CHAPTER 3 Architectural Design and Function of Human Back Muscles
Spinal muscles generate movements of the spine and provide the stability needed to protect vital anatomic structures. Muscles must work in coordination with the rest of the neuromusculoskeletal system (i.e., vertebrae, tendons, ligaments, and the nervous system) to provide these functions. Large movements of the head require appropriate muscle strength, vertebral geometry (e.g., facet joint orientation), ligament compliance, and neural control. The spinal muscles have been described as one of three subsystems (the others are the passive spinal column and neural control) that must work together to stabilize the spine.1
Muscle Architecture
Skeletal muscle is highly organized at the microscopic level, as evidenced by classic studies that have elucidated the properties of skeletal muscle fibers. With few exceptions, the arrangement of muscle fibers within and between muscles has received much less attention. The microscopic arrangement of muscle fibers relative to the axis of force generation is known as the architecture of a muscle.2 Although muscle fiber size (which is directly proportional to force generation) is relatively consistent among muscles of different sizes, architectural differences between muscles show much more variability and more strongly affect function. Muscle architecture is a primary determinant of muscle function, and understanding this structure-function relationship has great practical importance. This understanding not only clarifies the physiologic basis for production of force and movement but also provides a scientific rationale for surgery. Muscle architectural studies also provide guidelines for electrode placement during electromyographic measures of muscle activity, explain the mechanical basis of muscle injury during movement, and aid in the interpretation of histologic specimens obtained from muscle biopsies.
Basic Architectural Definitions
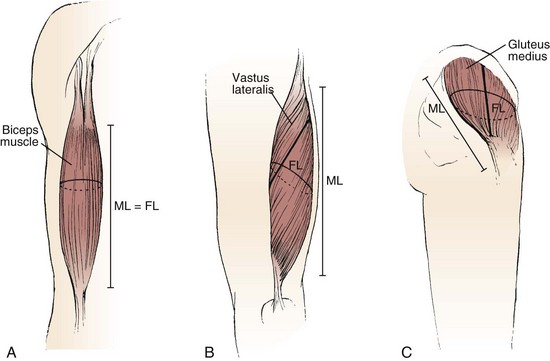
The various types of architectural arrangement are as numerous as the muscles themselves. For discussion purposes, we describe three general classes of muscle fiber architecture. Muscles composed of fibers that extend parallel to the force-generating axis of the muscle are described as having a parallel or longitudinal architecture (Fig. 3–1A). Muscles with fibers that are oriented at a single angle relative to the force-generating axis are described as having unipennate architecture (Fig. 3–1B). The angle between the fiber and the force-generating axis has been measured at resting length in mammalian muscles over different designs and varies from about 0 to 30 degrees. Most muscles fall into the third and most general category, multipennate muscles—muscles constructed of fibers that are oriented at several angles relative to the axis of force generation (Fig. 3–1C).
Experimental Determination of Skeletal Muscle Architecture
Quantitative studies of muscle architecture were pioneered by Gans and colleagues,2,3 who developed precise methodology for defining muscle architecture based on microdissection of whole muscles. The parameters usually included in an architecture analysis are muscle length, fiber or fascicle length, pennation angle (i.e., the fiber angle relative to the force-generating axis), and physiologic cross-sectional area (PCSA). Typically, muscles are chemically fixed in formalin to maintain fiber integrity during dissection. The muscles should be chemically fixed while attached to the skeleton to preserve their physiologic length, or physiologic length in the skeleton should be noted. After fixation, muscles are dissected from the skeleton, their mass is determined, and their pennation angle and muscle length are measured.
Pennation angle (θ) is measured by determining the average angle of the fibers relative to the axis of force generation of the muscle. Usually only the pennation angle of fibers on the superficial muscle surface is measured; however, this is only an estimate because pennation angles may vary from superficial to deep and from proximal to distal. This superficial to deep variation in pennation has been documented in several spinal muscles (see later). Although more sophisticated methods could be developed for measurement of pennation angle, it is doubtful they would provide a great deal more insight into muscle function because variations in pennation angle may not strongly affect function.2
Muscle length is defined as “the distance from the origin of the most proximal muscle fibers to the insertion of the most distal fibers.”4 Fiber length represents the number of sarcomeres in series, and experimental evidence suggests that muscle fiber length is proportional to fiber contraction velocity.3,5 Muscle length and fiber length are not the same because there is a variable degree of “stagger” seen in muscle fibers as they arise from and insert onto tendon plates (see Fig. 3–1B). Muscle fiber length can be determined only by microdissection of individual fibers from fixed tissues or by laborious identification of fibers by glycogen depletion on serial sections along the length of the muscle.6 Unless investigators are explicit when they refer to muscle fiber length, they are probably referring to muscle fiber bundle length (also known as fascicle length) because it is extremely difficult to isolate intact fibers, which run from origin to insertion, especially in mammalian tissue.6,7
Experimental studies of mammalian muscle suggest that individual muscle fibers do not extend the entire muscle length and may not even extend the entire length of a fascicle.6,7 Detailed studies of individual muscle fiber length have not been conducted in human spinal muscles, but studies in feline neck muscles illustrate that muscle fibers are often arranged in series, ending in tendinous inscriptions within the muscle or terminating intrafascicularly.8,9 Although the terms fiber length and fascicle length are often used interchangeably, technically they are identical only if muscle fibers span the entire length of a fascicle. In muscle architecture studies, bundles consisting of 5 to 50 fibers are typically used to estimate fiber length, which may be reported as either fiber length or fascicle length.
To permit such conclusions, fiber length measurements should be normalized to a constant sarcomere length, which eliminates fiber length variability owing to variation in fixation length. Fiber (or fascicle) lengths are usually normalized to the optimal sarcomere length, the length at which a sarcomere generates maximum force. This normalized length is referred to as optimal fiber (or fascicle) length and provides a reference value that can be related back to the physiologic length if the relationship between muscle length and joint position is noted. Based on measured architectural parameters and joint properties, the relationship between sarcomere length and joint angle can be calculated. Because sarcomere length strongly influences muscle force generation, an understanding of the relationship between sarcomere length change and movement has been used in many studies to provide added understanding of muscle design.10–14
Mechanical Properties of Muscles with Different Architectures
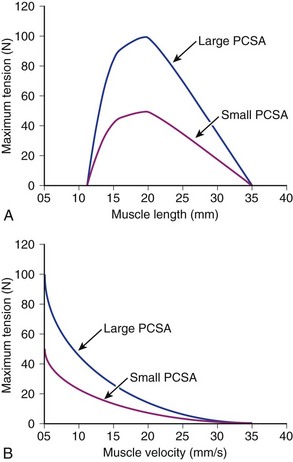
Comparison of Two Muscles with Different Physiologic Cross-Sectional Areas
Suppose that two muscles have identical fiber lengths and pennation angles but one muscle has twice the mass (equivalent to saying that one muscle has twice the number of fibers and twice the PCSA). Also suppose that the two muscles have identical fiber type distributions and that they generate the same force per unit area. The functional difference between these two muscles is shown in Figure 3–2. The muscle with twice the PCSA has an isometric length-tension curve with the same shape, but it is amplified upward by a factor of 2. The maximal tetanic tension (Po) of the larger muscle would be twice that of the smaller muscle. Similarly, comparison of isotonic force-velocity curves indicates that the differences between muscles would simply be an upward shift in Po for the larger muscle.
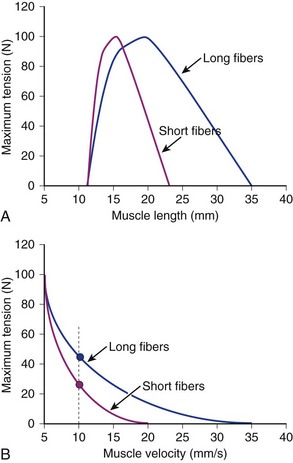
Comparison of Two Muscles with Different Fiber Lengths
If two muscles have identical PCSAs and pennation angles but fiber lengths that differ, the schematic in Figure 3–3 shows that the effect of increased fiber length is to increase muscle excursion and velocity. Peak force of the length-tension curves is identical between muscles, but the range of lengths over which the muscle generates active force is different. For the same reason that an increased fiber length increases active muscle range of the length-tension relationship, it results in an increase in the maximum velocity (Vmax) of the muscle. Experimental support for this concept was obtained indirectly through observations of the cat semitendinosus muscle:4 When the proximal semitendinosus head was activated, its Vmax was 224 mm/sec, whereas when only the distal semitendinosus head was activated, its Vmax was 424 mm/sec. When both heads were activated simultaneously, the whole muscle Vmax was 624 mm/sec, or the sum of the two velocities. The values for Vmax were proportional to the different lengths of the proximal and distal heads. These data indicate that the longer the fibers in series (equivalent to saying the greater number of sarcomeres in series), the greater the muscle contraction velocity. As expected, maximum isometric tension was essentially the same regardless of which activation pattern was used.
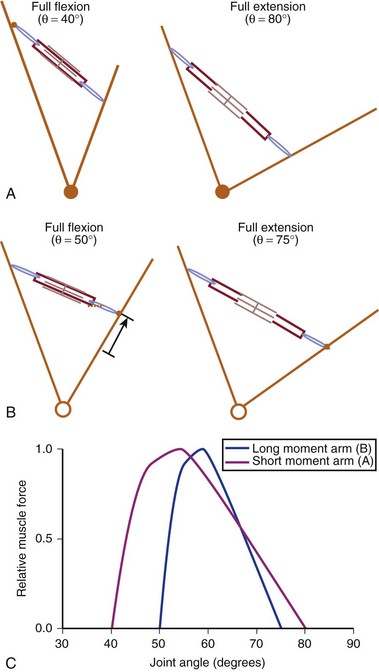
Interplay of Muscle Architecture and Moment Arms
In addition to its architecture, the potential moment generated by a muscle is influenced by its moment arm. Moment arm, the “mechanical advantage” of a muscle, is the distance from the line of action of a muscle to the joint axis of rotation and is directly related to a muscle’s change in length with joint rotation.15 In other words, the amount of muscle fiber length change that occurs as a joint rotates and, consequently, the range of joint angles over which the muscle develops active force depend on the muscle moment arm. This idea can be explained by comparing the situation in which two muscles with identical fiber lengths have different moment arms at a joint (Fig. 3–4). In the case in which the moment arm is greater, the muscle fibers change length much more for a given change in joint angle compared with a muscle with a shorter moment arm. As a result, the range of joint motion over which the muscle develops active force is smaller for the muscle with the larger moment arm despite the fact that the muscular properties of both muscles are identical.
The architectural design of a muscle and its placement in relation to the skeletal geometry are important determinants of its function. Although muscles with longer fibers can generate force over a greater range of lengths than muscles with shorter fibers (see Fig. 3–3A), this does not indicate that muscles with longer fibers are associated with joints that have larger ranges of motion. Muscles that appear to be designed for speed based on their long fibers may not actually produce large joint velocities if they are placed in the skeleton with a very large moment arm because joint excursion and joint angular velocity are inversely related to moment arm. A large moment arm results in a large joint moment, so that the muscle would be highly suited for torque production but at low angular velocities. Similarly, a muscle that appears to be designed for force production because of the large PCSA, if placed in position with a very small moment arm, may actually produce high joint excursions or angular velocities. Differences between muscle-joint systems require complete analysis of joint and muscular properties. These interrelated concepts of architecture and moment arm (gross anatomy) must be considered when examining the design and function of spinal muscles.
Anatomy and Architecture of Spinal Musculature
Intrinsic Spinal Muscles Found in the Lumbar, Thoracic, or Cervical Spine
Intrinsic muscles of the spine are dominated by the erector spinae, a group of interdigitated muscles that spans the entire length of the spine, from the sacrum and iliac crest to the skull. Another important group of muscles, the multifidus, are shorter and deeper and are described in more detail later. In the thoracolumbar region, the erector spinae and multifidus muscles constitute the bulk of the spinal musculature. These two distinct functional units have large differences in innervation that probably result in significant functional differences,16 although the detailed biomechanical function of these groups remains only partially elucidated.17 Lying deep to the multifidus are even smaller muscles, the rotators, interspinales, and intertransversarii. The cervical region is composed of other intrinsic muscles unique to it (see later).
The erector spinae are commonly considered to be composed of three muscles; from medial to lateral, they are the spinalis, longissimus, and iliocostalis. The anatomy and architecture of these muscles vary among different levels of the spine. The words “lumborum,” “thoracis,” “cervicis,” and “capitis” are appended to the muscle name to describe the anatomy more accurately. Although there are varying definitions of the composition of the erector spinae, a study by MacIntosh and Bogduk17 provides the most comprehensive descriptive anatomy of the lumbar erector spinae, and Delp and colleagues18 provided the first architectural measurements of these muscles. The continuation of the erector spinae in the cervical region was discussed briefly by Kamibayashi and Richmond.19
The spinalis muscle is the most medial division of the erector spinae. MacIntosh and Bogduk17 described the spinalis as mostly aponeurotic in the lumbar region, but Delp and colleagues18 obtained architecture measurements from the spinalis in the thoracic region (Table 3–1). The spinalis is generally absent in the cervical region.
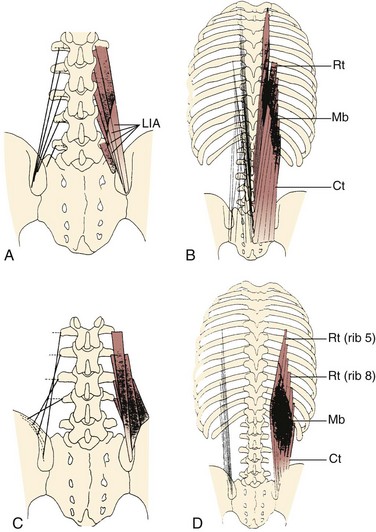
Caudal to rostral, the longissimus consists of the longissimus thoracis, cervicis, and capitis; the longissimus thoracis is divided into lumbar and thoracic portions. The lumbar fascicles of the longissimus thoracis (longissimus thoracis pars lumborum) are composed of five bands that arise from the lumbar transverse processes and attach in a caudal fashion onto the iliac crest (Fig. 3–5A). Each band arising from vertebra L1 to L4 is actually a small fusiform muscle that has an elongated and flattened caudal tendon of insertion. Bands from more rostral levels attach more medially on the iliac crest. The juxtaposition of these caudally located tendons form the lumbar intermuscular aponeurosis (see LIA in Fig. 3–5B).
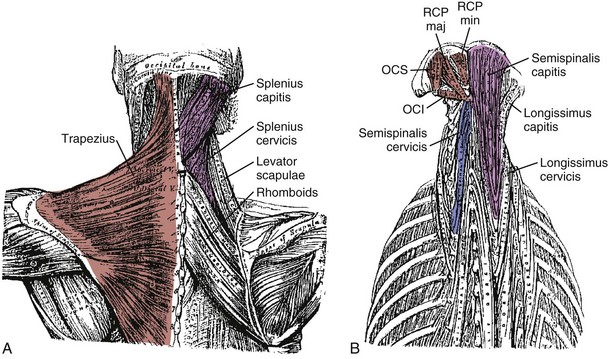
Fascicles of the thoracic component of longissimus thoracis (longissimus thoracis pars thoracis) arise from all thoracic transverse processes and most ribs and attach to lumbar spinous processes, the sacrum, or the ilium. These are long slender muscles with pronounced caudal tendons that juxtapose to form the strong erector spinae aponeurosis, which bounds the lumbar paraspinal muscles dorsally. In the upper thoracic and cervical region, the longissimus cervicis connects transverse processes of thoracic and cervical vertebrae, whereas the longissimus capitis originates on transverse processes and inserts on the mastoid process of the skull (Fig. 3–6).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree