Osteoarthritis
Ira Zaltz and Brian Larkin
Key Points
• Epidemiology and risk factors
• Clinical features and diagnosis
• Hip OA presents with groin pain, decreased range of motion, and gate alterations.
Introduction
A comprehensive definition of osteoarthritis (OA) was agreed upon at a 1994 workshop entitled “New Horizons in Osteoarthritis,” which was sponsored by the American Academy of Orthopaedic Surgeons; the National Institute of Arthritis, Musculoskeletal, and Skin Diseases; the National Institute on Aging; the Arthritis Foundation; and the Orthopaedic Research and Education Foundation:
“Osteoarthritic (OA) diseases are a result of both mechanical and biologic events that destabilize the normal coupling of degradation and synthesis of articular cartilage chondrocytes and extracellular matrix, and subchondral bone. Although they may be initiated by multiple factors, including genetic, developmental, metabolic, and traumatic, OA diseases involve all of the tissues of the diarthrodial joint. Ultimately, OA diseases are manifested by morphologic, biochemical, molecular, and biomechanical changes of both cells and matrix which lead to a softening, fibrillation, ulceration, loss of articular cartilage, sclerosis and eburnation of subchondral bone, osteophytes, and subchondral cysts. When clinically evident, OA diseases are characterized by joint pain, tenderness, limitation of movement, crepitus, occasional effusion, and variable degrees of inflammation without systemic effects.”1
OA, a common condition affecting millions of people, is associated with pain and disability that have a negative impact on quality of life, productivity, self-worth, and self-related health. The estimated cost of lost productivity and employment for American workers between the ages of 40 and 65 is greater than $7 billion.2 Currently, approximately 27 million Americans older than age 25 are limited by OA.3 As the population ages, the prevalence of OA is expected to increase substantially4; OA already trails only cardiovascular, cerebrovascular, and pulmonary disease in terms of disability in people older than age 65.5
Although the final clinical appearance and common pathway of hip OA may appear similar, multiple precipitating causes are known, including trauma, lower extremity misalignment, developmental abnormalities, synovial disease, infection, and crystalline arthropathy. It is now understood that subtle anatomic abnormalities that have different pathomechanical consequences and can be subclinical initially may initiate a continuum of disease that may not manifest until early adulthood or beyond. Thus the common pathway of OA leads to irreversible damage to the acetabular labrum and articular surface that causes joint dysfunction resulting in deformity, reduction in range of motion, and pain.
Many different treatment options are available to the clinician and patient; current accepted treatment goals are to improve function, decrease pain, and preserve the hip joint for as long as possible. Treatment modalities include a variety of medications, physical therapies and exercises, alternative modalities, and operative interventions ranging from arthroscopy to arthrotomy, osteotomy, arthrodesis, and arthroplasty.
This chapter will focus on the epidemiology and risk factors for osteoarthritis, the pathophysiology, the clinical features and diagnostic tests, the differential diagnosis, treatment options, and the prognosis of this common musculoskeletal disease.
Epidemiology and Risk Factors
Hip osteoarthritis (OA) is a common condition that affects millions of people worldwide and is present in virtually all races. Although the clinical course, presentation, and patient demographics vary considerably, all patients share a common degenerative pathway. Prevalence varies by race and regional population. In a population-based study in Finland, hip OA was found to affect 4.9% of people.6 In South Korea, the prevalence of hip OA was estimated to be 1.2%, with men affected more frequently than women.7 In England, the prevalence was 7% in males and 10% in females over the age of 45.8 Within the United States, the prevalence of radiographic hip OA in population studies has varied, ranging from 7%9 to 28%.10 A systematic literature review found that the prevalence of radiographic hip OA ranges between 0.9% and 27%, depending on the population studied, with a mean of 8% and a standard deviation of 7%.11
Much of the difficulty involved in arriving at an accurate estimate for the prevalence of hip OA is explained by differing clinical and radiographic criteria used to define the disease. Many attempts have been made to quantify the radiographic changes noted at the onset of hip OA. Kellgren and Lawrence relied on assessing progressive radiographic changes seen in OA. They used the following criteria: formation of marginal joint osteophytes; periarticular ossicles; narrowing of joint cartilage associated with sclerosis of subchondral bone; small pseudocystic areas with sclerotic walls usually found in the subchondral bone; and altered shape of the bone ends, particularly in the head of the femur. Their grading system ranges from 0 to 4 as follows: 0—no findings, 1—equivocal findings, 2—minimal findings, 3—moderate findings, and 4—severe findings.12 Tonnis used many of the same radiographic findings but defined his grades slightly differently.13 Grade 0 has no changes. Grade 1 has increased sclerosis of the femoral head and acetabulum, a slight decrease in the height of the cartilage, and slight osteophytes. Grade 2 reveals small cysts in the femoral head or acetabulum, a marked decrease in the height of the cartilage, and slight deviation from the round form of the femoral head. Grade 3 shows large cysts in the femoral head and acetabulum, a severe decrease in cartilage up to complete absence of the joint cleft, severe deviation of the round form of the femoral head, or avascular necrosis.
Both descriptive grading systems start with the formation of osteophytes, progress to narrowed joint spaces and small cystic changes, and conclude with severe changes in the shape of the bone ends on either side of the joint. Croft compared seven radiographic indices of hip OA in an attempt to quantify the radiographic factor most strongly associated with the clinical picture of OA.14 Four measures of the joint space, the maximum thickness of subchondral sclerosis, and the size of the largest osteophyte were analyzed; minimum joint space was determined to be the best predictor of symptomatic OA—a conclusion that has been supported in other studies.15–17 It is now understood that once radiographic evidence of arthrosis is visible, chondral and labral damage is irreversible, and plain radiographic findings of arthritis probably underestimate the actual prevalence of hip joint disease. Newer imaging modalities such as magnetic resonance imaging (MRI) have been improved in terms of their ability to diagnose OA before the onset of plain radiographic changes and will be used in the future for grading early findings of OA (Fig. 32-1). Newer biochemical imaging modalities such as delayed enhanced magnetic resonance imaging of cartilage (dGMERIC) are now used to estimate the quantity of negative charge within hyaline cartilage—a measure that correlates with chondral integrity.
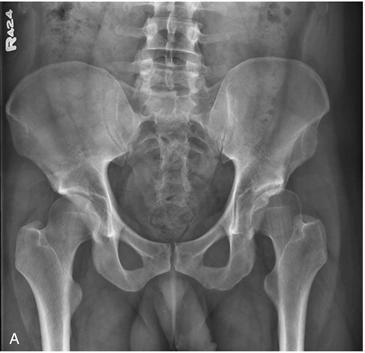
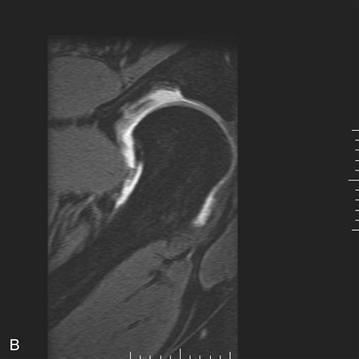
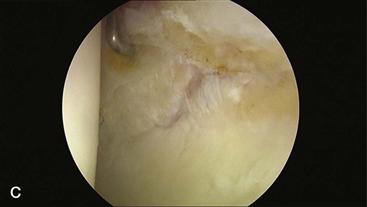
Figure 32-1 A, Anteroposterior (AP) pelvis demonstrating early arthritic changes in the left hip with sclerosis of the sourcil. B, Magnetic resonance imaging (MRI) arthrogram of the left hip demonstrating chondrolabral separation with paraganglion cyst formation. C, Hip arthroscopy images noting chondrolabral separation.
Many proposed occupational and avocational risk factors for the development of OA are associated with repetitive or sustained mechanical joint overload. Any anatomic abnormality may potentiate the effects of excessive, sustained extrinsic and intrinsic joint overload. Accordingly, these individuals are at risk for developing end-stage OA at a younger age.
Obesity.
The effect of body habitus on the development of OA has regional variations. In Australia, body weight, body mass index (BMI), fat mass, percentage fat, waist circumference, and waist-to-hip ratio have been correlated with increased risk of arthroplasty.18 These factors are more highly correlated with knee OA than hip OA. A linear relationship has been noted between BMI and the risk of severe OA in Swedish construction workers.19 Among the Dutch, obesity was associated with both self-reported OA and chronic pain; the higher the weight, the stronger was the association.20 Compared with Caucasians, Asians presenting with end-stage OA have worse joint pain and dysfunction and present at a younger age with a lower BMI.21 A systematic literature review for obesity as a specific risk factor for hip OA found moderate evidence of a positive association, with an odds ratio of approximately 2.22
Heavy manual labor.
It is assumed that hips subjected to increased levels of stress are more likely to develop OA. A history of heavy manual labor seems to be associated with an increased prevalence of hip OA. A study evaluating occupational risk and hip OA in French workers showed that female cleaners had the greatest prevalence of OA with an odds ratio of 6.2, followed by women in the clothing industry (odds ratio 5.0), male construction workers (odds ratio 2.9), and agriculture workers (odds ratio 2.8). Early onset of OA was seen in those exposed to heavy labor jobs, and almost 40% reported initial symptoms before the age of 50.23 A Finnish series demonstrated that the odds ratio of developing hip OA with a history of heavy manual labor was 6.7 compared with 5.0 for previous trauma to the hip.24 A systematic literature review looking at 16 studies demonstrated a moderate association between previous heavy workload and the occurrence of hip OA, with an odds ratio of 3.25
Gender.
In most series, women have a higher prevalence of hip OA than men.26–28 Females are particularly at risk after menopause, showing a progressive postmenopausal decline in joint space compared with age-matched males.15,29 The exact cause of this female preponderance is not known and is likely multifactorial. Alterations in hip joint biomechanics that increase the joint reactive forces in older females may cause joint overload.30 The predilection for hip dysplasia in females and its potential to predispose to OA may contribute.
Trauma.
A history of trauma to a joint surface increases the chance of developing late OA. Because the articular cartilage is avascular and has limited cellularity, its reparative capacity is limited. Consequently, trauma that alters the articular surface initiates the degenerative cascade. As the surface is exposed to physiologic joint forces, chondrocytes are unable to maintain a homeostatic environment. Consequently, degenerative changes develop and the degenerative biological cascade progresses.
Anatomic anomalies.
Certain anatomic variants that have been described can predispose to the development of osteoarthritis. Subtle anatomic variations contribute to the development of femoroacetabular impingement (FAI), which has been associated with subsequent development of cartilage degeneration and osteoarthritis.31,32 Certain conditions associated with abnormal acetabulum orientation, such as acetabular retroversion, coxa profunda, and coxa protrusion, predispose to FAI. Similarly, patients with acetabular dysplasia are at risk for damage to the chondrolabral junction and progressive development of OA. Murphy demonstrated that patients with a center edge angle of Wiberg measuring less than 16 degrees developed osteoarthritis by age 65.33 Decreasing anterior center edge angle and the presence of a labral tear have also been associated with the development of osteoarthritis.34 Other growth-related disorders such as Legg-Calvé-Perthes and slipped capital femoral epiphysis (SCFE) that alter the anatomy of the hip joint are associated with long-term hip dysfunction and development of osteoarthritis. The concept that FAI is a prearthritic condition has been suggested31,32,35 and is consistent with anatomic conditions associated with repetitive abnormal contact between the proximal femur and the acetabulum. Retroverted acetabuli have been shown to be present in 20% of patients undergoing total hip arthroplasty compared with 5% of the general population.36 Retroversion has been noted in 20% of patients with OA, 18% of patients with developmental dysplasia of the hip, 6% of patients with osteonecrosis, and 42% of patients with Legg-Calvé-Perthes.37 Furthermore, variations in proximal femoral anatomy that decrease head-neck offset or produce aspherical femoral heads can contribute to the development of FAI and subsequent OA.
Pathophysiology
Multiple anatomic and inflammatory factors predispose patients and contribute to the biomechanical environment associated with the development of hip arthritis. These may be inherited congenital anatomic variations or acquired abnormalities that may be cumulative over time. These factors eventually produce degenerative changes within the joint and the slow development of end-stage OA. Although each factor can affect the hip differently, if exposure is substantially injurious or of sufficient duration, the inflammatory cascade associated with the development of hip arthritis will follow.
Some of the early changes in hip OA are exemplified by femoroacetabular impingement. In anatomically predisposed hips, the proximal femur repetitively abuts the acetabular rim during normal range of motion or predisposing activities. Two primary pathomechanisms of FAI are predisposed: cam-type and pincer-type impingement. Cam-type impingement, a complex femoral-based deformity, results in impingement on a normal acetabulum. Resultant patterns of injury secondary to cam impingement include labral tears, chondral delamination, acetabular rim fracture, and proliferation of bone and fibrous tissue on the anterosuperior femoral neck.31,38 Pincer-type impingement, secondary to an acetabular-based deformity, results in damage to the acetabular rim, labrum, and peripheral articular cartilage. Most commonly, patients presenting with impingement have a combination of cam and pincer features that lead to coexisting patterns of acetabular damage. The femoral head articular cartilage is generally relatively well preserved until late in the cascade of hip OA.
Articular cartilage is the primary degenerative tissue affected in hip OA. Articular cartilage is an aneural and avascular multilayered structure composed of chondrocytes, glycoproteins, matrix molecules, and water that absorbs stress and distributes loads evenly through a joint. Articular cartilage, when properly lubricated, minimizes friction between articulating surfaces and allows for pain-free range of motion. Water, adsorbed within the extracellular matrix, facilitates chondrocyte nutrition and assists in joint lubrication. Shifts in water allow cartilage to deform in response to stress. Type II collagen accounts for approximately 95% of collagen within the articular cartilage. Type II collagen functions as a framework for the cartilage and provides tensile strength. Proteoglycans provide compressive strength and are synthesized by chondrocytes in subunits called glycosaminoglycans.
Articular cartilage has three distinct layers: superficial, middle, and deep. The superficial layer is composed of collagen fibers arranged tangentially to the articular surface and is thought to resist shear forces. The middle layer, with fibers oriented obliquely, is thought to resist compressive forces; the deep layer, with fibers oriented vertically, resists compressive forces as well. The articular cartilage is adherent to the subchondral bone plate by the tidemark, which is a histologically unique layer of calcified cartilage that merges with the subchondral bone.
In the arthritic hip, a combination of factors leads to end-stage OA. Articular cartilage depends on the integrity of its extracellular matrix for optimal function. Chondrocytes maintain a homeostatic environment by regulating competing catabolic and anabolic pathways (Fig. 32-2).39 Biomechanical abnormalities and associated progressive chondral injury lead to damage that exceeds the capacity of the chondrocyte for repair.40 These catabolic changes are mediated in part by cytokine production from both the synovium and the chondrocytes.41 Cytokines act on the release of pro-matrix metalloproteinases (MMPs) by the chondrocyte. Pro-MMPs are turned into active MMPs by a process that is regulated by the plasminogen activator/plasmin system and other proteases. Active MMPs induce matrix degradation and cartilage damage, while tissue inhibitors of MMPs (TIMPs) are able to bind the active site of degradative MMPs and thus slow cartilage degradation.
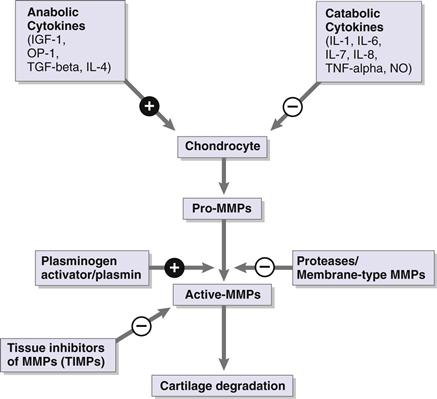
Figure 32-2 Competing anabolic and catabolic pathways leading to cartilage degradation.
Interleukin (IL)-1 and transforming growth factor-beta (TGF-β) are two of the known catabolic and anabolic stimuli for chondrocytes.42 Catabolic cytokines such as IL-1, tumor necrosis factor-alpha (TNF-α), and inducible nitric oxide synthase (iNOS) have been localized within the superficial zone of arthritic cartilage.43 These changes are thought to be caused by the biomechanical stresses seen in OA, as unloaded chondrocytes do not have iNOS present, whereas intermittent and static compression of chondrocytes can induce the upregulation of iNOS.44 Chondrocyte senescence also plays a role in this balance. As the chondrocyte ages, it is less responsive to anabolic stimuli.45 With continued improved detection of these inflammatory signaling pathways, a better understanding of this complex process will potentially guide future treatment options.
Early compositional changes seen in articular cartilage affected by osteoarthritis include an increase in water content and progressive disorganization of the collagen framework. As degeneration progresses, the articular surface becomes more susceptible to shear forces that aggravate cartilage degeneration. While proteoglycan synthesis increases, the overall content decreases and the chains become shorter secondary to marked increases in proteoglycan degradation.
As the articular surface is degenerating, changes in the subchondral bony architecture are noted. The cause of these changes is not clear, but the thickness of the subchondral bone plate increases and the trabecular architecture of the subchondral bone becomes disorderly as the layer of calcified cartilage begins to advance into the deep layer of hyaline cartilage. Subsequently, osteophytes begin to form at the articular margin, and subchondral cysts begin to form.46 Progressive edema of the marrow is also noted.
The consequences of increased subchondral bone plate thickness have not been clearly resolved. Radin proposed that the progressive thickness and volume increase the stiffness of the subchondral bone, which negatively influences the biomechanical environment of the articular cartilage.47 More recent contradictory studies48–50 have demonstrated that the progressive volume of subchondral bone is associated with decreasing subchondral stiffness. Within the subchondral bone plate, an increase in the vertical orientation of trabeculae, as well as thinning of the normal trabeculae, is thought to make the area relatively osteoporotic and less able to tolerate loading.51 Coincident with the increase in thickness of the subchondral bone plate, the zone of calcified cartilage, or the tidemark, advances. Although the mechanism of this phenomenon has not been elucidated, progressive tidemark advancement further diminishes the thickness of the residual articular cartilage. This leads to increasing mechanical stresses in the deep zone, likely contributing to further degradation of the articular cartilage.52
Other pathologic changes noted include bone marrow edema, subchondral cyst formation, microfractures, and osteophyte formation. Analysis of areas of bone marrow edema revealed that 70% of patients had pseudocysts in regions of bone marrow edema with microfractures of the trabecular bone in various stages of healing. These pathologic changes correlated with the area of most severe damage of the overlying cartilage.53 Studies of osteophyte formation in animal models have demonstrated initial proliferation of periosteal cells at the joint margin that differentiate into chondrocytes, which hypertrophy and ossify through endochondral ossification.54 Local production of growth factors, including TGF-β and bone morphogenic proteins (BMPs), has been implicated in the formation of osteophytes.55,56 Although osteophyte formation is ubiquitous and osteophytes are seen in areas of increased joint loading, the precise pathogenesis of osteophytes and their adaptive role remain unclear.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








