Bone Grafts in Hip Surgery
Paul Tee Hui Lee, Sandor Gyomorey, Oleg A. Safir, David J. Backstein and Allan E. Gross
Key Points
Introduction
Historical Perspective
Bone grafting is an ancient art. Anthropologist A. Jagharian at Erivan Medical Centre in Armenia examined a prehistoric Khuritic skull from the ancient Centre of Ishtkun and found a piece of animal bone filling a 7-mm defect with bony regrowth around the grafted bone.4 It has been suggested that the ancient Egyptians and Greeks attempted bone transplantation. In the modern age, the first documented bone graft was successfully performed in 1668 by a Dutch surgeon named Job Van Meekeren, who successfully inserted a fragment of a dog bone into the skull of an injured soldier.5 The first documented successful autograft transplant was performed in Germany in 1821 by Philips von Walter in experimentally created animal bone defects.7 The first documented allograft transplant was performed in 1879 by Sir William MacEwen in Scotland, who replaced the infected proximal two thirds of a humerus in a 4-year-old boy with the tibia from another child with rickets.6 In 1915, F. H. Albee published his work on autologous bone graft in the United States, which promoted the use of bone transplantation.8 In 1942, Inclan reported his experience in a large number of operations using autograft and allograft.9 Similar to other surgeons, Inclan considered the difficulties of finding suitable grafts for operations. When met with supply difficulties for autologous bone transplant, he used homologous bone graft between living patients of the same blood group. He did this because immune-related problems were known then, and graft from cadaver was prohibited on religious and sentimental grounds. Modern-day bone banks emerged in the 1960s and 1970s with improvements in cooling techniques combined with allograft processing; this led to the relative immunogenic impunity of allografts after transplantation. Several reports have helped to define safety and techniques in bone banking, thereby facilitating increased use of bone allografts.10–12 In recent times, despite its limitations, allogeneic bone graft is the most commonly used graft tissue.13 Several important clinical applications include restoration of bone stock in tumor surgery and in revision hip arthroplasty.
In hip surgery, the use of bone graft includes autografts, allografts, and bone substitutes. We will focus our discussion on the use of autologous and allogeneic bone grafts and will include various grafting techniques, their clinical applications and results, the basic scientific basis of these materials, current controversies, and future directions.
Bone Autograft
Clinical indications for the use of bone autograft include primary total hip replacement for hip dysplasia and protrusio to treat bone defects and achieve primary implant fixation stability and restore the center of rotation for the joint.1–3 Femoral head osteonecrosis can also be treated by vascularized and nonvascularized autologous bone grafts. Morselized autografts generally are used for treating smaller contained defects, and structural autografts are used for larger defects that may be uncontained. The resected host femoral head is the most frequently used form of autograft bone. It is readily available, adequate in size, cheap, biomechanically compatible, and easy to contour to fit defects, and it does not need processing. Total hip replacements performed in dysplastic hips using femoral head autograft have shown graft incorporation in most cases, with generally good clinical outcomes over the short term to midterm.1,17-27 Longer-term results have been mixed. Harris and associates reported a 10% revision rate (n = 47) at a mean of 7.1 years,1 a 20% revision rate (n = 46) at a mean of 11.8 years,19 and a 29% revision rate (n = 55) at a mean of 16.5 years.20 The authors expressed concern regarding low stem offset and posterior cup uncoverage in this cohort. Gross and colleagues21 reported a revision rate of 13% (n = 15) at a mean of 8.4 years’ follow-up and later, Nousiainen and co-workers27 reported a revision rate of 32% (n = 31) at a mean of 14 (8 to 18) years’ follow-up. Inao and associates22 reported 0% revision rate (n = 20) at a mean of 8.4 years; however, Iida and colleagues25 reported a 4% revision rate (n = 133) at a mean of 12.3 (8 to 24) years, and Akiyama and associates26 reported a 4% revision rate (n = 147) at a mean of 11.8 (6.3 to 15.4) years. Note that mean patient weight was 51 (29 to 78) kg in Iida’s cohort and 51.6 ± 7.9 kg in Akiyama’s. With regard to resorption of the autologous graft, results from various series were similarly mixed.17,19,21,27 Nevertheless, there is consensus among most authors that the bulk autograft incorporated well, restored pelvic bone stock, and facilitated revision surgery.
Allograft
Allografts, morselized or structural, have been used mainly to address significant bone loss in revision hip arthroplasty and post tumor resection.
Impaction Bone Grafting
Impaction bone grafting is used because it is considered a biological technique for restoring host bone stock. Metal wire meshes are used to enclose segmental bone defects, which then are filled with morselized bone allograft that is densely impacted before cementing of the acetabular or femoral component. Studies have indicated that the success of the construct depends upon graft incorporation, which relies on the quality of the host bone bed.
The choice of bone graft is controversial. A systematic review performed by the Cochrane Database demonstrated insufficient evidence to suggest differences between processed (freeze dried or irradiated) and unprocessed (fresh-frozen) bone for impaction grafting.119 However, it is recommended that the graft be used in the form of cancellous cubes that are 7 to 10 mm in width.
Results
Acetabular Side
The results of acetabular reconstructions using this technique for contained cavitary defects have generally been good, with survivorship approaching 85% to 90% at 20 years.28,29 Buma and colleagues31 took core biopsies from 8 patients after 1 to 72 months from surgery to show the different stages of graft incorporation. At 4 months after surgery, histologic analysis showed revascularization of the graft. Osteoclasts had removed parts of the graft, and woven bone had formed on the remnants of the graft and in the stroma that was invading the graft (Fig. 13-1). Between 8 and 28 months after surgery, the mixture of graft and new bone showed remodeling with time into normal trabecular bone structure with viable bone marrow that contained few or no remnants of the original graft (Fig. 13-2). Although some specimens showed the presence of graft-cement interface and local apposition of vital bone with the cement layer, a soft tissue interface predominated (see Fig. 13-2B and D).
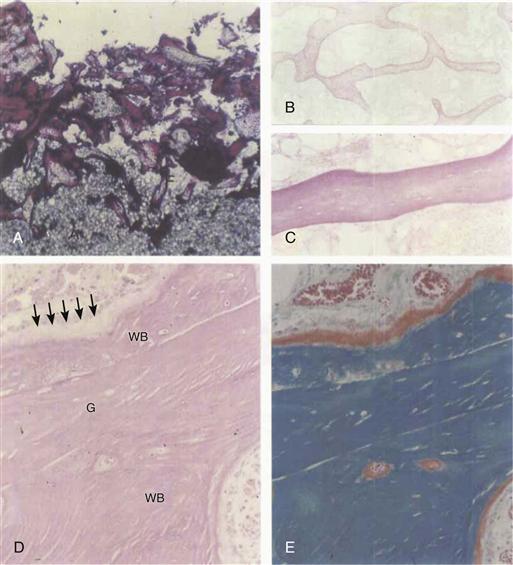
Figure 13-1 A through C, One month post acetabular reconstruction. A, Graft-cement interface in fuchsin-stained thick section. Note penetration of cement into the graft. B and C, No incorporation of graft in hematoxylin and eosin (H&E)-stained section. Note acellular medullary tissue in C (A and B, ×20; C, ×90). D and E, Case 2.4 months postoperatively. New woven bone (We) is formed on the remnants of the graft (G) by active osteoblasts (arrows). (D, H&E-stained section; E, Goldner-stained adjacent section.) Note red-stained osteoid indicating active bone formation (×225). (From Buma P, Lamerigts N, Schreurs BW, et al: Impacted graft incorporation after cemented acetabular revision: histological evaluation in 8 patients. Acta Orthop Scand 67:536–540, 1996, Fig. 2.)
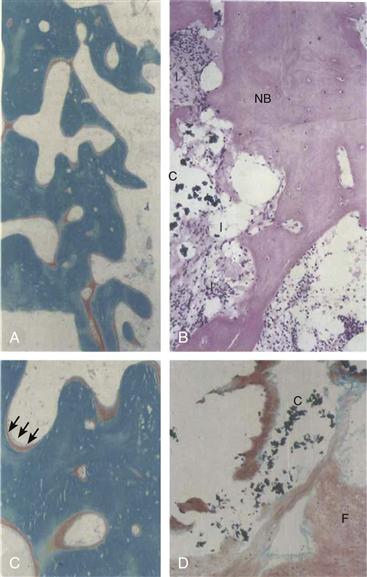
Figure 13-2 A through C, Eight months postacetabular reconstruction. The graft is incorporated into a new trabecular structure. If inspected with polarized light, the structure mainly consists of woven bone, with many active bone remodeling sites indicated by red osteoid staining (Goldner staining, ×30). C, Magnification of part of Figure 13-2A. Note active osteoblasts (arrows) (×55). B and D, 28 months postoperatively. At the graft-cement (C) interface, new bone (NB) is locally present, graft remnants are absent, and locally a soft tissue interface (I) and/or fibrocartilage (F) is present (hematoxylin and eosin [H&E] and Goldner staining, ×140). (From Buma P, Lamerigts N, Schreurs BW, et al: Impacted graft incorporation after cemented acetabular revision: histological evaluation in 8 patients. Acta Orthop Scand 67:536–540, 1996, Fig. 3.)
For uncontained segmental defects, concerns with impaction bone grafting include early mechanical failure, mesh rupture, frequent significant cup migration, and poor survivorship.32,33 Reports suggest that this technique is excellent for patients with small to moderate-sized contained cavitary defects, but poor for those with large uncontained segmental defects.
Femoral Side
Published reports have shown good clinical success in patients with significant cavitary femoral bone loss treated with impaction grafting34–37 and radiographic and histologic evidence of revascularization of impacted allograft.38
In a retrieval study of a well-fixed stem with good radiographic graft incorporation (Fig. 13-3), Ling and co-workers39 showed at 3.5 years’ follow-up that the grafted bone had become organized into three zones (Fig. 13-4): a newly regenerated cortical surface, an interface zone, and a deep zone. In the regenerated cortical surface, the bone was fully mineralized and revascularized. The fatty bone marrow was normal looking with no fibrosis. The bone was generally viable, as shown by more than 90% of filled osteocyte lacunae (Fig. 13-5), but occasional islands of dead bone were evident. The interface zone was irregular with direct contact between bone cement and osteoid. Foreign body giant cells were visible, and in some areas, there was a thin soft tissue lining (Fig. 13-6). No direct contact occurred between viable mineralized bone and cement. The deep layer included trabeculae of dead bone, entombed in cement at the time of its insertion. These trabeculae had empty lacunae but were linked to surrounding viable bone by bridges of soft tissue, osteoid, and bone (see Fig. 13-6).
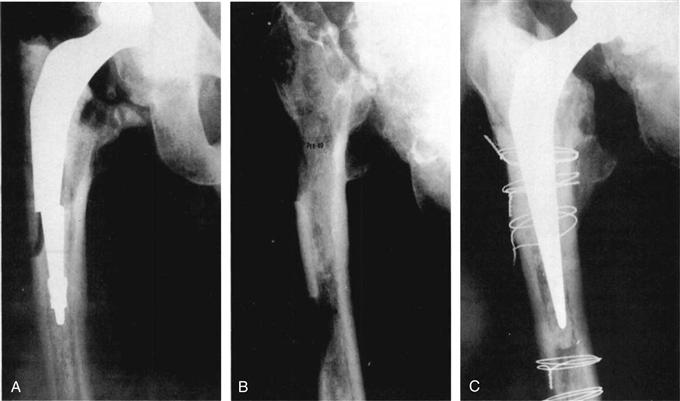
Figure 13-3 A, The fractured femoral stem with an adjacent cortical defect. B, The femur 1 year after removal of the prosthesis and cement. Two cortical defects are present: the distal one was created surgically to remove the distal cement. C, Two years after revision arthroplasty with impaction cancellous grafting. Cerclage wires, which secure the wire mesh, mark the levels of cortical defects; these appear to be filled by bone. (From Ling RS, Timperley AJ, Linder L: Histology of cancellous impaction grafting in the femur: a case report. J Bone Joint Surg Br 75:693–696, 1993, Fig. 1.)
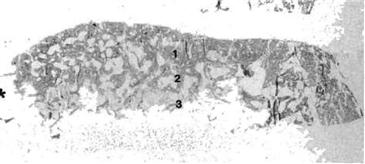
Figure 13-4 Low-power view of a histologic section taken at the level of the upper cortical window. Cement has been dissolved out, and staining indicates mineralized bone. At one point (*), cement has protruded between the cortex and the graft. The three zones of interest are shown: (1) cortical bone; (2) interface between living tissue and cement; and (3) bone trabeculae buried in the cement (Goldner stain, ×4). (From Ling RS, Timperley AJ, Linder L: Histology of cancellous impaction grafting in the femur: a case report. J Bone Joint Surg Br 75:693–696, 1993, Fig. 5.)
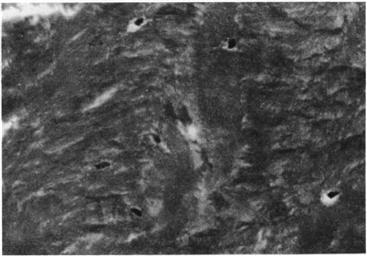
Figure 13-5 High-power view of regenerated bone showing filled osteocyte lacunae in a mineralized stroma with all the histologic characteristics of normal cortical bone (Goldner stain, ×600). (From Ling RS, Timperley AJ, Linder L: Histology of cancellous impaction grafting in the femur: a case report. J Bone Joint Surg Br 75:693–696, 1993, Fig. 6.)
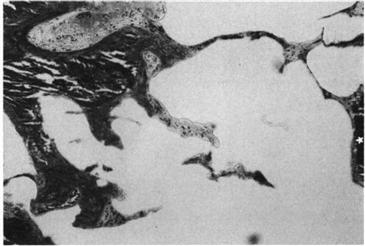
Figure 13-6 The interface between viable tissue and bone cement that has been dissolved out. Viable bone has been capped with osteoid, which is in direct contact with the (absent) cement surface. Some dead bone trabeculae (*) are probably remnants of the graft. Bridges of soft tissue and osteoid connect these trabeculae to living bone. Multinucleated giant cells are also seen in contact with the cement surface (Goldner stain, ×120). (From Ling RS, Timperley AJ, Linder L: Histology of cancellous impaction grafting in the femur: a case report. J Bone Joint Surg Br 75:693–696, 1993, Fig. 7.)
In cases with substantial and fully circumferential bone loss of the proximal femur, concerns with impaction grafting techniques remain because of the risk of prosthetic migration and periprosthetic fracture.40,41 Some would consider this a contraindication for impaction bone grafting. Few studies have shown reasonable results in this situation. Buttaro and associates42 reported the use of this technique for 15 cases of severe proximal femoral bone defects averaging 12 cm in length that were followed for an average of 43.2 months (range, 20 to 72 months). The Merle d’Aubigné and Postel score improved from a mean of 4.8 to 14.4 points, and implant survivorship was 100% at 1 year and 87% at 72 months. However, a high incidence of severe complications was reported, including 2 (13%) fractures, 3 (20%) dislocations, and 3 (20%) deep infections.
Structural Allografts
Structural allograft can provide adequate support for primary implant fixation stability in revision surgery with large uncontained segmental bone loss. Disadvantages include lack of universal availability, technical difficulties, and concerns over long-term graft resorption and collapse.
Acetabular Side
A minor column or shelf allograft is a structural graft that provides 50% or less support to the new acetabular component. The bulk graft is shaped intraoperatively, is snug-fitted into the defect, and is held with compression screws with washers (Fig. 13-7). Midterm results for treatment of uncontained segmental defects involving <50% of the acetabulum have been around 80% for cup survivorship at 10 years, with aseptic loosening as the endpoint.43,44 Similar results have been reported with the use of bulk femoral head and acetabular and distal femur allografts.43,44
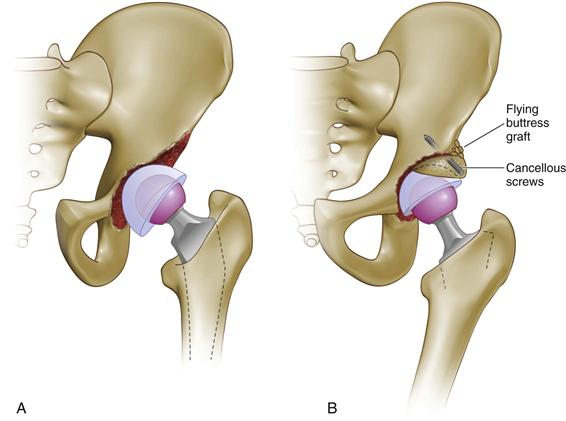
Figure 13-7 A, An uncontained defect involving less than 50% of the acetabulum. B, A minor column (shelf) graft supporting less than 50% of the cup is held with two cancellous screws. The flying buttress graft is cancellous autograft bone. (Redrawn from Gross AE, Duncan CP, Garbuz D, Mohamed MZ: Revision arthroplasty of the acetabulum in association with loss of bone stock. J Bone Joint Surg Am 80:440–451, 1998.)
Despite concerns about long-term graft resorption, collapse, and failure, mid- to long-term studies have shown that during rerevision surgery, most of the bulk allograft remained intact with host-graft boundaries obscured, enabling cup-only exchanges.43,44
A major column allograft is a structural graft that provides more than 50% support to the new acetabular component (Fig. 13-8). These allografts are used in revision cases with uncontained segmental defects involving more than 50% of the acetabulum and both columns, with or without pelvic discontinuity. The use of major column allografts without support by an anti–protrusio ilioischial cage has met with poor results, with short-term to midterm survivorships of 45% to 60%.45,46 Treatment of the same defects with cemented cups supported by reconstruction cages securely fixed to the ilium and ischium in conjunction has led to more encouraging midterm results, with survivorships of 77% to 87%.47,48 The cage protects the structural graft by providing pelvic stability in spanning across the ilium and ischium and by offloading the graft until bony interdigitation occurs.
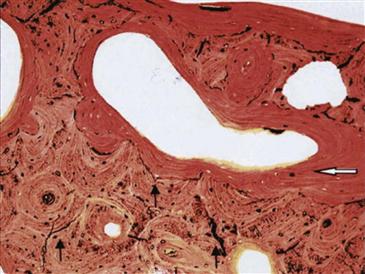
Figure 13-8 Low-power magnification photomicrograph of the allograft-host junction. Newly formed bone (white arrow) was present on the allograft surface and bridging to host. The microfractures (black arrows) are parallel or perpendicular to the newly formed osteons (van Gieson picrofuchsin, ×40). (From Hamadouche M, Blanchat C, Meunier A, et al: Histological findings in a proximal femoral structural allograft ten years following revision total hip arthroplasty: a case report. J Bone Joint Surg Am 84:269–273, 2002, Fig. 4.)
Hooten and colleagues49 examined two acetabuli that were revised with femoral head structural allografts and cementless cups at 25 and 48 months’ follow-up. Both grafts were functioning well at revision and were radiographically stable without evidence of graft collapse, cup loosening, or failure of union at the graft-host interface. Histologic examination showed that vascularity was increased at the host-graft interface, but evidence of bone union between the graft and the host was limited, in contrast to the radiographic appearance (Figs. 13-9 and 13-10). In areas where union had occurred, revascularization extended to 2 mm beyond the graft-host interface. Within the body of the graft, revascularization and remodeling were limited; the trabecular matrix appeared acellular, but the structural integrity was maintained at 48 months after surgery. In areas where the allograft was adjacent to an implant, fibrous tissue was oriented parallel to the implant surface. Bony ingrowth was limited to areas of porous coating that were in contact with viable host bone but did not occur between the ingrowth coating and the graft (Fig. 13-11).
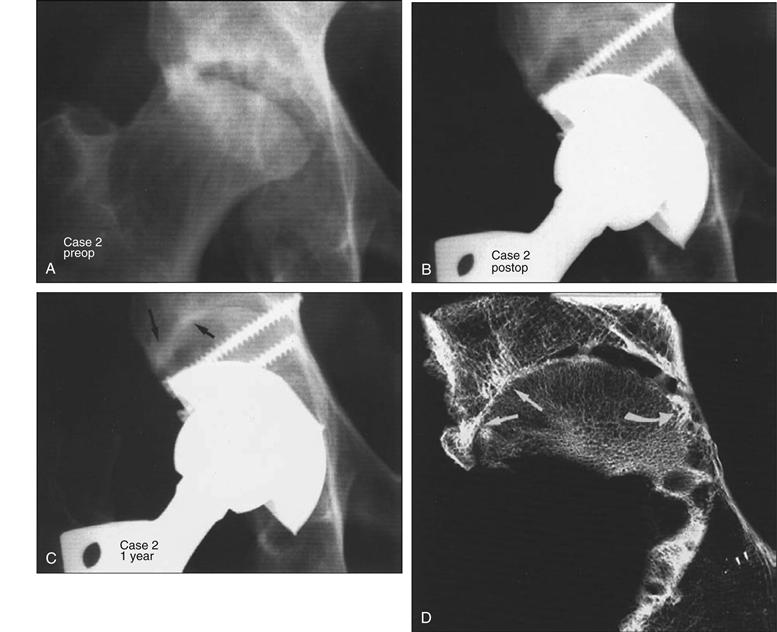
Figure 13-9 A, Preoperative radiograph showing superolateral acetabular deficiency with associated femoral head deficiency. B, Radiograph after femoral head allografting and cementless total hip arthroplasty. C, One year after surgery with trabecular bridging superolaterally (black arrows) and obscured interface medially, consistent with partial incorporation. D, Specimen radiograph showing persistence of the sclerotic border circumferentially around the femoral head allograft with partial graft healing in two areas (white arrows). (From Hooten JP Jr, Engh CA, Heekin RD, Vinh TN: Structural bulk allografts in acetabular reconstruction: analysis of two grafts retrieved at post-mortem. J Bone Joint Surg Br 78:270–275, 1996, Fig. 2A-D.)
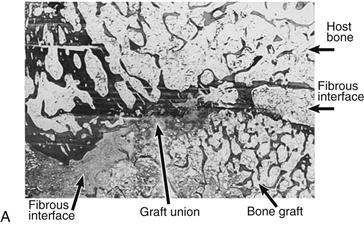
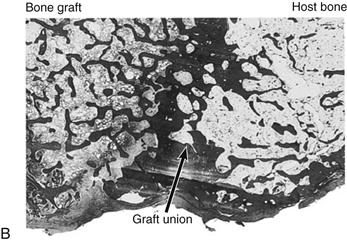
Figure 13-10 A, Appearance of the superolateral graft-host junction (area depicted by white arrows in Fig. 13-12D). B, Appearance of the superomedial graft-host junction (area depicted by black arrow in Fig. 13-12D) (hematoxylin and eosin, ×1.5). (From Hooten JP Jr, Engh CA, Heekin RD, Vinh TN: Structural bulk allografts in acetabular reconstruction: analysis of two grafts retrieved at post-mortem. J Bone Joint Surg Br 78:270–275, 1996, Fig. 4A and B.)
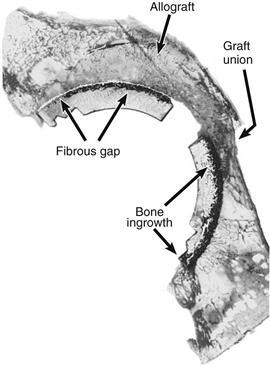
Figure 13-11 Graft-host junction (25-µm methylmethacrylate section, hematoxylin and eosin). (From Hooten JP Jr, Engh CA, Heekin RD, Vinh TN: Structural bulk allografts in acetabular reconstruction: analysis of two grafts retrieved at post-mortem. J Bone Joint Surg Br 78:270–275, 1996, Fig. 5.)
Femoral Side
Uncontained fully circumferential proximal femur segmental defects may be addressed with the use of a proximal femoral structural allograft in conjunction with a long-stem prosthesis. The stem is cemented into the proximal femur allograft to form an allograft-prosthesis composite. Fixation of the prosthesis to the host distal femur is maintained by a stem extension, which is implanted into the medullary canal and augmented with a step or oblique cut at the graft-host junction, multiple cerclage wires at the graft-host junction, and the optional addition of cortical (fibular or ulnar) strut grafts.
This method of treatment has shown good long-term survivorship and functional outcomes.50–53 Safir and associates54 reported 82% survivorship (n = 93) at a mean follow-up of 16 years (range, 15 to 22 years) with stem revision as the endpoint. Two (4%) infections, 6 (11%) aseptic loosenings, 3 (6%) nonunions, and 4 (7%) dislocations were reported. Complication rates were considered acceptable in view of the complexity of the clinical condition.
The main advantages of treatment with proximal femoral allografts over distally fixed proximal femoral replacements (cementless) include lower dislocation rates,55,56 better abductor function, less frequent Trendelenburg limp,57 and better survivorship with a lower rate of loosening of the distal stem.57,58
Hamadouche and colleagues,59 in a case report, showed the histologic appearance of a proximal femoral allograft via biopsies taken during stem revision for loosening after 10 years following surgery. Three layers were seen within the allograft: inner, intermediate, and superficial layers. The inner layer of the bone graft consisted of the zone of contact between the graft and bone cement and was composed of trabeculae of dead bone and cellular debris without evidence of remodeling (Fig. 13-12). The superficial or interfacial layer, corresponding to the region of contact between the graft and the host bone, was composed of areas of remodeling bone and osteoclasts, osteoblasts, and fatty bone marrow (see Fig. 13-8). Evidence of creeping substitution was noted with new bone and osteoid formation. Revascularization occurred through the formation of woven bone to a depth of 5 mm. Between inner and superficial layers within the substance of the graft lay the intermediate layer, which consisted of a sparsely cellular area with resorption and no remodeling. Of note, numerous microfractures were seen in the unremodeled area of the allograft, especially under high magnification, but not in the remodeled area. These microfractures were parallel or perpendicular to the bone lamellae and bypassed newly formed osteons at the level of the cement line.
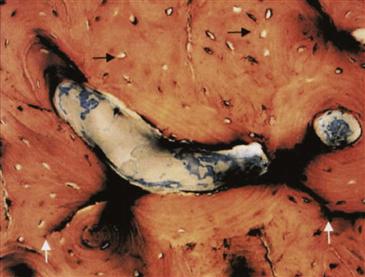
Figure 13-12 Photomicrograph of an unremodeled area of the allograft, showing empty lacunae (black arrows) and microfractures (white arrows). Cellular debris is present in the middle of the image (Stevenel blue and van Gieson picrofuchsin, ×100). (From Hamadouche M, Blanchat C, Meunier A, et al: Histological findings in a proximal femoral structural allograft ten years following revision total hip arthroplasty: a case report. J Bone Joint Surg Am 84:269–273, 2002, Fig. 3.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








