The correct embryonic development of the musculoskeletal system requires a coordinated morphogenesis of the fundamental tissues of the body. Throughout the human body, there are four major types of tissues: CT proper has a loose, flexible matrix, called ground substance. The most common cell within CT proper is the fibroblast. Fibroblasts produce collagen, elastin, and reticular fibers: The various characteristics of collagen differ depending on whether it is loose or dense collagen. The anatomic and functional characteristics of loose and dense collagen are summarized in Table 1-1. Collagenous and elastic fibers are sparse and irregularly arranged in loose CT but are tightly packed in dense CT.10 TABLE 1-1 Loose and Dense Collagen Joint Type Anatomic Location Fiber Orientation Mechanical Specialization Dense irregular connective tissue Composes the external fibrous layer of the joint capsule, forms ligaments, bone, aponeuroses, and tendons Parallel, tightly aligned fibers Ligament: binds bones together and restrains unwanted movement at the joints; resists tension in several directions Tendon: attaches muscle to bone Loose irregular connective tissue Found in capsules, muscles, nerves, fascia, and skin Random fiber orientation Provides structural support The various types of CT, as they relate to the musculoskeletal system, are described as follows:
CHAPTER 1
The Musculoskeletal System
OVERVIEW
 Epithelial. Covers all internal and external body surfaces and includes structures such as the skin and the inner lining of the blood vessels.
Epithelial. Covers all internal and external body surfaces and includes structures such as the skin and the inner lining of the blood vessels.
 Connective. Connective tissue (CT), which includes four different classes: connective tissue proper, bone, cartilage, and blood tissue. In the embryo, muscle tissue and its fascia form as a differentiation of the paraxial mesoderm that divides into somites on either side of the neural tube and notochord. The cartilage and bone of the vertebral column and ribs develop from the sclerotome which is the anterior (ventral) part of the somite.1 The dermomyotome, which is the posterior (dorsal) part of the somite, gives rise to the overlying dermis of the back and the skeletal muscles of the body and limbs.1 Connective tissue provides structural and metabolic support for other tissues and organs of the body.
Connective. Connective tissue (CT), which includes four different classes: connective tissue proper, bone, cartilage, and blood tissue. In the embryo, muscle tissue and its fascia form as a differentiation of the paraxial mesoderm that divides into somites on either side of the neural tube and notochord. The cartilage and bone of the vertebral column and ribs develop from the sclerotome which is the anterior (ventral) part of the somite.1 The dermomyotome, which is the posterior (dorsal) part of the somite, gives rise to the overlying dermis of the back and the skeletal muscles of the body and limbs.1 Connective tissue provides structural and metabolic support for other tissues and organs of the body.
 Muscle. Muscles are classified functionally as either voluntary or involuntary, and structurally as either smooth, striated (skeletal), or cardiac. There are approximately 430 skeletal muscles in the body, each of which can be considered anatomically as a separate organ. Of these 430 muscles, about 75 pairs provide the majority of body movements and postures.2
Muscle. Muscles are classified functionally as either voluntary or involuntary, and structurally as either smooth, striated (skeletal), or cardiac. There are approximately 430 skeletal muscles in the body, each of which can be considered anatomically as a separate organ. Of these 430 muscles, about 75 pairs provide the majority of body movements and postures.2
 Nervous. Nervous tissue provides a two-way communication system between the central nervous system (brain and spinal cord) and muscles, sensory organs, and various systems (see Chapter 3).
Nervous. Nervous tissue provides a two-way communication system between the central nervous system (brain and spinal cord) and muscles, sensory organs, and various systems (see Chapter 3).
CONNECTIVE TISSUE
 Collagen is a group of naturally occurring proteins. The collagens are a family of extracellular matrix (ECM) proteins that play a dominant role in maintaining the structural integrity of various tissues and in providing tensile strength to tissues. The ECM is formed from glycosaminoglycans (GAGs) subunits, long polysaccharide chains containing amino sugars, and are strongly hydrophilic to allow rapid diffusion of water-soluble molecules and easy migration of cells. Proteoglycans, which are a major component of the ECM, are macromolecules that consist of a protein backbone to which the GAGs are attached. There are two types of GAGs: chondroitin sulfate and keratin sulfate.3,4 A simple way to visualize the proteoglycan molecule is to consider a test tube brush, with the stem representing the protein core and the GAGs representing the bristles.5,6 Glycoproteins, another component of the ECM, consist of fibronectin and thrombospondin and function as adhesive structures for repair and regeneration.7
Collagen is a group of naturally occurring proteins. The collagens are a family of extracellular matrix (ECM) proteins that play a dominant role in maintaining the structural integrity of various tissues and in providing tensile strength to tissues. The ECM is formed from glycosaminoglycans (GAGs) subunits, long polysaccharide chains containing amino sugars, and are strongly hydrophilic to allow rapid diffusion of water-soluble molecules and easy migration of cells. Proteoglycans, which are a major component of the ECM, are macromolecules that consist of a protein backbone to which the GAGs are attached. There are two types of GAGs: chondroitin sulfate and keratin sulfate.3,4 A simple way to visualize the proteoglycan molecule is to consider a test tube brush, with the stem representing the protein core and the GAGs representing the bristles.5,6 Glycoproteins, another component of the ECM, consist of fibronectin and thrombospondin and function as adhesive structures for repair and regeneration.7
 Elastic fibers are composed of a protein called elastin. As its name suggests, elastin provides elastic properties to the tissues in which it is situated.8 Elastin fibers can stretch, but they normally return to their original shape when the tension is released. Thus, the elastic fibers of elastin determine the patterns of distention and recoil in most organs, including the skin and lungs, blood vessels, and CT. Bundles of collagen and elastin combine to form a matrix of CT fascicles. This matrix is organized within the primary collagen bundles as well as between the bundles that surround them.9
Elastic fibers are composed of a protein called elastin. As its name suggests, elastin provides elastic properties to the tissues in which it is situated.8 Elastin fibers can stretch, but they normally return to their original shape when the tension is released. Thus, the elastic fibers of elastin determine the patterns of distention and recoil in most organs, including the skin and lungs, blood vessels, and CT. Bundles of collagen and elastin combine to form a matrix of CT fascicles. This matrix is organized within the primary collagen bundles as well as between the bundles that surround them.9
 Reticular fibers are composed of a type of collagen, which is secreted by reticular cells. These fibers crosslink to form a fine meshwork, called reticulin, which acts as a supporting mesh in bone marrow, and the tissues and organs of the lymphatic system, and the liver.
Reticular fibers are composed of a type of collagen, which is secreted by reticular cells. These fibers crosslink to form a fine meshwork, called reticulin, which acts as a supporting mesh in bone marrow, and the tissues and organs of the lymphatic system, and the liver.
Fascia
Fascia, for example, the thoracolumbar fascia and the plantar fascia, is viewed as a loose CT that provides support and protection to a joint, and acts as an interconnection between tendons, aponeuroses, ligaments, capsules, nerves, and the intrinsic components of muscle.11,12 Fascia may be categorized as fibrous or nonfibrous, with the fibrous components consisting mainly of collagen and elastin fibers, and the nonfibrous portion consisting of amorphous ground substance.13 Three different types of fascia have been identified, namely, superficial, deep, and visceral fascia. Various three-dimensional biomechanical models of the human fascial system have been developed, which correlate dysfunctional movement with various interrelated abnormal amounts of tension throughout the network of fascia. In particular, deep fascia has been implicated in being involved with the deep venous return, in having a possible role in proprioception, and responding to mechanical traction induced by muscular activity in different regions.14 Histological studies of deep fascia in the limbs show that it consists of elastic fibers and undulated collagen fibers arranged in layers.15 Each collagen layer is aligned in a different direction, and this permits a certain degree of stretch as well as a capacity to recoil.16
Tendons
Tendons are dense, regularly arranged connective tissues, composed of 70% water and 30% dry mass that attach muscle to the bone at each end of the muscle. Tendons produce joint motion by transferring force from muscle to bone, and, when stretched, store elastic energy that contributes to movement. Also, tendons enable the muscle belly to be an optimal distance from the joint upon which it is acting. The collagen fibers of tendons (70–80% of the collagen in tendons is type I, with the remaining 20–30% of dry weight composed of proteoglycans, GAGs, elastin, and other collagens—being type III, V, and VII) are arranged in a quarter-stagger arrangement, which gives it a characteristic banding pattern and provides high strength and stability.17 Tenoblasts, or immature tendon cells, transform into tenocytes that synthesize collagen and components of the ECM network.7 The ECM surrounds collagen and tenocytes and is composed of several components for specific functions (e.g., glycoproteins, and Tenascin-C, which may play a role in collagen fiber orientation and alignment). Tendon structure is highly regular with collagen-forming triple helices (tropocollagen), which pack together to form microfibrils, which interdigitate to form fibrils, which coalesce to form fibers, which combine to form fascicles, which are bundled together to form a tendon.18 The thickness of each tendon varies and is proportional to the size of the muscle from which it originates. Vascularity within the tendon is relatively sparse and corresponds with the lower metabolic/turnover rate of these tissues. Within the fascicles of tendons, which are held together by loose CT called endotenon, the collagen components are oriented in a unidirectional way. Endotenon contains blood vessels, lymphatics, and nerves and permits longitudinal movements of individual fascicles when tensile forces are applied to the structure. The CT surrounding groups of fascicles, or the entire structure, is called the epitenon. The epitenon contains the vascular, lymphatic, and nerve supplies to the tendon. A peritendinous sheath (paratenon), which is composed of loose areolar connective tissue in addition to sensory and autonomic nerve fibers, surrounds the entire tendon.19 This sheath consists of two layers: an inner (visceral) layer and an outer (parietal) layer with occasional connecting bridges (mesotenon). If there is synovial fluid between these two layers, the paratenon is called tenosynovium; if not, it is termed tenovagium.9
Tendons are metabolically active and are provided with a rich and vascular supply during development.20 Tendons receive their vascular supply through the musculotendinous junction (MTJ), the osteotendinous junction, and the vessels from the various surrounding tissues including the paratenon and mesotenon.18 Tendons in different areas of the body receive different amounts of blood supply, and tendon vascularity can be compromised by the junctional zones and sites of friction, torsion, or compression—a number of tendons are known to have reduced tendon vascularity, including the supraspinatus, the biceps, the Achilles, the patellar, and the posterior tibial tendon.18
The mechanical properties of tendon come from its highly oriented structure. Tendons display viscoelastic mechanical properties that confer time- and rate-dependent effects on the tissue. Specifically, tendons are more elastic at lower strain rates and stiffer at higher rates of tensile loading (see Chapter 2). Tendons deform less than ligaments under an applied load and are able to transmit the load from muscle to bone.9 Material and structural properties of the tendon increase from birth through maturity and then decrease from maturity through old age.18 Although tendons withstand strong tensile forces well, they resist shear forces less well and provide little resistance to a compression force (see Chapter 2).
A tendon can be divided into three main sections:21
 The bone–tendon junction. At most tendon–bone interfaces, the collagen fibers insert directly into the bone in a gradual transition of material composition. The physical junction of tendon and bone is referred to as an enthesis,22 and is an interface that is vulnerable to acute and chronic injury.23 One role of the enthesis is to absorb and distribute the stress concentration that occurs at the junction over a broader area.
The bone–tendon junction. At most tendon–bone interfaces, the collagen fibers insert directly into the bone in a gradual transition of material composition. The physical junction of tendon and bone is referred to as an enthesis,22 and is an interface that is vulnerable to acute and chronic injury.23 One role of the enthesis is to absorb and distribute the stress concentration that occurs at the junction over a broader area.
 The tendon midsubstance. Overuse tendon injuries can occur in the midsubstance of the tendon, but not as frequently as at the enthesis.
The tendon midsubstance. Overuse tendon injuries can occur in the midsubstance of the tendon, but not as frequently as at the enthesis.
 MTJ. The MTJ is the site where the muscle and tendon meet. The MTJ comprises numerous interdigitations between muscle cells and tendon tissue, resembling interlocked fingers. Despite its viscoelastic mechanical characteristics, the MTJ is very vulnerable to tensile failure (see Chapter 2).24,25
MTJ. The MTJ is the site where the muscle and tendon meet. The MTJ comprises numerous interdigitations between muscle cells and tendon tissue, resembling interlocked fingers. Despite its viscoelastic mechanical characteristics, the MTJ is very vulnerable to tensile failure (see Chapter 2).24,25
Ligaments
Skeletal ligaments are fibrous bands of dense CT that connect bones across joints. Ligaments can be named for the bones into which they insert (coracohumeral), their shape (deltoid of the ankle), or their relationships to each other (cruciate).26 The gross structure of a ligament varies according to location (intra-articular or extra-articular, capsular), and function.27 Ligaments, which appear as dense white bands or cords of CT, are composed primarily of water (approximately 66%), and of collagen (largely type I collagen [85%], but with small amounts of type III) making up most of the dry weight. The collagen in ligaments has a less unidirectional organization than it does in tendons, but its structural framework still provides stiffness (resistance to deformation—see Chapter 2).28 Small amounts of elastin (1% of the dry weight) are present in ligaments, with the exception of the ligamentum flavum and the nuchal ligament of the spine, which contain more. The cellular organization of ligaments makes them ideal for sustaining tensile loads, with many containing functional subunits that are capable of tightening or loosening in different joint positions.29 At the microscopic level, closely spaced collagen fibers (fascicles) are aligned along the long axis of the ligament and are arranged into a series of bundles that are delineated by a cellular layer, the endoligament, and the entire ligament is encased in a neurovascular biocellular layer referred to as the epiligament.26 Ligaments contribute to the stability of joint function by preventing excessive motion,30 acting as guides or checkreins to direct motion, and providing proprioceptive information for joint function through sensory nerve endings (see Chapter 3) and the attachments of the ligament to the joint capsule.31–33 Many ligaments share functions. For example, while the anterior cruciate ligament of the knee is considered the primary restraint to anterior translation of the tibia relative to the femur, the collateral ligaments and the posterior capsule of the knee also help in this function (see Chapter 20).26 The vascular and nerve distribution to ligaments is not homogeneous. For example, the middle of the ligament is typically avascular, while the proximal and distal ends enjoy a rich blood supply. Similarly, the insertional ends of the ligaments are more highly innervated than the midsubstance.
Cartilage
Cartilage tissue exists in three forms: hyaline, elastic, and fibrocartilage.
 Hyaline cartilage, also referred to as articular cartilage, covers the ends of long bones and permits almost frictionless motion to occur between the articular surfaces of a diarthrodial (synovial) joint.34 Articular cartilage is a highly organized viscoelastic material composed of cartilage cells called chondrocytes, water, and an ECM.
Hyaline cartilage, also referred to as articular cartilage, covers the ends of long bones and permits almost frictionless motion to occur between the articular surfaces of a diarthrodial (synovial) joint.34 Articular cartilage is a highly organized viscoelastic material composed of cartilage cells called chondrocytes, water, and an ECM.
Articular cartilage, the most abundant cartilage within the body, is devoid of any blood vessels, lymphatics, and nerves.5,6 Most of the bones of the body form first as hyaline cartilage, and later become bone in a process called endochondral ossification. The normal thickness of articular cartilage is determined by the contact pressures across the joint—the higher the peak pressures, the thicker the cartilage.27 Articular cartilage functions to distribute the joint forces over a large contact area, thereby dissipating the forces associated with the load. This distribution of forces allows the articular cartilage to remain healthy and fully functional throughout decades of life. The patellar has the thickest articular cartilage in the body.
Articular cartilage may be grossly subdivided into four distinct zones with differing cellular morphology, biomechanical composition, collagen orientation, and structural properties, as follows:
 The superficial zone. The superficial zone, which lies adjacent to the joint cavity, comprises approximately 10–20% of the articular cartilage thickness and functions to protect deeper layers from shear stresses. The collagen fibers within this zone are packed tightly and aligned parallel to the articular surface. This zone is in contact with the synovial fluid and handles most of the tensile properties of cartilage.
The superficial zone. The superficial zone, which lies adjacent to the joint cavity, comprises approximately 10–20% of the articular cartilage thickness and functions to protect deeper layers from shear stresses. The collagen fibers within this zone are packed tightly and aligned parallel to the articular surface. This zone is in contact with the synovial fluid and handles most of the tensile properties of cartilage.
 The middle (transitional) zone. In the middle zone, which provides an anatomic and functional bridge between the superficial and deep zones, the collagen fibril orientation is obliquely organized. This zone comprises 40–60% of the total cartilage volume. Functionally, the middle zone is the first line of resistance to compressive forces.
The middle (transitional) zone. In the middle zone, which provides an anatomic and functional bridge between the superficial and deep zones, the collagen fibril orientation is obliquely organized. This zone comprises 40–60% of the total cartilage volume. Functionally, the middle zone is the first line of resistance to compressive forces.
 The deep or radial layer. The deep layer comprises 30% of the matrix volume. It is characterized by radially aligned collagen fibers that are perpendicular to the surface of the joint, and which have a high proteoglycan content. Functionally the deep zone is responsible for providing the greatest resistance to compressive forces.
The deep or radial layer. The deep layer comprises 30% of the matrix volume. It is characterized by radially aligned collagen fibers that are perpendicular to the surface of the joint, and which have a high proteoglycan content. Functionally the deep zone is responsible for providing the greatest resistance to compressive forces.
 The tidemark. The tidemark distinguishes the deep zone from the calcified cartilage, the area that prevents the diffusion of nutrients from the bone tissue into the cartilage.
The tidemark. The tidemark distinguishes the deep zone from the calcified cartilage, the area that prevents the diffusion of nutrients from the bone tissue into the cartilage.
 Elastic (yellow) cartilage is a very specialized CT, primarily found in locations such as the outer ear, and portions of the larynx.
Elastic (yellow) cartilage is a very specialized CT, primarily found in locations such as the outer ear, and portions of the larynx.
 Fibrocartilage, also referred to as white cartilage, functions as a shock absorber in both weight-bearing and nonweight-bearing joints. Its large fiber content, reinforced with numerous collagen fibers, makes it ideal for bearing large stresses in all directions. Fibrocartilage is an avascular, alymphatic, and aneural tissue and derives its nutrition by a double-diffusion system.36 Examples of fibrocartilage include the symphysis pubis, the intervertebral disk, and the menisci of the knee.
Fibrocartilage, also referred to as white cartilage, functions as a shock absorber in both weight-bearing and nonweight-bearing joints. Its large fiber content, reinforced with numerous collagen fibers, makes it ideal for bearing large stresses in all directions. Fibrocartilage is an avascular, alymphatic, and aneural tissue and derives its nutrition by a double-diffusion system.36 Examples of fibrocartilage include the symphysis pubis, the intervertebral disk, and the menisci of the knee.
Bone
Bone is a highly vascular form of CT, composed of collagen, calcium phosphate, water, amorphous proteins, and cells. It is the most rigid of the CTs (Table 1-2). Despite its rigidity, bone is a dynamic tissue that undergoes constant metabolism and remodeling. The collagen of bone is produced in the same manner as that of ligament and tendon but by a different cell, the osteoblast.10 At the gross anatomical level, each bone has a distinct morphology comprising both cortical bone and cancellous bone. Cortical bone is found in the outer shell. Cancellous bone is found within the epiphyseal and metaphyseal regions of long bones, as well as throughout the interior of short bones.24 Skeletal development occurs in one of the two ways:
TABLE 1-2 | General Structure of Bone | ||
Site | Comment | Conditions | Result |
Epiphysis | Mainly develops under pressure Apophysis forms under traction Forms bone ends Supports articular surface | Epiphyseal dysplasias Joint surface trauma Overuse injury Damaged blood supply | Distorted joints Degenerative changes Fragmented development Avascular necrosis |
Physis | Epiphyseal or growth plate Responsive to growth and sex hormones Vulnerable prior to growth spurt Mechanically weak | Physeal dysplasia Trauma Slipped epiphysis | Short stature Deformed or angulated growth or growth arrest |
Metaphysis | Remodeling expanded bone end Cancellous bone heals rapidly Vulnerable to osteomyelitis Affords ligament attachment | Osteomyelitis Tumors Metaphyseal dysplasia | Sequestrum formation Altered bone shape Distorted growth |
Diaphysis | Forms shaft of bone Large surface for muscle origin Significant compact cortical bone Strong in compression | Fractures Diaphyseal dysplasias Healing slower than at metaphysis | Able to remodel angulation Cannot remodel rotation Involucrum with infection Dysplasia gives altered density and shape |
Data from Reid DC. Sports Injury Assessment and Rehabilitation. New York, NY: Churchill Livingstone; 1992. | |||
 Intramembranous ossification. Mesenchymal stem cells within mesenchyme or the medullary cavity of a bone initiate the process of intramembranous ossification. This type of ossification occurs in the cranium and facial bones and, in part, the ribs, clavicle, and mandible.
Intramembranous ossification. Mesenchymal stem cells within mesenchyme or the medullary cavity of a bone initiate the process of intramembranous ossification. This type of ossification occurs in the cranium and facial bones and, in part, the ribs, clavicle, and mandible.
 Endochondral ossification. The first site of ossification occurs in the primary center of ossification, which is in the middle of the diaphysis (shaft). About the time of birth, a secondary ossification center appears in each epiphysis (end) of long bones. Between the bone formed by the primary and secondary ossification centers, cartilage persists as the epiphyseal (growth) plates between the diaphysis and the epiphysis of a long bone. This type of ossification occurs in the appendicular and axial bones.
Endochondral ossification. The first site of ossification occurs in the primary center of ossification, which is in the middle of the diaphysis (shaft). About the time of birth, a secondary ossification center appears in each epiphysis (end) of long bones. Between the bone formed by the primary and secondary ossification centers, cartilage persists as the epiphyseal (growth) plates between the diaphysis and the epiphysis of a long bone. This type of ossification occurs in the appendicular and axial bones.
The periosteum is formed when the perichondrium, which surrounds the cartilage, becomes the periosteum. Chondrocytes in the primary center of ossification begin to grow (hypertrophy) and begin secreting alkaline phosphatase, an enzyme essential for mineral deposition. Calcification of the matrix follows, and apoptosis (a type of cell death involving a programmed sequence of events that eliminates certain cells) of the hypertrophic chondrocytes occurs. This creates cavities within the bone. The exact mechanism of chondrocyte hypertrophy and apoptosis is currently unknown. The hypertrophic chondrocytes (before apoptosis) also secrete a substance called vascular endothelial cell growth factor that induces the sprouting of blood vessels from the perichondrium. Blood vessels forming the periosteal bud invade the cavity left by the chondrocytes, and branch in opposite directions along the length of the shaft. The blood vessels carry osteoprogenitor cells and hemopoietic cells inside the cavity, the latter of which later form the bone marrow. Osteoblasts, differentiated from the osteoprogenitor cells that enter the cavity via the periosteal bud, use the calcified matrix as a scaffold and begin to secrete osteoid, which forms the bone trabecula. Osteoclasts, formed from macrophages, break down the spongy bone to form the medullary cavity (bone marrow). The function of bone is to provide support, enhance leverage, protect vital structures, provide attachments for both tendons and ligaments, and store minerals, particularly calcium. From a clinical perspective, bones may serve as useful landmarks during the palpation phase of the examination. The strength of bone is related directly to its density. Of importance to the clinician, is the difference between maturing bone and mature bone. The epiphyseal plate or growth plate of a maturing bone can be divided into four distinct zones:37
 Reserve zone: produces and stores matrix.
Reserve zone: produces and stores matrix.
 Proliferative zone: produces matrix and is the site for longitudinal bone cell growth.
Proliferative zone: produces matrix and is the site for longitudinal bone cell growth.
 Hypertrophic zone: subdivided into the maturation zone, degenerative zone, and the zone of provisional calcification. It is within the hypertrophic zone that the matrix is prepared for calcification and is here that the matrix is ultimately calcified. The hypertrophic zone is the most susceptible of the zones to injury because of the low volume of bone matrix and the high amounts of developing immature cells in this region.38
Hypertrophic zone: subdivided into the maturation zone, degenerative zone, and the zone of provisional calcification. It is within the hypertrophic zone that the matrix is prepared for calcification and is here that the matrix is ultimately calcified. The hypertrophic zone is the most susceptible of the zones to injury because of the low volume of bone matrix and the high amounts of developing immature cells in this region.38
 Bone metaphysis: the part of the bone that grows during childhood.
Bone metaphysis: the part of the bone that grows during childhood.
Skeletal Muscle Tissue
The microstructure and composition of skeletal muscle have been studied extensively. The class of tissue labeled skeletal muscle consists of individual muscle cells or fibers that work together to produce the movement of bony levers. A single muscle cell is called a muscle fiber or myofiber. As muscle cells differentiate within the mesoderm, individual myofibers are wrapped in a CT envelope called endomysium. Bundles of myofibers, which form a whole muscle (fasciculus), are encased in the perimysium (Fig. 1-1). The perimysium is continuous with the deep fascia. This relationship allows the fascia to unite all of the fibers of a single motor unit and, therefore, adapt to variations in form and volume of each muscle according to muscular contraction and intramuscular modifications induced by joint movement.15 Groups of fasciculi are surrounded by a connective sheath called the epimysium (Fig. 1-1). Under an electron microscope, it can be seen that each of the myofibers consists of thousands of myofibrils (Fig. 1-1), which extend throughout its length. Myofibrils are composed of sarcomeres arranged in series.39
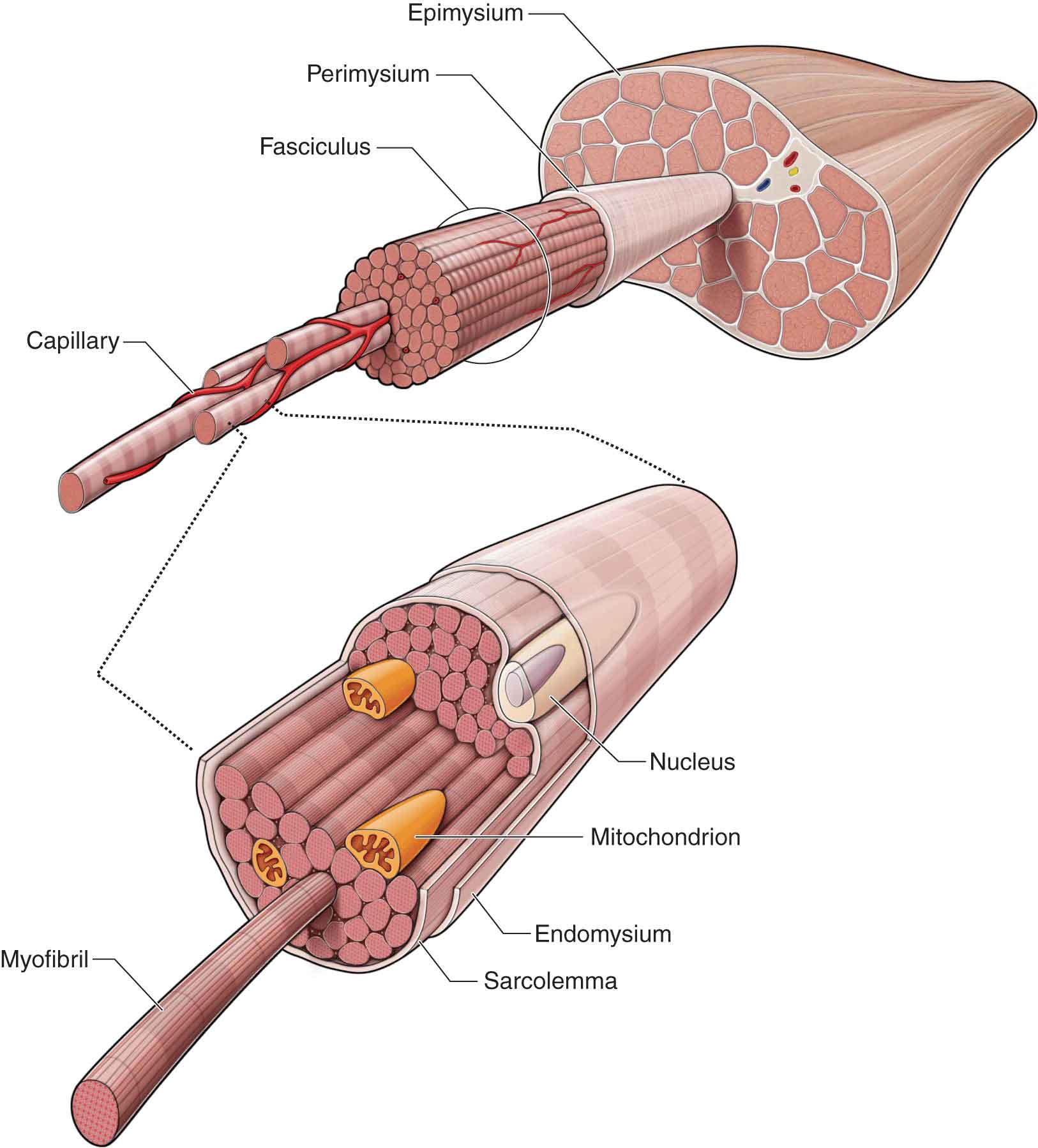
FIGURE 1-1 Microscopic structure of the muscle.
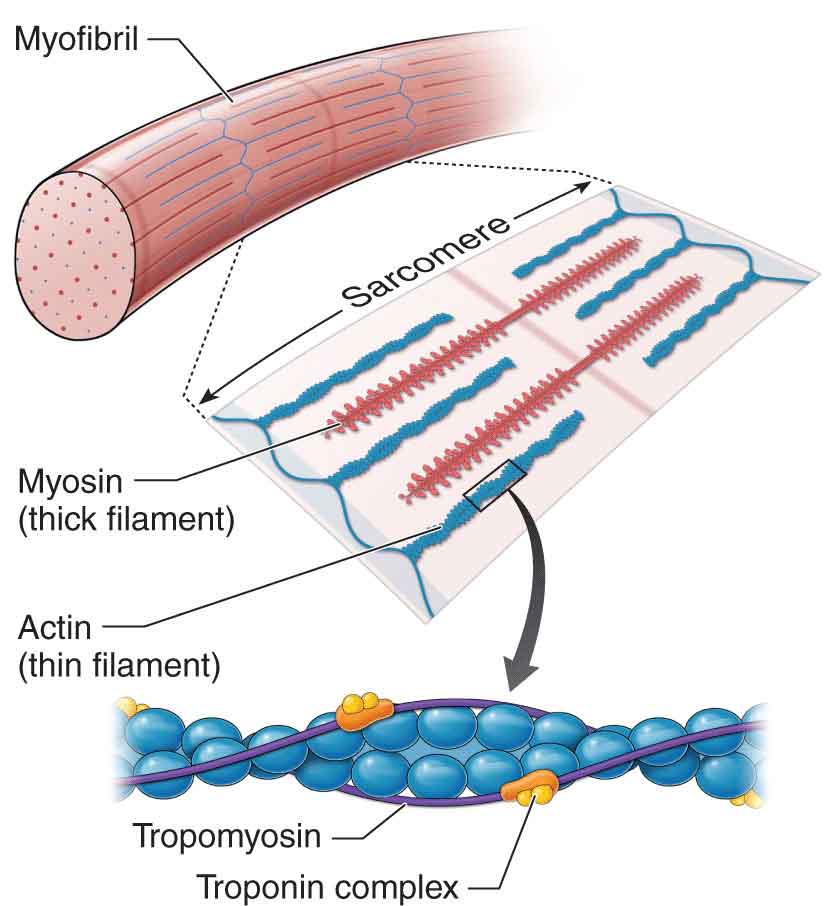
FIGURE 1-2 Troponin and tropomyosin action during a muscle contraction.
All skeletal muscles exhibit four characteristics:40
- Excitability, the ability to respond to stimulation from the nervous system.
- Elasticity, the ability to change in length or stretch.
- Extensibility, the ability to shorten and return to normal length.
- Contractility, the ability to shorten and contract in response to some neural command. The tension developed in skeletal muscle can occur passively (stretch) or actively (contraction). When an activated muscle develops tension, the amount of tension present is constant throughout the length of the muscle, in the tendons, and at the sites of the musculotendinous attachments to the bone. The tensile force produced by the muscle pulls on the attached bones and creates torque at the joints crossed by the muscle. The magnitude of the tensile force is dependent on a number of factors.
One of the most important roles of CT is to transmit mechanically the forces generated by the skeletal muscle cells to provide movement. Each of the myofibrils contains many fibers called myofilaments, which run parallel to the myofibril axis. The myofilaments are made up of two different proteins: actin (thin myofilaments) and myosin (thick myofilaments) that give skeletal muscle fibers their striated (striped) appearance (Fig. 1-2).39
The striations are produced by alternating dark (A) and light (I) bands that appear to span the width of the muscle fiber. The A bands are composed of myosin filaments, whereas the I bands are composed of actin filaments. The actin filaments of the I band overlap into the A band, giving the edges of the A band a darker appearance than the central region (H band), which contains only myosin. At the center of each I band is a thin, dark Z line. A sarcomere (Fig. 1-2) represents the distance between each Z line. Each muscle fiber is limited by a cell membrane called a sarcolemma (Fig. 1-1). The protein dystrophin plays an essential role in the mechanical strength and stability of the sarcolemma.41 Dystrophin is lacking in patients with Duchenne muscular dystrophy.
The basic function of muscle is to contract. The word contraction, used to describe the generation of tension within muscle fibers, conjures up an image of shortening of muscle fibers during a resistance exercise. However, a contraction can produce shortening or lengthening of the muscle, or no change in the muscle length. Thus, three types of contraction are commonly recognized: isometric, concentric, and eccentric (see Chapter 12).
 Isometric contraction. Isometric exercises provide a static contraction with a variable and accommodating resistance without producing any appreciable change in muscle length.42
Isometric contraction. Isometric exercises provide a static contraction with a variable and accommodating resistance without producing any appreciable change in muscle length.42
 Concentric contraction. A concentric contraction produces a shortening of the muscle. This occurs when the tension generated by the agonist muscle is sufficient to overcome an external resistance and to move the body segment of one attachment toward the segment of its other attachment.42
Concentric contraction. A concentric contraction produces a shortening of the muscle. This occurs when the tension generated by the agonist muscle is sufficient to overcome an external resistance and to move the body segment of one attachment toward the segment of its other attachment.42
 Eccentric contraction. An eccentric contraction occurs when a muscle slowly lengthens as it gives in to an external force that is greater than the contractile force it is exerting.42 In reality, the muscle does not lengthen, it merely returns from its shortened position to its normal resting length. Eccentric muscle contractions, which are capable of generating greater forces than either isometric or concentric contractions,43–45 are involved in activities that require a deceleration to occur. Such activities include slowing to a stop when running, lowering an object, or sitting down. Because the load exceeds the bond between the actin and myosin filaments during an eccentric contraction, some of the myosin filaments probably are torn from the binding sites on the actin filament while the remainder are completing the contraction cycle.46 The resulting force is substantially larger for a torn cross-bridge than for one being created during a normal cycle of muscle contraction. Consequently, the combined increase in force per cross-bridge and the number of active cross-bridges results in a maximum lengthening muscle tension that is greater than the tension that could be created during a shortening muscle action.46,47
Eccentric contraction. An eccentric contraction occurs when a muscle slowly lengthens as it gives in to an external force that is greater than the contractile force it is exerting.42 In reality, the muscle does not lengthen, it merely returns from its shortened position to its normal resting length. Eccentric muscle contractions, which are capable of generating greater forces than either isometric or concentric contractions,43–45 are involved in activities that require a deceleration to occur. Such activities include slowing to a stop when running, lowering an object, or sitting down. Because the load exceeds the bond between the actin and myosin filaments during an eccentric contraction, some of the myosin filaments probably are torn from the binding sites on the actin filament while the remainder are completing the contraction cycle.46 The resulting force is substantially larger for a torn cross-bridge than for one being created during a normal cycle of muscle contraction. Consequently, the combined increase in force per cross-bridge and the number of active cross-bridges results in a maximum lengthening muscle tension that is greater than the tension that could be created during a shortening muscle action.46,47
Four other contractions are worth mentioning:
 Isokinetic contraction. An isokinetic contraction occurs when a muscle is maximally contracting at the same speed throughout the whole range of its related lever.42 Isokinetic contractions require the use of special equipment that produces an accommodating resistance. Both high-speed/low-resistance and low-speed/high-resistance regimens result in excellent strength gains.48–51 The major disadvantage of this type of exercise is its expense. Also, there is the potential for impact loading and incorrect joint axis alignment.52 Isokinetic exercises may also have questionable functional carryover.53
Isokinetic contraction. An isokinetic contraction occurs when a muscle is maximally contracting at the same speed throughout the whole range of its related lever.42 Isokinetic contractions require the use of special equipment that produces an accommodating resistance. Both high-speed/low-resistance and low-speed/high-resistance regimens result in excellent strength gains.48–51 The major disadvantage of this type of exercise is its expense. Also, there is the potential for impact loading and incorrect joint axis alignment.52 Isokinetic exercises may also have questionable functional carryover.53
 Econcentric contraction. This type of contraction combines both a controlled concentric and a simultaneous eccentric contraction of the same muscle over two separate joints.54 Examples of an econcentric contraction include the standing hamstring curl, in which the hamstrings work concentrically to flex the knee while the hip tends to flex eccentrically, lengthening the hamstrings. When rising from a squat, the hamstrings work concentrically as the hip extends and work eccentrically as the knee extends. Conversely, the rectus femoris work eccentrically as the hip extends and work concentrically as the knee extends.
Econcentric contraction. This type of contraction combines both a controlled concentric and a simultaneous eccentric contraction of the same muscle over two separate joints.54 Examples of an econcentric contraction include the standing hamstring curl, in which the hamstrings work concentrically to flex the knee while the hip tends to flex eccentrically, lengthening the hamstrings. When rising from a squat, the hamstrings work concentrically as the hip extends and work eccentrically as the knee extends. Conversely, the rectus femoris work eccentrically as the hip extends and work concentrically as the knee extends.
 Isolytic contraction. An isolytic contraction is an osteopathic term used to describe a type of eccentric contraction that makes use of a greater force than the patient can overcome. The difference between an eccentric contraction and an isolytic contraction is that, in the former, the contraction is voluntary whereas, in the latter, it is involuntary. The isolytic contraction can be used in certain manual techniques to stretch fibrotic tissue (see Chapter 10).
Isolytic contraction. An isolytic contraction is an osteopathic term used to describe a type of eccentric contraction that makes use of a greater force than the patient can overcome. The difference between an eccentric contraction and an isolytic contraction is that, in the former, the contraction is voluntary whereas, in the latter, it is involuntary. The isolytic contraction can be used in certain manual techniques to stretch fibrotic tissue (see Chapter 10).
Structures called cross-bridges serve to connect the actin and myosin filaments. Increased synthesis of actin and myosin stimulates new myofibrils that are added to the external layers of the pre-existing myofibrils.55 The myosin filaments contain two flexible, hinge-like regions, which allow the cross-bridges to attach and detach from the actin filament. During contraction, the cross-bridges attach and undergo power strokes, which provide the contractile force. During relaxation, the cross-bridges detach. This attaching and detaching is asynchronous, so that some are attaching while others are detaching. Thus, at each moment, some of the cross-bridges are pulling, while others are releasing.
The regulation of cross-bridge attachment and detachment is a function of two proteins found in the actin filaments: tropomyosin and troponin (Fig. 1-2). Tropomyosin attaches directly to the actin filament, whereas troponin is attached to the tropomyosin rather than directly to the actin filament.
Each muscle fiber is innervated by a somatic motor neuron. One neuron and the muscle fibers it innervates constitute a motor unit or functional unit of the muscle. Each motor neuron branches as it enters the muscle to innervate a number of muscle fibers.
The release of a chemical acetylcholine from the axon terminals at the NMJ causes electrical activation of the skeletal muscle fibers. When an action potential propagates into the transverse tubule system (narrow membranous tunnels formed from and continuous with the sarcolemma), the voltage sensors on the transverse tubule membrane signal the release of Ca2+ from the terminal cisternae portion of the SR (a series of interconnected sacs and tubes that surround each myofibril).56 The released Ca2+ then diffuses into the sarcomeres and binds to troponin, displacing the tropomyosin, and allowing the actin to bind with the myosin cross-bridges (Fig. 1-2). Whenever a somatic motor neuron is activated, all of the muscle fibers that it innervates are stimulated and contract with all-or-none twitches. Although the muscle fibers produce all-or-none contractions, muscles are capable of a wide variety of responses, ranging from activities requiring a high level of precision, to activities requiring high tension.
At the end of the contraction (the neural activity and action potentials cease), the SR actively accumulates Ca2+ and muscle relaxation occurs. The return of Ca2+ to the SR involves active transport, requiring the degradation of adenosine triphosphate (ATP) to adenosine diphosphate (ADP)*.56 Because SR function is closely associated with both contraction and relaxation, changes in its ability to release or sequester Ca2+ markedly affect both the time course and magnitude of force output by the muscle fiber.57
*The most readily available energy for skeletal muscle cells is stored in the form of ATP and phosphocreatine (PCr). Through the activity of the enzyme ATPase, ATP promptly releases energy when required by the cell to perform any type of work, whether it is electrical, chemical, or mechanical.
On the basis of their contractile properties, two major types of muscle fiber have been recognized within skeletal muscle based on their resistance to fatigue: type I (tonic, slow-twitch fibers), and type II (phasic fast-twitch fibers). Type II muscle fibers are further divided into two additional classifications (Types IIA and IIB) (Table 1-3). Scott et al.58 subdivide type II fibers into three classifications, including a type IIIC.
TABLE 1-3 | Comparison of Muscle Fiber Types | ||
Characteristics | Type I | Type II A | Type II B |
Size (diameter) | Small | Intermediate | Very large |
Resistance to fatigue | High | Fairly high | Low |
Capillary density | High | High | Low |
Glycogen content | Low | Intermediate | High |
Twitch rate | Slow | Fast | Fast |
Energy system | Aerobic | Aerobic | Anaerobic |
Maximum muscle shortening velocity | Slow | Fast | Fast |
Major storage fuel | Triglycerides | Creatine phosphate glycogen | Creatine phosphate glycogen |
Type I fibers are richly endowed with mitochondria and have a high capacity for oxygen uptake. They are, therefore, suitable for activities of long duration or endurance (aerobic), including the maintenance of posture. In contrast, fast-twitch fibers, which generate a great amount of tension within a short period, are suited to quick, explosive actions (anaerobic), including such activities as sprinting. The type II (fast-twitch) fibers are separated based on mitochondria content into those that have a high complement of mitochondria (type IIA) and those that are mitochondria-poor (type IIB). This results in type IIB fibers having a tendency to fatigue more quickly than the type IIA fibers (Table 1-3).
Theory dictates that a muscle with a large percentage of the total cross-sectional area occupied by slow-twitch type I fibers should be more fatigue resistant than one in which the fast-twitch type II fibers predominate.
Different activities place differing demands on a muscle (Table 1-4).59 For example, dynamic movement activities involve a predominance of fast-twitch fiber recruitment, whereas postural activities and those activities requiring stabilization entail more involvement of the slow-twitch fibers. In humans, most limb muscles contain a relatively equal distribution of each muscle fiber type, whereas the back and trunk demonstrate a predominance of slow-twitch fibers. Although it would seem possible that physical training may cause fibers to convert from slow twitch to fast twitch or the reverse, this has not been shown to be the case.60 However, fiber conversion from type IIB to type IIA, and vice versa, has been found to occur with training.61
TABLE 1-4 | Functional Division of Muscle Groups |
Movement Group | Stabilization Group |
Primarily type IIa | Primarily type I |
Prone to adaptive shortening | Prone to develop weakness |
Prone to develop hypertonicity | Prone to muscle inhibition |
Dominate in fatigue and new movement situations | Fatigue easily |
Generally cross two joints | Primarily cross one joint |
Examples | Examples |
Gastrocnemius/Soleus | Fibularis (peronei) |
Tibialis posterior | Tibialis anterior |
Short hip adductors | Vastus medialis and lateralis |
Hamstrings | Gluteus maximus, medius, and minimus |
Rectus femoris | Serratus anterior |
Tensor fascia lata | Rhomboids |
Erector spinae | Lower portion of trapezius |
Quadratus lumborum | Short/deep cervical flexors |
Pectoralis major | Upper limb extensors |
Upper portion of trapezius | Rectus abdominis |
Levator scapulae | |
Sternocleidomastoid | |
Scalenes | |
Upper limb flexors | |
Data from Jull GA, Janda V. Muscle and motor control in low back pain. In: Twomey LT, Taylor JR, eds. Physical Therapy of the Low Back: Clinics in Physical Therapy. New York, NY: Churchill Livingstone; 1987:258–278. | |
The effectiveness of muscle to produce movement depends on some factors. These include the location and orientation of the muscle attachment relative to the joint, the limitations or laxity present in the musculotendinous unit, the type of contraction, the point of application, and the actions of other muscles that cross the joint.2
