were in the group of 28 patients who exhibited no slip progression over the follow-up period. Progressive slip was noted in 12 patients (30%), although none of the 12 patients exhibited clinical deterioration. The majority of the patients in this study showed slight improvement in their clinical symptoms over time. In general, no correlation was noted between slip progression and clinical deterioration. No slip progression was noted in patients with intervertebral disc narrowing, spur formation, subcartilaginous sclerosis, or ligamentous ossification, suggesting that these anatomic factors were protective against further slip and represented a mechanism of spinal restabilization. The lack of correlation between slip progression and progression of symptoms has also been reported by other authors.18,23,24 The generally favorable prognosis of DS was confirmed by a North American Spine Society (NASS) work group consensus statement that summarized evidence-based clinical guidelines on the diagnosis and treatment of DS.18 Although it did not distinguish between natural history and conservative care, that committee reported that most patients without neurologic deficits did well without surgery.
Clinical Features
The clinical features of DS are the same as those of spinal stenosis. DS, like spinal stenosis, may be either asymptomatic or may produce low back and/or leg pain. Back pain with DS is typically mechanical and may be aggravated by back extension or by arising from a bent posture. It is to be distinguished from discogenic back pain, which is typically provoked by flexion or sitting. Only a small percentage of patients with DS may experience low back pain (LBP). A recent report that investigated a small cohort of patients from the Framingham Heart Study concluded that there did not appear to be an association between LBP and DS, as diagnosed by computed tomography (CT).25
Leg pain may be either radicular or referred in a characteristic pattern of neurogenic claudication. Neurogenic claudication, also known as pseudoclaudication, is a clinical condition consisting of leg pain associated with walking.26 Neurogenic claudication must be distinguished from vascular claudication, which has slightly different clinical features, a different etiology, and completely different treatment (Table 65–1). Neurogenic claudication is defined as lower extremity pain, paresthesias, or weakness associated with walking or standing.27,28 Pain is the predominant symptom, being present in up to 94% of patients with spinal stenosis, with numbness (63%) and weakness (43%) being less common.27,28 Bilateral involvement is common. Patients with neurogenic claudication may present with either unilateral radicular pain or with diffuse, nondermatomal symptoms beginning in the buttocks and extending a variable distance into the legs. Radicular pain is typically dermatomal in distribution and is often unilateral. It is the presenting type of symptom in 6% to 13% of symptomatic patients with stenosis.28 It is often seen with lateral recess stenosis, foraminal stenosis, or concomitant disc herniation. The clinical effects of spinal canal narrowing are magnified by the presence of a degenerative slip that further narrows the spinal canal.
TABLE 65–1 Characteristics of Vascular vs. Neurogenic Claudication
| Evaluation | Vascular | Neurogenic |
|---|---|---|
| Walking distance | Fixed | Variable |
| Palliative factors | Standing | Sitting/bending |
| Provocative factors | Walking | Walking/standing |
| Walking uphill | Painful | Painless |
| Bicycle test | Positive (painful) | Negative (painless) |
| Pulses | Absent | Present |
| Skin | Loss of hair/shiny | Normal |
| Weakness | Rarely | Occasionally |
| Back pain | Occasionally | Commonly |
| Back motion | Normal | Limited |
| Pain character | Cramping/distal-to-proximal | Numbness/aching/proximal-to-distal |
| Atrophy | Uncommon | Occasionally |
Typical neurogenic claudication is less dermatomal in character than is radicular pain. It is frequently bilateral and may have a radicular component to it. Symptoms are typically produced by standing or walking and are relieved by sitting or bending forward (see Table 65–1). Indeed, patients may preferentially assume a stooped-over posture when walking or standing to ameliorate symptoms (“grocery cart sign”). Other leg symptoms such as weakness or numbness may also occur in association with standing or walking. Night pain is an uncommon feature of spinal stenosis, although it has been described in patients with lateral stenosis (lateral recess stenosis or foraminal stenosis). Unusual symptoms of spinal stenosis such as priapism associated with intermittent claudication during walking have also been reported.
The relationship of symptoms to posture can be explained on the basis of variation in canal size with posture.29,30 Cadaveric studies have demonstrated that spinal canal cross-sectional area, midsagittal diameter, subarticular sagittal diameter, and foraminal size are significantly reduced in extension and are increased with flexion.29 Similarly, neural compression is greater in extension than in flexion. An association between posture and epidural pressure measurements has also been demonstrated. In vivo studies relating posture to epidural pressure measurements have shown that epidural pressures at the level of stenosis were higher in standing compared with lying and sitting and were increased with extension and decreased with flexion.29
Neurogenic claudication should be distinguished from vascular claudication because their causes and treatments are different (see Table 65–1). Although both conditions may present as leg pain associated with walking, only patients with neurogenic claudication typically have leg pain with standing. Leg pain associated with neurogenic claudication is highly position dependent, whereas vascular claudication is unaffected by back flexion or extension. Leg pain with cycling in a sitting position is common with vascular claudication but is unaffected by neurogenic claudication.31 Patients with vascular claudication will typically have more leg pain produced by walking uphill than downhill, whereas patients with a neurogenic claudication will typically have less pain walking uphill, owing to the slightly flexed posture of the lumbar spine that results in neuroforaminal widening and reduced neural compression. Patients with neurogenic claudication may actually have increased leg pain when walking down an incline owing to associated lumbar lordosis and consequent neuroforaminal narrowing.
Radiographic Diagnosis
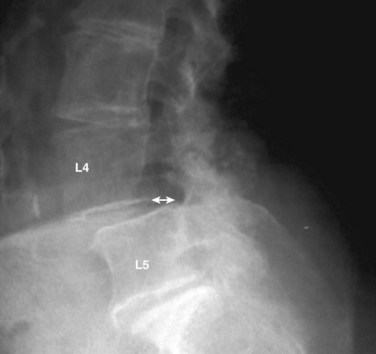
The diagnosis of DS is a radiographic diagnosis that is made on the lateral lumbar radiograph (Fig. 65–1). Although the slippage (anterolisthesis) may also be evident on a supine lateral radiograph, it is important that the lateral x-ray be performed in the standing position because there can be a dynamic component to the slip, causing it to reduce in the supine position and appear normal.32 It is not unusual for a patient to present with a normally aligned supine magnetic resonance imaging (MRI) study as the only radiographic study. Unless standing lumbar radiographs are obtained, however, the presence of a degenerative slip could be missed. In one recent study, 22% of L4-5 degenerative slips, as documented by standing lateral flexion-extension radiographs, were not detectable on supine MRI.33 Other dynamic radiographic views such as sitting or standing flexion-extension views and distraction-compression radiography may also be considered.
MRI may show increased signal within a facet joint at the level of the slip.33,34 Large facet effusions greater than 1.5 mm were found to be highly predictive of L4-5 DS, even in the absence of a measurable slip on the supine MRI.33 Therefore a patient who presents with only an MRI should be suspected of having a DS if a large facet effusion is detected, and standing lumbar radiographs should be obtained. Alternatively, the slip can be documented by upright or axial loaded MRI.34,35
Although the actual measurement of translation is generally straightforward, the distinction between what is normal dynamic translation and abnormal segmental motion (instability) is not.36,37 There is no consensus as to what constitutes clinically significant radiographic instability of the lumbar spine, nor even what is considered to be the normal range of translation between motion segments.36,37 As with routine radiographs, there exists a spectrum of normal translation that can exist in the absence of symptoms.36,37 One study showed that more than 90% of asymptomatic volunteers exhibited between 1 and 3 mm of translation on flexion-extension lateral lumbar radiographs and that a dynamic change of greater than 4 mm was therefore considered abnormal.36
Treatment
As mentioned previously, both the natural history of DS and, until recently, its optimal treatment are incompletely understood. The well-publicized Spine Patient Outcomes Research Trial (SPORT) was a prospective evaluation of the 2-year38 and 4-year18,39 outcomes of 607 patients with DS. Half of the patients were enrolled in a randomized cohort and half in an observational cohort. Pre-enrollment nonoperative care was not specified, and the type of surgery or nonoperative treatment during the study period was left to the discretion of the treating physicians. This study was hampered by a significant cross-over and nonadherence to treatment between the two groups, leading to both an as-treated and an intent-to-treat analysis of the data. When both the randomized and observational cohorts were combined, the as-treated analysis revealed that the surgically treated patients had significantly better outcome for both pain and function at 2-year and 4-year follow-ups. This study did not allow comparison of types of treatments, so it did not answer the question of which surgical treatments provided better outcomes.
That same SPORT trial examined radiographic predictors of outcome in both surgically and nonoperatively treated patients.40 Radiographic features examined included degree of slip (grade I vs. grade II), disc height (<5 mm vs. >5 mm), and mobility (stable vs. hypermobile). As noted previously, surgically treated patients had better outcomes than nonsurgically treated patients across all three radiographic parameters examined. For nonoperative patients, those with a grade I slip did better than those with a grade II slip, and those with a hypermobile slip did better than those with a stable slip.
In a long-term follow-up of patients with DS, progressive slip was noted in 34% of the 145 nonsurgically managed patients who were observed for a minimum of 10 years.41 This study was not a true natural history study because it included patients who had various nonsurgical interventions. Seventy-five percent of the patients were neurologically normal at the beginning of the study, and the majority (76%) remained so at final follow-up. Of the 34% who had neurologic symptoms, 83% experienced neurologic deterioration and had a poor outcome. There was no correlation between slip progression and clinical symptoms. This study suggested that conservative (nonsurgical) treatment in neurologically normal patients can result in satisfactory clinical outcome at an average of 10 years’ follow-up in the majority of patients.
Decompression Without Fusion
A recent review of Medicare patients undergoing surgery for stenosis, either with or without spondylolisthesis, from 2002 to 2007 found that only 21% of patients undergoing surgery for stenosis with spondylolisthesis in 2007 had simple decompression surgery, compared with 79% having some form of decompression with fusion.42 Although the trend in type of surgical procedure performed over this 6-year period was not examined for patients with stenosis associated with DS, the study noted that for patients with stenosis the trend was one of increasing complexity of surgical procedure. The rate of complex fusion, defined as fusion involving more than two levels or a 360-degree fusion, increased 15-fold from 2002 to 2007. It is likely that a similar increase in the rate of complex surgery occurred for patients with stenosis associated with DS.
One reason for considering decompression without fusion in select patient populations is that it is less invasive than fusion and reduces the morbidity and mortality associated with spinal fusion in elderly patients.42–46 In the recent retrospective review of Medicare claims for patients undergoing surgery for spinal stenosis between 2002 and 2007, patients having a complex fusion had greater morbidity, more life-threatening complications, greater likelihood of rehospitalization within 30 days of surgery, and higher costs compared with patients having decompression alone or decompression with simple fusion (defined as one- or two-level fusion through a single surgical approach).42
A meta-analysis of the literature on DS between 1970 and 1993 found only 11 papers, encompassing 216 patients, reporting outcome measures after decompression without fusion that met their inclusion criteria (Table 65–2).19 One of these studies was retrospective and nonrandomized,20 two were prospective and randomized,47,48 and the remaining eight were retrospective, nonrandomized, and uncontrolled. Overall, 69% of patients in this meta-analysis reported satisfactory outcome with decompression alone, with 31% having an unsatisfactory result.
TABLE 65–2 Results of Decompression without Fusion for Degenerative Spondylolisthesis: Meta-Analysis of Literature 1970-1993 (11 Articles)

One report that supported decompression without fusion for DS reviewed an elderly (average age, 67 years) population of 290 patients, 250 of whom had a one-level slip and 40 of whom had a two-level slip.49 The data from that study were self-reported by the surgeons and were retrospective. The decompressive procedures included laminectomy in 249 patients and fenestration procedures in 41 patients. Fenestration procedures typically involved bilateral laminotomy with partial medial facetectomy and foraminotomy. Only patients with a “stable” slip having less than 4-mm translation and less than 10 to 12 degrees of angulation on dynamic lateral radiographs were included. At an average follow-up of 10 years (range, 1 to 27 years), 69% of patients reported excellent outcome, 13% good outcome, 12% fair, and 6% poor. The authors concluded that 82% excellent/good outcome was acceptable in this elderly population, in whom fusion is associated with higher morbidity and mortality.
Similar results were reported in a recent retrospective review of 49 elderly patients (mean age, 68.7 years) with symptomatic degenerative lumbar spondylolisthesis, without evidence of hypermobility on flexion-extension radiographs, and who underwent decompression without fusion.50 At a mean follow-up of 3.73 years, 73.5% of the patients reported excellent or good results, although 10% underwent revision surgery with an instrumented fusion. The study concluded that limited decompression alone can be helpful in a select group of elderly patients without hypermobility.
Bilateral decompression through a unilateral approach has also been described in patients with spinal stenosis, both with and without DS. A retrospective study using that technique compared patients with and without spondylolisthesis and showed similar functional outcome between the two groups at 2 years.51 Although there was a statistically significant increase in the percentage slip postoperatively in the patients with spondylolisthesis, it did not appear to produce an adverse functional outcome. Nevertheless, the presence of an increased slip at 2 years is a cause for concern and caution.
One prospective study assigned a group of 67 patients with spinal stenosis to either laminectomy or multilevel laminotomy and included a small subgroup of patients with DS.52 Nine of the patients assigned to the laminotomy group crossed over to the laminectomy group, which allowed for some difficulty in interpreting the results. However, no patient who underwent multilevel laminotomies developed instability as a result of the surgery, compared with three patients who developed instability following laminectomy. The authors recommended multilevel laminotomies for patients with developmental stenosis, mild to moderate degenerative stenosis, or DS. Bilateral laminectomy was recommended for patients with severe degenerative stenosis or marked DS.
Another recent study prospectively evaluated 54 consecutive patients who underwent decompression without fusion for spinal stenosis.53 In the small subgroup of 15 patients who had concomitant DS, 87% (13 of 15 patients) showed no change in the amount of preoperative slip. Overall, 88% of the 54 patients reported good/excellent clinical outcome and the results were comparable between patients with and without DS. The study concluded that degenerative spinal stenosis including patients with DS can be decompressed effectively without the need for fusion.
Noninstrumented Posterolateral Fusion
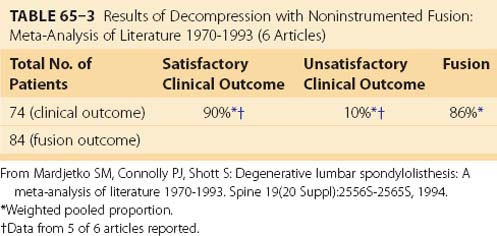
Although the beneficial role of fusion in the surgical treatment of spinal stenosis associated with DS is less controversial than the role of fusion in the treatment of other degenerative back conditions, incontrovertible evidence supporting fusion is sparse. An attempted meta-analysis of literature reported between 1970 and 1993 found only six studies meeting the inclusion criteria that reported results of decompression with noninstrumented fusion for DS.19 In that review, 90% of patients having decompression with noninstrumented fusion reported satisfactory clinical outcome and 86% achieved a solid arthrodesis, although the fusion rate varied widely, ranging from 3047 to 100% (Table 65–3).54 Patients undergoing decompression with noninstrumented fusion achieved a statistically significantly better clinical outcome than those treated with decompression alone (90% vs. 69%, respectively).
TABLE 65–3 Results of Decompression with Noninstrumented Fusion: Meta-Analysis of Literature 1970-1993 (6 Articles)
Many studies on the surgical treatment of DS report unfavorable outcome after decompression without fusion. One early, small study by two groups of surgeons from two different institutions included two populations of patients with spinal stenosis and DS: one group underwent decompression alone and the other had decompression and fusion.20 In the patients undergoing decompression alone, 5 of 11 (45%) were rated as good (satisfactory) and 6 of 11 (55%) as fair/poor (unsatisfactory). In contrast, 5 of 8 patients (63%) undergoing decompression with in situ posterolateral fusion achieved a satisfactory outcome. This study suggested that patients did better when their decompression was accompanied by noninstrumented fusion.
Several studies have supported the position that patients undergoing fusion with decompression for DS do clinically better than those undergoing decompression alone.20,48,55 It is difficult to gain a clear understanding of this issue from a review of existing literature, however, because well-done studies reporting surgical outcome after surgery for DS are uncommon.
Although most studies report no correlation between clinical outcome and the amount of slip progression, one study suggested that poor outcome was associated with slip progression.47 That study was a prospective randomized study that included a subgroup of 11 patients undergoing decompression and noninstrumented fusion for DS. Of the 10 patients available for follow-up, only 3 (30%) reported improved functional outcome and 7 had an increase in their preoperative spondylolisthesis, suggesting that slip progression was associated with poor clinical outcome.
A landmark prospective, randomized study comparing decompression alone with decompression and noninstrumented posterolateral spinal fusion in the treatment of L3-4 and L4-5 DS with spinal stenosis reported superior results when concomitant fusion was performed with the decompression.48 Satisfactory outcome was more than twice as common in the fused group compared with the unfused group (96% vs. 44%, respectively). Furthermore, the percentage of excellent results was significantly and dramatically greater in the fused group (44% excellent) than in the unfused group (8% excellent; P < 0.0001) (Table 65–4). This study concluded that the results of surgical decompression with in situ arthrodesis were superior to those of decompression alone in the treatment of spinal stenosis associated with L3-4 or L4-5 DS. Outcome was influenced by neither the age or sex of the patient nor the preoperative height of the disc space. The authors concluded that the decision for concomitant arthrodesis should be based purely on the presence or absence of a preoperative slip rather than on other preoperative factors such as the age or sex of the patient, disc height, or intraoperative factors such as the amount of bone resected during the decompression. This study showed that the results of decompression with an attempted arthrodesis produced superior results to decompression alone, even if the fusion was unsuccessful (pseudarthrosis). Although postoperatively there was a significant (P = 0.002) increase in the slip in patients not receiving an arthrodesis compared with those undergoing fusion, 36% of the arthrodesis group were also noted to have a pseudarthrosis, although all had an excellent or good result.
TABLE 65–4 Prospective, Randomized Comparison of Decompression vs. Decompression and Noninstrumented Spinal Fusion for Degenerative Spondylolisthesis
| Outcome | Arthrodesis (n = 25) | No Arthrodesis (n = 25) |
|---|---|---|
| Excellent | 11 (44%) | 2 (8%) |
| Good | 13 (52%) | 9 (36%) |
| Fair | 1 (4%) | 12 (48%) |
| Poor | 0 (0%) | 2 (8%) |
| Mean increase in slip (preoperative to postoperative) | 0.5 mm | 2.6 mm (P = .002) |
From Herkowitz HN, Kurz LT: Degenerative lumbar spondylolisthesis with spinal stenosis: A prospective study comparing decompression with decompression and intertransverse process arthrodesis. J Bone Joint Surg Am 73:802-808, 1991.
A long-term review of 96 patients undergoing decompressive surgery for spinal stenosis followed for at least 5 years included a subset of patients with associated DS.56 Although this subgroup was not fully analyzed separately and the study itself was retrospective, nonrandomized, and uncontrolled, some important trends were noted. Twenty-six patients (27%) of the entire group were considered failures: 16 because of recurrent neural symptoms and 10 because of low back pain. The incidence of DS was significantly greater in that surgical failure group (12 of 26 patients [46%]) than in the surgical successes (16 of 64 [25%]). The authors concluded that because of the higher incidence of recurrent symptoms in patients with preexisting DS, all patients with an associated slip should be fused.
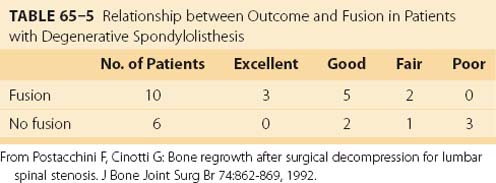
One well-recognized cause of long-term failure of decompression for spinal stenosis is subsequent bone regrowth causing recurrent neural compression. One study reported the relationship between bone regrowth, occurring an average of 8.6 years after surgical decompression for spinal stenosis, and long-term outcome.54 Of the 40 patients in the study, 16 had preoperative DS, 10 of whom had concomitant arthrodesis. Although all 16 patients with preexisting DS showed some bone regrowth, the degree of regrowth was less severe in the 10 patients undergoing arthrodesis than in the 6 patients who were not fused (Table 65–5). Furthermore, the proportion of satisfactory results was significantly higher in patients who had spinal fusion. Although this study was retrospective and not randomized, it suggested that arthrodesis stabilized the spine, resulting in less bone regrowth causing recurrent stenosis, and produced superior long-term results.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree