CHAPTER 13 Osteochondral Lesions of the Talar Dome
Cartilage Replacement Using Osteochondral Autogenous Transplantation and Mosaicplasty
TERMINOLOGY
The term osteochondritis dissecans was first used in 1887, when Konig1 described an inflammatory lesion of cartilage and underlying bone in the knee joint that resulted in loss of structural integrity, causing pain and dysfunction. Since then, it has been recognized that lesions of the bone and cartilage may appear spontaneously or may be caused by a traumatic event to the joint surface.2 The general term osteochondral lesion (OCL) has gained more widespread use than osteochondritis dissecans.
ETIOLOGY, EPIDEMIOLOGY, AND CLASSIFICATION
In the ankle joint, an OCL can occur spontaneously or as the result of trauma, and there appears to be a relationship between the location of the lesion and the cause.3,4 In a comprehensive review of the early literature that included more than 500 cases of OCL, Flick and Gould5 found that 98% of lateral lesions and only 70% of medial lesions were associated with a traumatic event. This and other reports2,6 led to the associations of traumatic OCLs with lateral lesions and spontaneous OCLs with medial lesions.
The incidence of OCL depends on the etiologic factors involved. In an early study, Bosien and colleagues7 reported the incidence of OCL to be 6.5% with ankle sprains, and Hinterman and coworkers8 and Leontaritis and associates9 reported the incidence of OCL as 69% and 74%, respectively.
The most recognized classification system is that of Berndt and Harty,2 which is useful for research purposes and preoperative planning. It is based on radiographic imaging or intraoperative findings. Stage I is a small, subchondral compression; stage II is partial fragment detachment; stage III is complete fragment detachment with no displacement; and stage IV is complete fragment detachment with displacement.
PATIENT EVALUATION
History and Physical Examination
Physical examination should begin with assessment of the standing alignment to identify an associated coronal plane deformity. Inspection of the ankle with an acute injury usually reveals swelling and ecchymosis, but the ankle sometimes appears completely normal, as is often the case with delayed presentations. An attempt to elicit tenderness with palpation should be made by focusing on the common OCL sites. Posteromedial lesions may be tender in response to palpation when the ankle is dorsiflexed and the region posterior to the medial malleolus is palpated. Anterolateral lesions may be tender when the joint is palpated laterally with the ankle in plantar flexion. These findings are often nonspecific because the tenderness is likely related to joint synovitis rather than the OCL itself. The joint should be taken through a range of motion to assess stiffness and to feel for crepitus and mechanical signs of clicking or locking. Ligamentous stability should be assessed to ascertain evidence of associated laxity, particularly of the lateral ligament complex. The examination is completed with a thorough neurovascular assessment and examination of the contralateral ankle for comparison.
Diagnostic Imaging
MRI affords an accurate assessment of the ankle’s soft tissue structures, including the articular cartilage and supporting ligaments. Multiplanar evaluation is possible with no radiation exposure to the patient. Sequences can identify the size and location, and they provide information about the stability of the lesion. DeSmet and colleagues10 prospectively showed an excellent correlation between staging with MRI and arthroscopic results, correctly predicting stable and unstable lesions in 92% of cases. In some circumstances, MRI may overestimate the size of the OCL due to associated edema, and CT may be better suited for defining the dimensions of the defect.
TREATMENT
The use of non–weight-bearing articular surfaces as a donor site for osteochondral autogenous transplantation (OAT) grafts for the treatment of OCL began in the 1980s. Yamashita and coworkers11 published a case report on two patients who had an OAT graft harvested from the non–weight-bearing portion of the medial femoral condyle and transplanted to an OCL of the same knee.
In 1993, Matsusue and associates12 reported their 3-year follow-up of cases treated with a new technique that involved transplantation of multiple osteochondral plugs harvested from the same knee. They used the press-fit method of cylindrical osteochondral plugs described by Garrett13 for allografts. They harvested two osteochondral autogenous graft (OAG) plugs from the lateral femoral patellar groove and one from the anterolateral side of the intercondylar notch.
In 1996, Bobic14 was the first to publish a case series (12 patients) describing multiple OAT grafts for the treatment of knee OCLs associated with anterior cruciate ligament reconstruction. In 1997, Hangody and colleagues15 published their first report of their prospective case series. They described 44 patients with 1 to 5 years of follow-up after multiple OAG plugs were used to treat OCLs of the knee. Most patients had good or excellent outcomes. They were the first to call the procedure mosaicplasty. We now commonly use this term for the technique of using multiple OAT graft plugs to treat an OCL. Some surgeons make distinction between OAT and mosaicplasty, using the former to describe the use of only one graft plug and the latter to describe the use of multiple graft plugs. These terms can be used interchangeably, but for clarification, we use the term mosaicplasty osteochondral autogenous transplantation (MOAT) when referring to transplantation with multiple graft plugs and OAT when referring to transplantation of a single graft plug.
Evidence-Based Studies of Osteochondral Autogenous Transplantation and Mosaicplasty Osteochondral Autogenous Transplantation
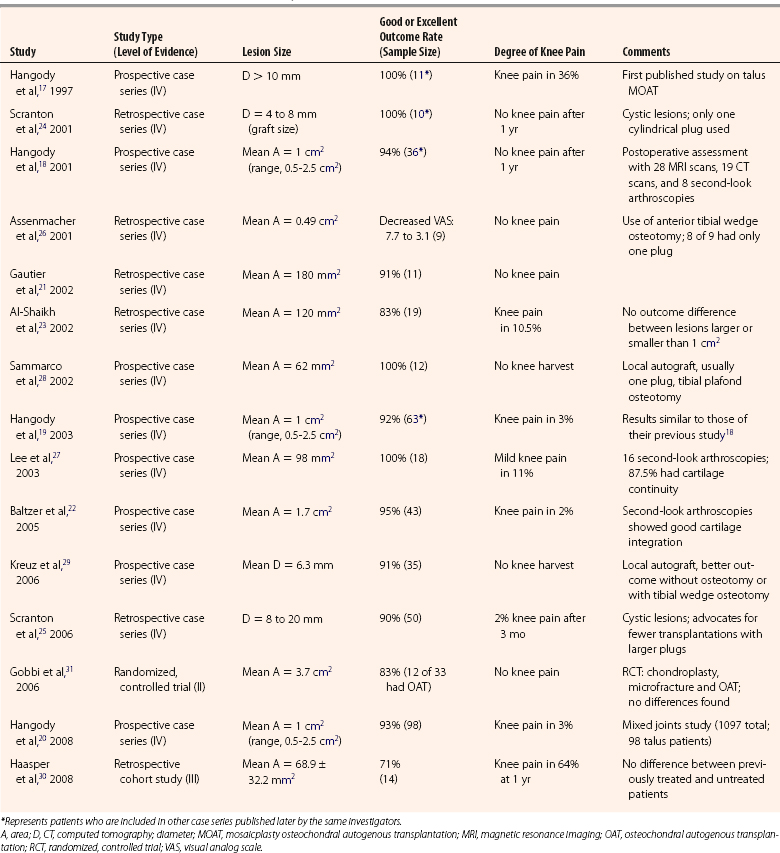
Given the success of OAT and mosaicplasty in the knee, there was a logical extension of this technique to the ankle. In reviewing the literature on OAT and MOAT in the ankle before February 2009, 15 studies were identified. All articles were reviewed and assigned a classification (i.e., I through IV) using the Journal of Bone and Joint Surgery levels of evidence for primary research questions.16 The 15 studies included 1 level II study, 1 level III study, and 13 level IV studies (Table 13-1).
TABLE 13-1 Clinical Outcome Studies of Mosaicplasty Osteochondral Autogenous Transplantation for Talar Osteochondral Lesions
Level IV Evidence
In 1997, Hangody and coworkers17 were the first to report on use of MOAT for treatment of OCL of the talus. In a preliminary report of a prospective case series (level IV), MOAT was done on 11 patients with OCLs larger than 10 mm diameter. The outcome results after at least 1 year of follow-up were excellent and promising. This study included second-look arthroscopies in three patients and one cartilage biopsy. Arthroscopy showed good incorporation of the grafts, with articular surface congruency and normal cartilage appearance. Biopsy showed normal articular hyaline cartilage (i.e., collagen type II) and proteoglycans and fibrocartilage bonding at the graft-recipient junction. Donor-site morbidity was a concern, because 3 of 11 patients had occasional knee pain and 2 of 11 had knee pain with activities (one with a 10-degree extension lag).
In 2001,18 2003,19 and 2008,20 Hangody and coworkers expanded their prospective case series report (level IV) after 2 to 9 years of follow-up after MOAT for OCL of the talus. The report included 98 patients with 92% to 94% good or excellent clinical results and less than 3% had knee pain after 1 year. Further assessment included 28 MRI scans, 19 CT scans, 8 second-look arthroscopies, and 4 biopsies. CT, MRI, and arthroscopy showed good incorporation of the grafts, with articular surface congruency and normal cartilage appearance. Biopsies showed normal articular cartilage collagen (type II) and proteoglycans. Three postoperative knee arthroscopies showed a donor-site defect covered by fibrocartilage congruent with the surrounding hyaline cartilage.
In smaller case series (level IV), Gauthier and colleagues21 and Baltzer and coworkers22 described 11 (retrospective) and 43 (prospective) cases, respectively, that had MOAT for OCL of the talus. More than 90% of their patients were satisfied with the results. In another retrospective case series (level IV), Al-Shaihk and associates23 performed MOAT on 19 patients with larger OCLs (mean area of 120 mm2). Eighty-three percent of patients were satisfied with the results, and two patients (10.5%) reported mild knee pain. They found no difference in outcomes between patients with lesions larger than 1 cm2 and those with lesions smaller than 1 cm.2
In 200124 and 2006,25 Scranton and colleagues confirmed earlier promising results with a retrospective case series (level IV) that evaluated 50 patients. Their technique included the transplantation of only one cylindrical plug in patients having cystic lesions that were 4 to 20 mm of diameter. The results were equally impressive, and the investigators advocated arthroscopic harvesting and the use of larger and fewer plugs to minimize peripheral graft chondrocytes death and to simplify the technique. In a retrospective case study (level IV), Assenmacher and coworkers26 reported the use of single-plug OAT in 8 of 9 patients, who had an average decrease in the visual analog scale (VAS) score of 7.7 to 3.1 after the procedure. They also described the use of an anterior tibial wedge osteotomy (i.e., plafondplasty) in five patients for lesions in the central third of the talar dome to avoid a malleolar osteotomy. Preoperative MRI showed an excellent correlation for OCL grading, and postoperative MRI showed integration of the grafts.
In a prospective case series (level IV) reported by Lee and associates,27 18 patients had 100% good or excellent outcomes. Two of the 18 patients (11%) had mild knee pain, and 14 (87.5%) of 16 second-look arthroscopies showed cartilage continuity with the native cartilage.
To minimize the risk of knee pain associated with violating an asymptomatic joint to harvest the OAG, Sammarco and colleagues28 performed a local osteochondral graft for an OCL with a mean area of 62 mm,2
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








