CHAPTER 21 Advanced Shoulder Arthroscopy
EVOLUTION OF SHOULDER ARTHROSCOPY
Shoulder arthroscopy started by exploring and dealing with shoulder pathologies in the glenohumeral space.1 The limits of the glenohumeral joint are the glenoid, the humeral head, the capsule and the musculotendinous structures. It contains the cartilage, the glenohumeral ligaments, the labrum, and the tendon of the long head of the biceps. The first reconstructive procedure was the arthroscopic treatment of anterior shoulder instability.2
The second step of arthroscopic shoulder surgery extended into the subacromial space, with the treatment of impingement by arthroscopic acromioplasty.3 Because superior cuff tears create a communication between the subacromial and glenohumeral space, arthroscopic rotator cuff repair started with dealing with supraspinatus tendon tears.4 Further development led to the arthroscopic repair of large superior tears and later to the exploration and repair of the posterior (infraspinatus and teres minor tendon) and the anterior (subscapularis tendon) rotator cuff.
THE SHOULDER AS A HOUSE
The house has a first floor, a second floor, and a basement.
Medially, the coracoid process is a key landmark with the attachment of two musculotendinous structures: the pectoralis minor and the conjoint tendon. These structures allow defining the infracoracoid and supracoracoid spaces. The infracoracoid space is the second room on the first floor anterior and medial to the glenohumeral joint. It contains laterally the presubscapular bursa and more medially the plexus including the axillary nerve and the subscapularis nerves. The important medial border of the presubscapular bursa is at the level of the medial border of the coracoid process. The ceiling of this infracoracoid space is the coracoid process and the pectoralis minor tendon. The conjoint tendon is the anterior wall and the subscapularis tendon is the posterior wall.
THE ARTHROSCOPIC PORTALS
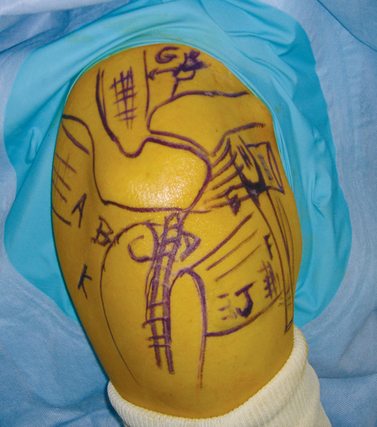
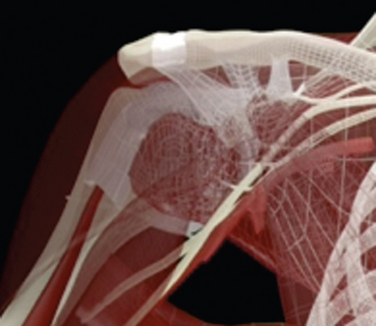
Since the beginning of shoulder arthroscopy, many portals have been described. Some are routinely used and others provide more specific access. We name the different portals in a standardized way (Fig. 21-1). Five portals, named A to E, surround the acromion.
Posteriorly the K portal is the posterolateral inferior portal, located lateral and inferior to the B portal. To avoid the anterior branch of the axillary nerve, it is placed lateral to the posterior border of the deltoid muscle and not lower than 6 cm from the acromion. This portal provides access to the axillary pouch for the repair of inferior humeral avulsion of the glenohumeral ligament (HAGL lesion), interposition arthroplasty, latissimus dorsi transfer, or simply removing loose bodies.
SHOULDER PATHOLOGY BY AREA
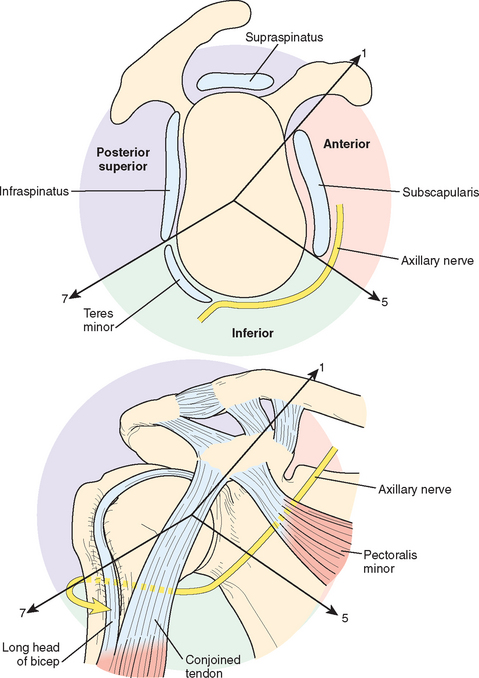
The concept of the shoulder as a house helps the surgeon to create a 3D perspective of the shoulder. The classic strategy for addressing disorders in the shoulder is to think by pathology, but it is also possible to think topographically. We divide this house into three distinct anatomic areas: the posterosuperior, the anterior, and the inferior areas. The safe zone, meaning a nerve-free zone, is laterally from the glenoid in the anterior and posterosuperior area (Fig. 21-2).
Three lines originating in the center of the glenoid and directed to the base of the coracoid and the inferior borders of the subscapularis and teres minor, respectively, together with the central axis going through the glenoid center, describe three planes. The three planes separate the three areas within the 3D shoulder. The plane through the coracoid base separates the anterior from the posterosuperior area, the plane through the inferior border of the subscapularis separates the anterior from the inferior area, and the plane through the inferior border of the teres minor separates the posterosuperior area from the inferior area.
Anterior Shoulder Area
The oblique plane of the coracoid base and the oblique plane of the inferior border of the subscapularis muscle span the space of the anterior shoulder area. Regarding the glenoid, this area spans from the 1-o’clock to the 5-o’clock position (see Fig. 21-2). Everything outside—meaning lateral to the glenoid—is considered a safe zone in terms of nerves.
Starting at the center, the anterior glenoid is followed by the anterior capsulolabral complex and the subscapularis tendon. Only the superior third of the subscapularis, being the largest and most powerful muscle of the rotator cuff,5 is visible from the intra-articular position. Its origin broadly covers most of the anterior scapula, and it inserts on a large footprint area that practically covers the entire lesser tuberosity. More medially is the coracoid process, with the attachment of four structures: the conjoint tendon, the coracoacromial ligament, the coracoclavicular ligament, and the pectoralis minor tendon. Medial to the coracoid process, the plexus and its vascular components rest on the subscapularis muscle. Under the subscapularis, the axillary nerve travels through the quadrilateral space in the inferior area. More lateral is the long head of the biceps in its bicipital groove, leading down to the humeral attachment of the pectoralis major.
In the anterior shoulder, placement of the arthroscope through an adequate anterolateral D portal allows simultaneous access to the intra- and extra-articular anterior shoulder (Fig. 21-3). This is only possible by opening the rotator cuff interval between the coracohumeral ligament and the subscapularis tendon. This concept is very important to note. It allows superior orientation not only for placing sequential portals safely but also for locating the neurovascular structures to shuttle between the intra- and extra-articular spaces.
Cartilage Lesions (Interposition Capsuloplasty)
Biological resurfacing of the glenoid and humeral hemiarthroplasty as an open surgical treatment for osteoarthritis in younger and active patients have been performed with acceptable results.6–8 At present, arthroscopic treatment of cartilage lesions of the glenoid with labral lesions is limited to reconstructions by covering the cartilage defect as much as possible by the labrum. In some institutions, GraftJacket is used to cover the glenoid cartilage defect arthroscopically, but no long follow-up is available regarding the outcome of this artificial component.9
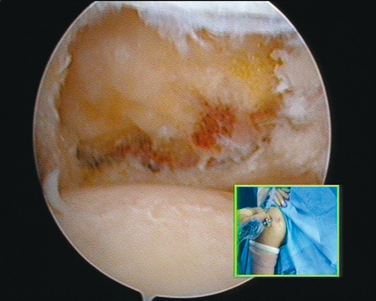
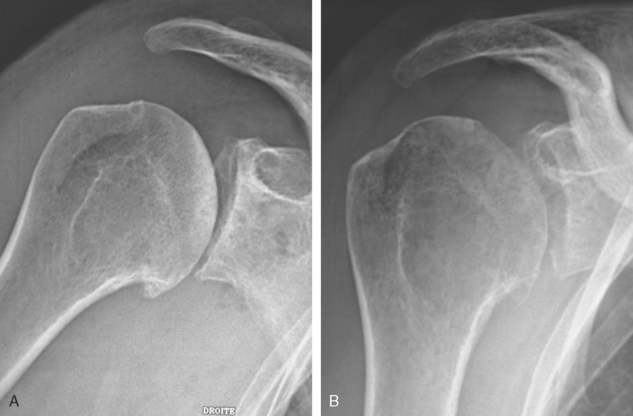
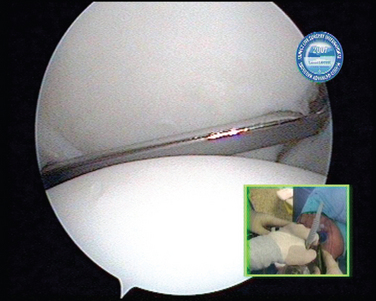
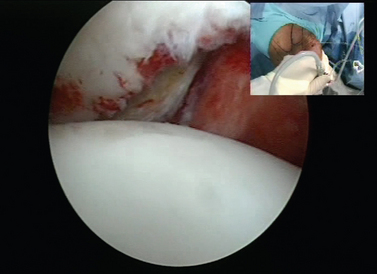
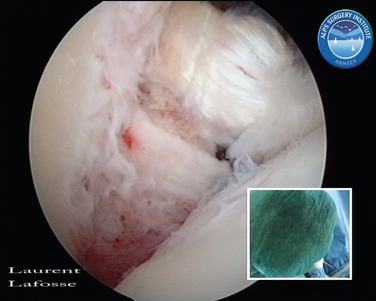
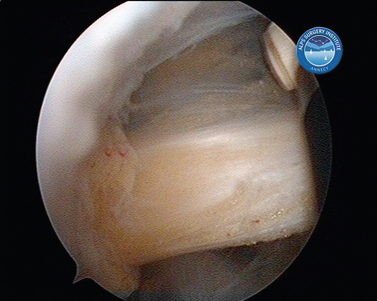
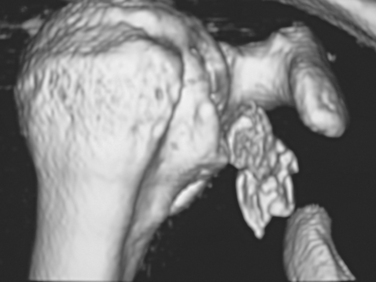
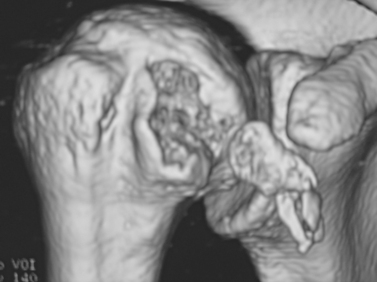
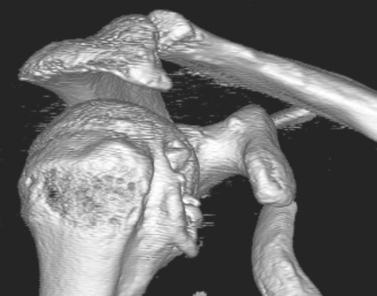
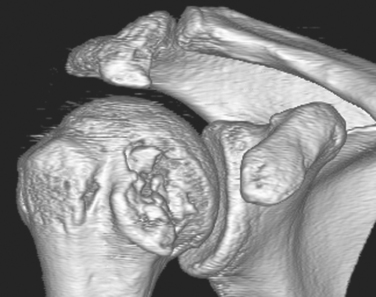
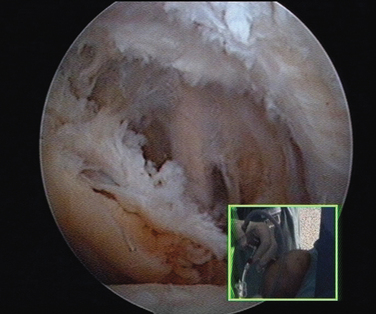
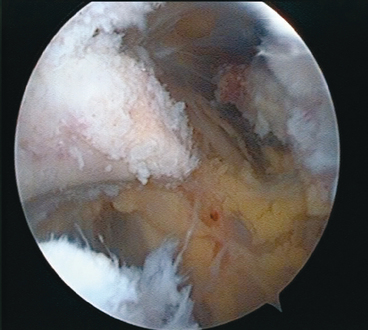
Encouragement by capsular interposition with hemiarthroplasty forged the arthroscopic interposition capsuloplasty. The technique is based on the concept of a capsular flap. It is a unique concept of resurfacing that addresses cartilage lesions selectively on the glenoid side. This seems a logical evolution, because the humeral side is not always the cause of pain and malfunction. We define the indication for this procedure to be younger patients with severe cartilage defects (Fig. 21-4) of the glenoid with or without involvement of the humeral side. The procedure is restricted to patients with only minor osteophytic changes.
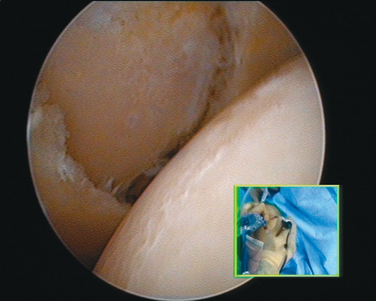
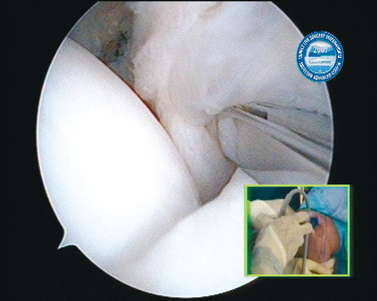
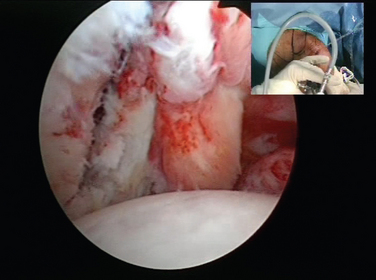
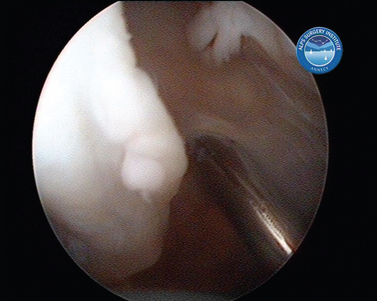
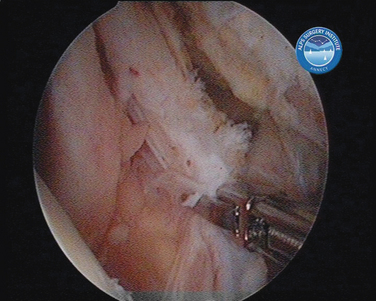
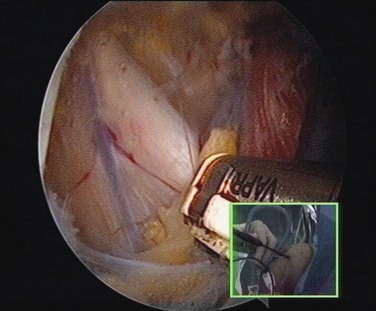
In shoulder instability, the cartilage lesion is caused by trauma from dislocation or is a consequence of chronic wear in persistent instability. In our experience, cartilage erosion associated with anterior or posterior instability is mostly limited to the anterior or the posterior cartilage area, whereas in idiopathic arthritis, the cartilage degeneration often starts inferiorly before becoming a global glenoid erosion. The capsular harvesting site for the interposition capsuloplasty is managed according to the area of the glenoid cartilage defect. The capsulolabral tissue is detached from both the glenoid (Fig. 21-5) and the humeral side (Fig. 21-6). The aim is always to apply the capsulolabral flap on an abraded bony surface, leaving it partially attached to the surrounding connective tissue. This has two major advantages: The biological graft only needs to be attached to the glenoid defect, and its vascularization is preserved.
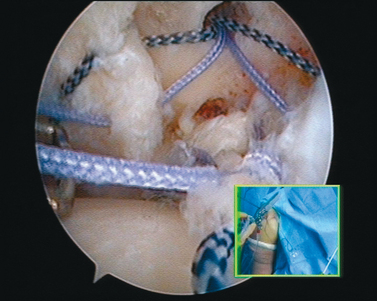
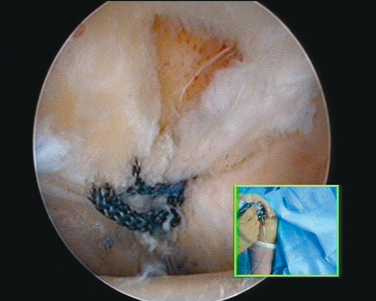
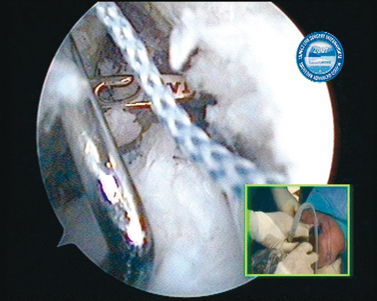
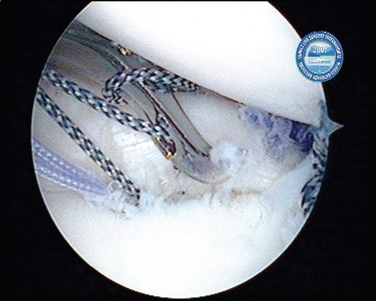
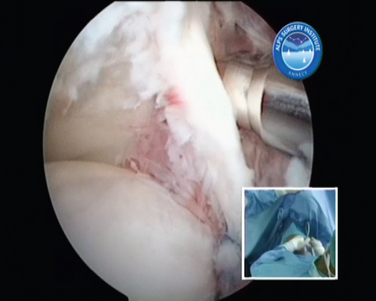
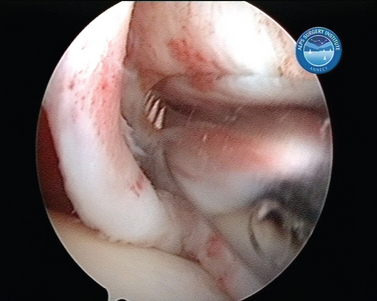
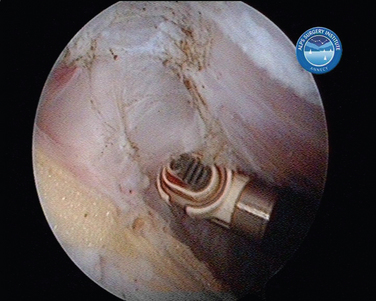
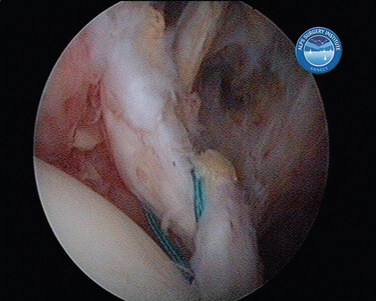
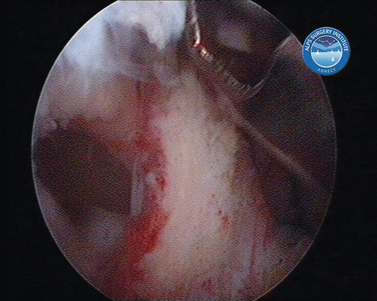
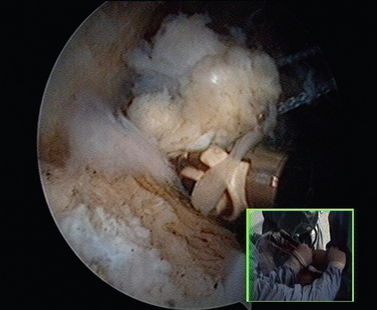
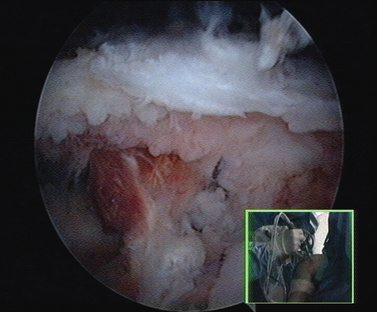
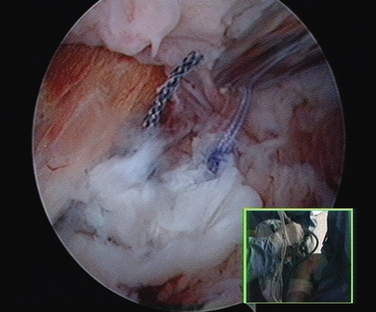
When the cartilage is missing over the entire glenoid (see Fig. 21-4), the entire glenoid is abraded (Fig. 21-7) and the anterior, posterior, and inferior capsule is used (Fig. 21-8) and fixed with three anchors placed along a vertical line in the middle of the glenoid. The anchor sutures are applied in a side-to-side lasso-loop fashion (Figs. 21-9 and 21-10). This allows a strong fixation of the three flaps one to another and to the glenoid surface. This procedure is performed with a superior view through the D portal (Fig. 21-11) and with instrumentation through the A and E portals (Fig. 21-12). Preoperative and postoperative radiographic views are shown in Figure 21-13.
Capsulolabral Double Row Fixation (Cassiopeia)
Most arthroscopic techniques address and repair only the labral aspect, employing a single-row technique. The technique in terms of anchor placement and reduction of the labrum—more on the cartilage, on the rim, or further medial—differs from surgeon to surgeon. The amount of capsulolabral tissue taken and shifted with the sutures varies.
To our knowledge, no present arthroscopic technique restores the insertion in an anatomic fashion. More specifically, the capsulolabral unit is generally only fixed to a narrow line. In contrast, open Bankart procedures do fix the unit not only on the glenoid rim but also to the medial scapular neck. This difference might even explain the reports of a relatively higher failure rate with arthroscopic single-row techniques.10
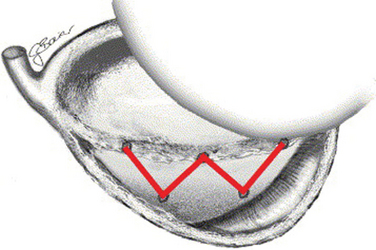
We use a double-row fixation technique with two transglenoidal suture points on the scapular neck and three anchor fixations on the glenoid rim. The fixation points create a W-shaped pattern, resembling the Cassiopeia constellation (Fig. 21-14). This way, the capsulolabral complex is fixed to a large surface on the anterior glenoid rim and the anterior scapular neck, restoring the physiologic insertion.
With this technique, we are able to combine an intra- and extra-articular procedure. A medial extracapsular fixation, being the hypothesized advantage of open repair, is managed arthroscopically and combined with a lateral anchor fixation to the glenoid rim. It allows the inferiorly displaced anterior bundle of the inferior glenohumeral ligament to be shifted back superiorly and permits a strong fixation of the entire anteroinferior capsulolabral complex.
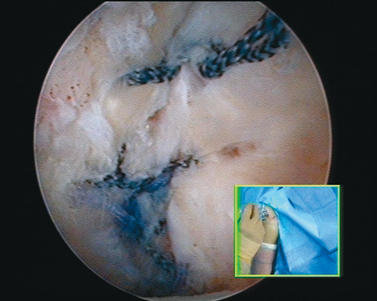
Technically, the anterior access of the medial row is difficult due to the anteversion of the glenoid and the neurovascular structures and conjoint tendon blocking a perpendicular approach. We therefore use a retrograde technique. The medial row fixation consists of a suture shuttled through two trans-scapular bone tunnels and through the medial capsule. The tunnels are created from posterior to anterior with a dedicated target device (Fig. 21-15) in order to control the position of the tunnel. The instrumentation further allows protection of the suprascapular nerve posteriorly by the cannulated system (Figs. 21-16 and 21-17). Anteriorly, the capsule can be lifted off the anterior scapula neck, allowing complete visual control as the guidewires exit the bone. In correct application, there is no danger to the brachial plexus and its accompanying vessels. The two ends of a braided suture are shuttled retrograde through the bone tunnels (Fig. 21-18). In the next step, they are passed through the capsule far inferiorly and medially (Fig. 21-19). Only after the labrum is reinserted with the lateral row at the glenoid rim using suture anchors (Fig. 21-20) are the medial row sutures tightened.
The entire procedure is visualized through a suprabicipital D portal. This allows visual access to the anterior and posterior glenoid, the scapular neck, and the space between the subscapularis muscle and the capsulolabral complex for management of the medial sutures.
Bony Fractures of the Glenoid Rim
Sufficient methods have been developed for treating small fractures. Dealing with them is an integrated part in suture anchor refixation of the labrum.11 In cases of recent fracture, the refixation of the capsulolabral bone unit is performed. In old fractures, resection of the fragment is favored, despite possible risk of chronic instability due to the loss of glenoid support. In a more functional glenoid defect and in a high-demand patient, we consider the arthroscopic Latarjet procedure (see later) for bone reconstruction.
Open surgical treatment through a direct anterior approach requires detachment of the subscapularis. For sufficient arthroscopic access, the axillary nerve and artery are at risk.12 The anterograde transaxillary mini-invasive approach is therefore known to be associated with neurovascular complications.13 A direct arthroscopic approach is further hindered by the coracoid and the conjoint tendon, which course even farther laterally as they descend (Fig. 21-21). Medial to those structures lies the brachial plexus. This prevents the arthroscopist from directly accessing the anterior or inferior glenoid through the interval or subscapularis in a perpendicular fashion as needed for screw fixation.
We have, concurrently with others,14 developed for larger fractures of the glenoid rim (Fig. 21-22) a transglenoid technique. In contrast to other groups, we use a retrograde approach, avoiding the difficulties encountered with an anterior approach and avoiding neurovascular complications. Ours is an arthroscopic approach for screw fixation based on the Cassiopeia technique15 (see earlier). Furthermore, it mediates a perpendicular approach to the fracture surface. This allows a congruent reduction of the often triangular fragment to the glenoid.
Visualization for this technique is performed through the suprabicipital D portal. For temporary fixation, guidewires are brought in a retrograde fashion through the scapular neck, perforating the fragment through the posterior A portal. During this step, the fragment needs to be reduced and stabilized with a tissue grasper passed through the E portal. We use a cannulated screw system for final fixation. Complementary fixation is achieved by soft tissue attachment through suture anchors. This technique does not permit compression of the fracture, but it offers a safe reduction and a solid scaffold (Fig. 21-23).
Anterior HAGL Lesions
Arthroscopically, anterior HAGL lesions are located at the lesser tuberosity deep and just medial to the subscapularis insertion and continue to the anatomic neck (Figs. 21-24 and 21-25). A high index of suspicion for an underlying anterior HAGL lesion should be present in all cases with a contradiction between a history of post-traumatic recurrent dislocations and no or limited capsulolabral lesions at the glenoid side. Open surgical repair through an anterior approach requires detachment of the subscapularis tendon.16,17 Only a few reports exist on arthroscopic HAGL repair.18,19
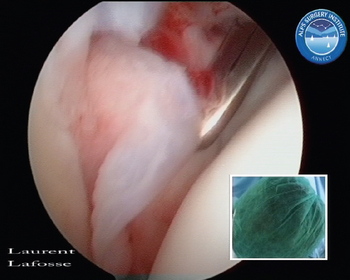
FIGURE 21-24 Posterior view of anterior humeral avulsion of the glenohumeral ligament (HAGL lesion). Inset, External view.
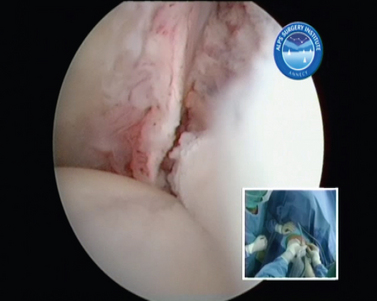
FIGURE 21-25 Anterolateral view of anterior humeral avulsion of the glenohumeral ligament (HAGL lesion). Inset, External view.
The repair is visualized through the posterior A and the anterolateral D portal above the subscapularis tendon by opening the rotator cuff interval and partially resecting the coracohumeral ligament. A single-row suture anchor repair through the anterior E portal is applied (Figs. 21-26 to 21-29). To enhance the exposure, the arm can be brought into slight internal rotation. Possible difficulties encountered with arthroscopic treatment are due to the narrow space at the humeral insertion, poor tissue quality, or retraction. If the given circumstances do not allow a sufficient anatomic reconstruction, an arthroscopic Latarjet procedure (see later) is proposed as a nonanatomic alternative stabilization procedure to prevent failure or stiffness.
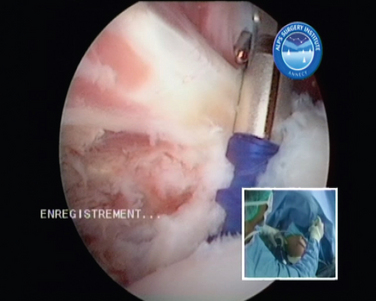
FIGURE 21-26 Repair of anterior humeral avulsion of the glenohumeral ligament (HAGL lesion) I. Inset, External view.
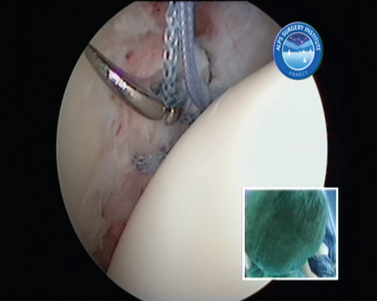
FIGURE 21-27 Repair of anterior humeral avulsion of the glenohumeral ligament (HAGL lesion) II. Inset, External view.
Subscapularis Pathologies
Recognition and treatment of lesions of the subscapularis tendon are more recent in comparison with the other rotator cuff tendons. We know now that the impact of subscapularis tears is very important. It is not only the single internal rotator and anterior constraint of the rotator cuff, but also in diameter and strength it is the most important muscle of the rotator cuff. Failure of its function can therefore insufficiently be compensated and is associated with anterior instability and deterioration of the shoulder function.
So to manage all subscapularis tears arthroscopically, we developed an extra-articular approach. As a function of the stage of the rupture, gradual steps are necessary to obtain a repair. We propose a classification system of rupture stages, and we base the surgical steps needed for repair on these stages (Table 21-1).20 Type I tears are localized to the superior third of the subscapularis tendon and are partial tears of the deep fibers at the insertion onto the lesser tuberosity. These tears never display tendon retraction because the superficial fibers of the subscapularis remain intact. Type II tears are complete ruptures of the superior third of the tendon. Type III tears are complete tears of the superior two thirds of the tendon. The intact inferior one third of the tendon still limits the degree of retraction. Type IV tears are complete tears of the entire subscapularis tendon from its insertion, combined with retraction of the tendon edge to the level of the glenoid rim without anterior subluxation of the humeral head. Type V tears are complete tears of the subscapularis tendon with retraction and an eccentric humeral head, anteriorly subluxated, with possible impingement on the coracoid process.
TABLE 21-1 Classification of Subscapularis Tendon Tear on the Basis of Preoperative CT and Intraoperative Evaluation
| Type | Lesion |
|---|---|
| I | Partial avulsion of superior one third |
| II | Complete avulsion of superior one third |
| III | Complete avulsion of superior two thirds |
| IV | Complete avulsion of the tendon with head centered and fatty degeneration classified as less than or equal to type III |
| V | Complete avulsion of the tendon but uncentered head with coracoid impingement and fatty degeneration classified as greater than or equal to type III |
In a type IV rupture (Fig. 21-30) a 360-degree release (Figs. 21-31 to 21-34) is the most crucial step in the repair (Figs. 21-35 and 21-36). Great caution is necessary to preserve not only the subscapular nerves but also all of the brachial plexus with its collateral and terminal branches.
We consider type V ruptures not repairable. A partial repair with a pectoralis minor transfer (see later) for augmentation can be performed arthroscopically if a repair is indicated, depending on such other factors as patient’s age or presence and stage of ruptures of other rotator cuff tendons.
Lesser Tuberosity Fracture
Isolated avulsion fractures of the lesser tuberosity (Figs. 21-37 and 21-38) are rare injuries. The underlying trauma mechanism is usually an abduction–external rotation trauma. In elderly and middle-aged patients, it is more likely for the tendon to rupture, but in a younger patient, the high tensile force of the tendon can lead to an avulsion fracture. If the fracture includes the superior third of the lesser tuberosity, it will inevitably affect the stability of the long head of the biceps. This can be due to two reasons: damage of the bony medial wall of the bicipital groove and soft tissue lesions of the reflection pulley system.
As an equivalent to a type IV subscapularis tendon tear, a lesser tuberosity fracture potentially will result in a total insufficiency of the subscapularis muscle and merges with a repair before irreversible retraction. The most common treatment for displaced avulsion fractures of the lesser tuberosity is open reduction and internal fixation using cannulated screws. The arthroscopic technique described in the literature21 uses a posterior intra-articular portal for visualization.
We use an arthroscopic technique with alternating visualization from the posterior intra-articular A portal and subacromial anterolateral D portal. This allows a combined view and intra-articular and extra-articular access to the entire anterior shoulder including the area medial to the coracoid and conjoint tendon. This has marked advantages for the release and mobilization of the subscapular musculotendinous unit and of the surrounding neurovascular structures. The reduction and stabilization can be performed by traction sutures and by suture anchors only. In case of larger fractures, an arthroscopically guided cannulated screw fixation is an attractive effective option. Good reduction and firm fixation can be achieved arthroscopically (Figs. 21-39 and 21-40). If associated biceps instability is present, the arthroscopic osteosynthesis is always performed in conjunction with biceps tenodesis.
Pectoralis Minor Transfer
Due to atrophy, fatty degeneration, or denervation of the subscapularis muscle, a chronic subscapularis tendon tear is not always totally reparable. These complete tears of the subscapularis tendon with retraction can lead to an eccentric humeral head that is displaced anteriorly and superiorly on the glenoid because of disruption of the force couple of the rotator cuff. The humeral head comes in contact with the coracoid, which makes it a type V subscapularis tendon lesion in our proposed classification (Fig. 21-41). Deterioration of shoulder function, instability, and degenerative articular changes can be present or can develop. Some type IV lesions can be augmented after repair by this transfer.
The recommended treatment options range from débridement to reversed shoulder arthroplasty. In young patients with higher functional demand, open pectoralis major transfer is an acknowledged option.22 Open pectoralis minor transfer with or without transfer of the pectoralis major has also been described but is not normally used.23
Traction sutures are passed in a lasso-loop technique at the superior and inferior border of the distal pectoralis minor tendon before it is detached from the coracoid (Figs. 21-42 and 21-43). First the traction sutures (Fig. 21-44) and then the pectoralis minor tendon are shuttled underneath the coracoid and posterior to the conjoint tendon (Fig. 21-45). In the next step, these same sutures are used to arm suture anchors for fixation to the lesser tuberosity. A coracoplasty is performed in all cases to prevent development of lateral thoracic outlet syndrome behind the conjoint tendon. This is an important issue because the combined bulk of the muscles can cause a compression of the brachial plexus or a subcoracoid impingement.
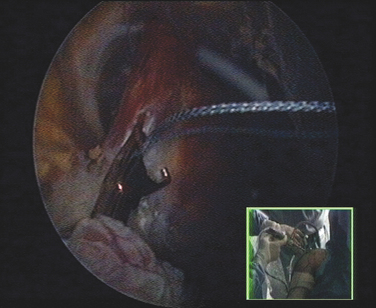
FIGURE 21-44 Shuttling of the pectoralis minor traction sutures underneath the coracoid process. Inset, External view.
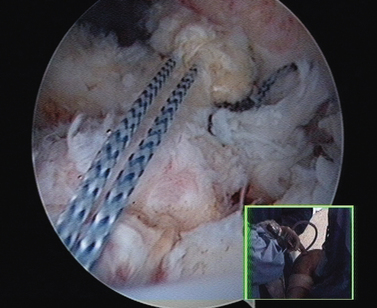
FIGURE 21-45 Shuttling of the pectoralis minor over the subscapularis partial repair. Inset, External view.
Depending on where the musculocutaneous nerve passes the conjoint tendon, significant tension might be placed on this nerve and need to be dealt with by a release. This technique allows a nonanatomic reconstruction by transferring the pectoralis minor as augmentation of the partially repaired subscapularis (Figs. 21-46 and 21-47). There are two distinct biomechanical consequences: The transferred tendon is reoriented to approach the force vector of the subscapularis muscle, and a passive restraint for anterior humeral excursion is restored.
Coracoid Fracture
Coracoid fractures are rare. Most coracoid fractures are seen after severe trauma and are associated with other lesions of the shoulder, such as glenohumeral or acromioclavicular dislocation, intra- and extra-articular scapula fractures, or subscapularis tendon tears. Treatment of those coracoid fractures is part of the reconstruction of the shoulder. Fractures of the lateral coracoid are often minimally displaced due to stabilizing insertions of the acromioclavicular ligament, conjoint tendon, and pectoralis minor.
In our experience, the coracoid, as a key landmark, is perfectly accessible to arthroscopic exploration of the anterior area. Stabilization of these rare fractures (Figs. 21-48 to 21-50) can therefore be performed arthroscopically. For osteosynthesis, depending on the fracture type, suture anchors or cannulated screws may be used (Figs. 21-51 to 21-53). The associated lesions may be treated during the same procedure. Other advantages of arthroscopic treatment are the good visual control in case of associated articular glenoid fractures and easy access to the suprascapular notch in case of suprascapular nerve entrapment in certain fracture patterns.24,25
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree