Acetabular Revision
Rings, Cages, and Custom Implants
Derek R. Johnson, Douglas A. Dennis and Raymond H. Kim
Key Points
Introduction
The goal of acetabular revision surgery is to achieve a stable, pain-free, and functional construct. This can frequently be achieved with the use of larger hemispherical acetabular components. However, in the setting of massive periacetabular bone loss, stable fixation with the use of a standard hemispherical acetabular component may not be possible, and other reconstruction options must be considered.
Numerous treatment methods are available for management of massive acetabular defects in revision total hip arthroplasty (THA). Unfortunately, the clinical results of managing these complex cases are highly variable and are often associated with a high incidence of complications. Treatment methods utilized in an attempt to reconstruct these difficult cases have included implantation of jumbo acetabular components1 with or without the use of massive structural allografts,2–5 bipolar hemiarthroplasty,6 acetabular impaction bone grafting,7 oblong acetabular components,8–10 noncustom acetabular reconstruction rings or cages,11–14 and use of custom triflanged acetabular components (CTACs).15–19 This chapter will focus on the treatment of massive periacetabular bone loss in revision THA with use of noncustom rings and cages and custom triflanged acetabular components.
Bone Loss Classification
The severity of acetabular bone loss in revision total hip arthroplasty (THA) has been classified by Paprosky5 (Table 92-1) and the American Academy of Orthopedic Surgeons (AAOS; Table 92-2).20 The bone loss is classified according to the magnitude of bone loss, the amount of acetabular component migration, and the degree of destruction of the anterior and posterior columns.
Table 92-1
Paprosky Classification of Acetabular Defects
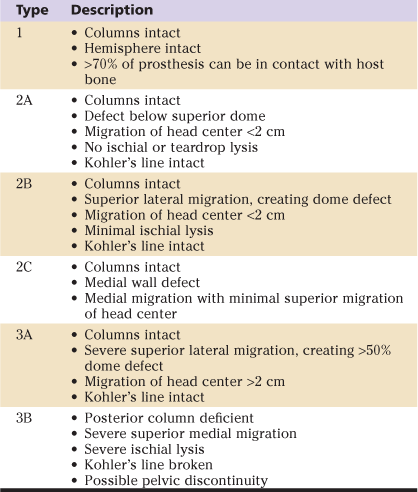
Table 92-2
AAOS Classification of Acetabular Defects

AAOS, American Academy of Orthopaedic Surgeons.
Indications
The presence of massive periacetabular bone loss (Paprosky 3B and AAOS III and IV) that precludes the ability to obtain a stable acetabular reconstruction with a traditional hemispherical cup is the primary indication for the use of acetabular cages or a custom triflanged acetabular component (CTAC). The use of bulk allograft and impaction grafting with traditional acetabular components in these cases has failure rates as high as 36% in some series.2-4,7 The ability of these allografts to incorporate and withstand physiologic loads is questionable and is a likely cause of the unacceptably high failure rates. With the use of cages or a CTAC, however, the ability to bridge the defects and obtain fixation to remaining available host bone is theoretically attained, providing stable fixation and preventing early component migration. An additional advantage of the use of acetabular cages or a CTAC is the ability to place the acetabular component at a correct anatomic level to restore hip biomechanics and stability more accurately.
Contraindications
The outstanding results and proven long-term success of hemispherical components in revision THA make these devices the components of choice in the presence of adequate host bone. The cost and complexity of cages and a CTAC limit their indication only to those cases in which adequate host bone for hemispherical cups is not available. In cases in which adequate pelvic bone quality to obtain screw fixation does not exist, or in the setting of persistent infection, cages and CTAC are contraindicated. Finally, use of these devices alone without additional efforts at pelvic stabilization (column plating) is relatively contraindicated in situations with massive bone loss associated with the presence of a pelvic discontinuity.
Preoperative Planning
History and Physical Examination
As with any arthroplasty evaluation, the initial step in management is a thorough history and physical examination. History should include the reason for initial arthroplasty, as well as the reason for any subsequent revision arthroplasty procedures. A detailed investigation for infection is essential, including white blood cell count, erythrocyte sedimentation rate, and C-reactive protein. If these tests are elevated, or if the history indicates infection (previous infection or wound complication, early failure of previous surgery, or constitutional symptoms), hip aspiration and joint fluid analysis are necessary. Previous operative records should be reviewed to note the exposure used and any complications or unusual techniques involved, and to determine the type, size, and fixation method of the currently implanted components. Careful examination should be performed, including inspection of the skin for the presence of fluctuance, warmth, and erythema and for the location and condition of previous skin incisions. The neurovascular status of the limb must be assessed, as well as the symmetry of leg lengths. Also, the motor function of the lower extremities must be evaluated, particularly the function of the abductor musculature, as this can be predictive of future postoperative problems with hip stability.
Radiographic Evaluation
Plain radiographs, including an anteroposterior view of the pelvis and anteroposterior and lateral views of the hip along with the entire prosthesis, should be obtained. If a femoral component revision is planned, anteroposterior and lateral views of the femur undergoing instrumentation are necessary. Iliac and obturator oblique views are helpful in classifying bone loss and in determining whether a pelvic discontinuity is present. Computed tomography (CT) with metal subtraction techniques allows the surgeon to evaluate osseous defects more precisely, including the presence of a pelvic discontinuity, and is essential for the design of a CTAC should this treatment method be selected (Fig. 92-1A and B). Specific CT scan protocols are often required for the production of a CTAC and should be ordered as such to avoid subjecting the patient to multiple CT scans. In cases with significant leg length discrepancy, full-length orthoroentgenograms may be necessary for preoperative leg length assessment.
Figure 92-1 A, Anteroposterior radiograph of a failed total hip arthroplasty (THA) with acetabular component loosening and massive periacetabular bone loss. B, Three-dimensional images created from a thin cut computed tomography (CT) scan, demonstrating severe acetabular protrusio and a Paprosky 3B acetabular defect.
In scenarios of severe acetabular component protrusio, a CT scan with a concomitant angiogram may be required.21 This would aid in determining whether intrapelvic vessels or other visceral structures are in close proximity to the failed acetabular component. Anticipation of potential vascular injury would warrant a preoperative vascular surgery consultation and consideration of a retroperitoneal exposure to free vital intrapelvic structures from the acetabular component before it is removed.
Component Design
Numerous designs of noncustom acetabular rings and antiprotrusio cages are available and selected when stable fixation on remaining host bone is not possible (Fig. 92-2). These devices gain stability through fixation to the ischium and ilium, bridge areas of acetabular bone loss, provide support for the acetabular component, and allow for pelvic bone grafting in an environment protected from excessive stress. They are typically designed with some degree of flexibility, allowing the surgeon to manipulate the shape of the cage somewhat intraoperatively to maximize fixation and fit. Polyethylene acetabular components are then cemented into the ring or cage.

Figure 92-2 Photographs of a Mueller acetabular ring (A), a Ganz roof ring (B), and an antiprotrusio cage (C) utilized for reconstruction of large acetabular bone defects.
The design of a CTAC is finalized preoperatively rather than intraoperatively as with cages. After completion of a thin cut CT with metal subtraction, the manufacturer creates three dimensional (3D) images, as well as a one-to-one computer-aided design (CAD) solid model of the hemipelvis. This 3D model allows for accurate assessment of bone loss and remaining pelvic bone and is utilized to facilitate the design of the CTAC. The surgeon, in conjunction with the design engineer, uses the 3D model to create a clay or acrylic prototype of the CTAC. The surgeon is then able to manipulate the prototype within the 3D hemipelvis and make recommendations to the design engineer regarding cup orientation, the hip center, and the number and position of flange fixation screws. In addition to their use in preoperative planning, these models may be sterilized and used intraoperatively as a trial component to assess position and fit (Fig. 92-3A through G).
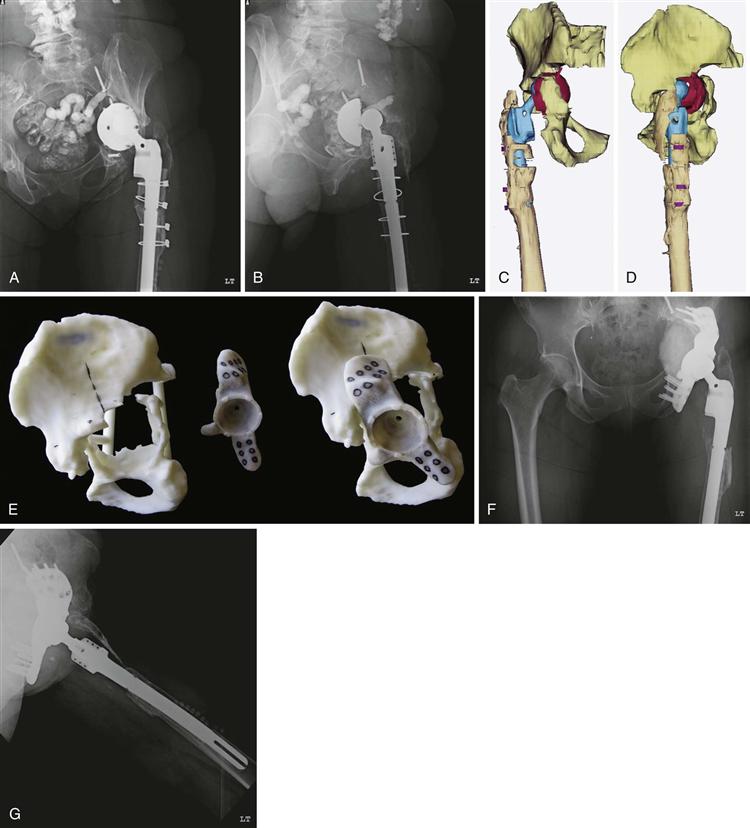
Figure 92-3 Preoperative anteroposterior (A) and lateral (B) radiographs demonstrating a failed total hip arthroplasty (THA) with acetabular component migration and massive periacetabular bone loss. Three-dimensional anteroposterior (C) and lateral (D) images demonstrating severe acetabula protrusio and a Paprosky 3B acetabular defect. E, Photographs of a one-to-one computer-aided design (CAD) model of a hemipelvis, demonstrating marked periacetabular bone loss, along with the acrylic custom triflanged acetabular component (CTAC) designed for acetabular reconstruction. Postoperative anteroposterior (F) and lateral (G) radiographs following reconstruction with a CTAC.
Patient-specific considerations, including leg length discrepancy, planned retention or revision of the femoral component, length of the contralateral leg, and size of the current acetabular component, determine the position of the hip center. The vertical head center location can be established by first determining the approximate anatomic position of the head center using the superior aspect of the obturator foramen as a reference point. The remaining bone of the anterior and posterior columns determines the head center in the coronal plane, whereas flange geometry and component face diameter guide the position of the head center in the sagittal plane. Evaluation of the contralateral hip center, if not distorted by aberrant anatomy or previous joint arthroplasty, can be helpful in determination of the true anatomic hip center. Although it may be desirable to reconstruct the hip center anatomically from a hip biomechanics standpoint, this may not be possible in all cases, particularly if the hip center has been displaced superiorly for an extended period of time and the femoral component is to be maintained. In this situation, a compromise may be needed with some superior positioning of the vertical hip center to reduce the newly reconstructed hip without the risk of neurovascular injury. Obtaining a preoperative anteroposterior radiograph with traction applied to the operative limb may be helpful in assessing laxity of the hip and in subsequently determining where the hip center of the CTAC should be placed.
Establishing anteversion and abduction angles of the cup is necessary to set face orientation. The abduction angle is set 35 to 40 degrees from the horizontal plane, using the plane of the obturator foramen as a reference. The anteversion angle is set at 25 to 30 degrees, using the plane of the iliac wing and the obturator foramen as references.
Upon finalization of implant design, the surfaces of the titanium alloy stock are milled. The iliac and ischial flanges contain multiple rows of screw holes for 6.5-mm screws. Current designs allow the inclusion of threaded holes to accommodate locking screws into some, or all, of the holes. Four to six screw holes in the ischial flange are preferred because they have proved to be the most common site of fixation loss. Two rows of three to four screw holes have proved sufficient for fixation of the iliac flange. The pubic flange is smaller and does not contain screw holes. Dome screw holes may also be placed, depending on the adequacy of the iliac bone stock, to create interlocking-screw fixation with the iliac flange screws. The inner geometry has a modular locking mechanism that can accept any of the modular polyethylene or metal liners typically available for standard acetabular components.
The bone interface of the CTAC, including the flanges, has a porous ingrowth surface to foster osteointegration and may be enhanced by the addition of a hydroxyapatite coating. Although osseous integration has not been proven through histologic retrieval analyses with the use of a CTAC, the ingrowth surface does provide a theoretical fixation advantage not afforded with the use of cages. We suspect that some magnitude of bone ingrowth occurs based on the lack of component migration observed radiographically in most cases now followed out to 10 years. Current CTAC designs allow for easier insertion at the time of operation and provide space behind the implant for additional bone graft. A critical design characteristic involves creating a central dome that has intimate contact with the remaining ilium superiorly to reduce shear stresses on the three fixation flanges. In a hip with massive acetabular bone loss or even pelvic discontinuity, this iliac shelf may be the only structurally sound bone on which the CTAC may be able to rely.
Throughout the development process of the CTAC, multiple iterations of the prototype are often required, with extensive communication between the surgeon and the design engineer. This is necessary for accurate component preparation, which is critical because the operation is dependent upon the availability of a single component at the time of the operation. Because of the need for careful planning and production of the custom implant, the patient should be made aware that the preoperative process may take up to 2 months to complete. In addition to the extensive planning necessary for using CTAC, the implant is significantly more expensive than cages and “off-the-shelf” implants. For this reason, many hospitals may require prior authorization for its use or may preclude its use altogether.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








