29 Key Points 1. Organizational, regulatory and financial barriers must be overcome to bring new basic scientific discoveries that have the promise of improving the outcome of spinal cord injury to clinical trials. The present chapter describes the steps taken to overcome these barriers and to develop a clinical trials network for new treatments for spinal cord injury. The rapid advance of knowledge of the cellular and molecular responses of the spinal cord to injury has led to therapies that have improved functional recovery after spinal cord injury (SCI) in laboratory studies.1 Some of these therapies have been used in small numbers of SCI patients.2 However, most of these promising developments have not been brought to phase 2 or 3 clinical trials that have the design and statistical power to demonstrate efficacy because of the formidable organizational, regulatory, and financial barriers that must be overcome to conduct such trials. This chapter describes the steps in building the North American Clinical Trials Network (NACTN) for Treatment of Spinal Cord Injury. NACTN was created in 2004 with the support of the Christopher Reeve Foundation with the goal of overcoming these barriers. The Telemedicine and Advanced Technology Research Center (TATRC) United States Army Medical Research and Materiel Command (USAMRMC) of the Department of Defense has supported NACTN since 2006. Since 2007, Walter Reed Army Medical Center (WRAMC) has been part of NACTN with the goal of comparing the natural history of military and civilian injuries. NACTN’s mission is to carry out clinical trials of the comparative effectiveness of new therapies for SCI using a consortium of neurosurgery departments at university-affiliated medical center hospitals with medical, nursing, and rehabilitation personnel who are skilled in the evaluation and management of SCI. NACTN is the only standing network for clinical trials for SCI in North America. NACTN is composed of clinical, data management, and pharmacological centers. Fig. 29.1 NACTN clinical centers. An increasing number of clinical trial networks have been formed to study a wide variety of diseases.3 This growth has been encouraged by the National Institutes of Health (NIH)4 and the Institute of Medicine,5 and it has been facilitated by Internet communication and the formation of societies and journals devoted to clinical trials.6 The prototype of a clinical trial network for SCI was the National Acute Spinal Cord Injury Study (NASCIS) supported by the NIH, which published its first trial of corticosteroid therapy for SCI in 1984.7 NASCIS did not continue as a clinical trial organization for testing new therapies after the publication of its third corticosteroid trial in 1997.8 In the past decade, SCI clinical trial networks have been organized in Europe—the European Multicenter Study about Spinal Cord Injury (EMSCI)9—and in Canada and the United States—the Surgical Treatment in Acute Spinal Cord Injury Study (STASCIS),10 which was formed to study early surgical decompression of cord injuries. NACTN is collaborating with these organizations in studies of outcome after SCI. 1. Recruitment of network components: Clinical Centers, Data Management Center, Pharmacological Center 2. Creation of a Data Registry of the natural history of SCI patients 3. NACTN’s first clinical trial, of a neuroprotective drug, riluzole Each stage has involved multiple steps, as described next. 1. Emergency transport systems providing rapid transport of SCI patients to a level 1 emergency department at the clinical center 2. An intensive care unit with neurointensivists 3. Neurosurgical, orthopedic, critical care, and physiatry staff with research and clinical expertise in neurotraumatology 4. A closely affiliated rehabilitation hospital 5. Mutual trust of the centers in each other based upon previous joint experience in research The initial five clinical centers were expanded to nine centers between 2006 and 2008. Each center is led by a principal investigator and at some centers a co-principal investigator. Each clinical center has a study coordinator, usually a nurse-clinician, and two clinical research assistants. Case report forms were developed to capture aspects of the medical history that have prognostic value. Forms were designed to obtain detailed information about complications that occur during the course of treatment. Clinical examinations were chosen to quantify the course of functional recovery in patients. A data archival system was developed to create a registry that can be interrogated to provide a historical control group to aid in clinical trial design. Multiple meetings were held by the investigators to choose the data elements that would be collected and to design the case report forms. The investigators had the benefit of consulting existing SCI registries, including those of the Model Spinal Cord Injury Systems,11 the International Spinal Cord Injury Data Set Elements that are currently being incorporated into the NIH–National Institute of Neurological Disorders and Stroke (NINDS) Common Data Elements12 and those of STASCIS and EMSCI. Developing prognostic algorithms required collecting comprehensive data that included a medical history, medications, the radiology of the injury, medical and surgical treatment, the physiological response to injury and complications, as well as neurological examinations from the earliest feasible time to examinations during rehabilitation and for a year after injury. The case report forms are divided between measures obtained during the acute hospitalization following injury and functional outcome measures obtained during rehabilitation. The case report forms are available from the coordinating center to investigators who are developing clinical trials. Data are collected under 12 headings: Functional outcomes are measured at specified times: The Manual of Operations (MOO) was written to serve as a guide for completing the data forms. It is a 55-page document that summarizes the research protocol and inclusion/exclusion criteria, and gives explicit instructions about each form. The center study coordinators are also in frequent telephone and e-mail communication with the Clinical Coordinating Center and the Data Management and Statistical Center (DMC) to clarify questions that arise. A multipart paper/optical scanning system was judged to be the best initial system for data collection because it would allow for multiple protocol iterations and adjustments required as experience was gained in data collection. The data collection system is a high-security system. Data from patients that are submitted to the DMC are identified only by a code number for the center and a number for the patient. Each data collection form was designed in Verity TeleForm software that uses an optical recognition system to digitally capture paper-based images and their content by scanner and allows customized field-by-field data verification. The data processing activities are managed by an integrated data system that uses TeleForm for data entry and verification and then uploads data to a secure Structured Query Language (SQL) server, which simultaneously generates Microsoft (MS) Access files and tables. This integrated system is augmented by a parallel data quality control system. The NACTN registry receives completed paper forms via mail from the clinical centers. Data forms are reviewed for completeness before being sent to the optical scanner. Forms are held by the electronic data processing system until they are audited by a verification program that allows field-by-field data verification. Each scanned form is maintained in the system as a TIF file, allowing computerized images of the data forms to be stored electronically. Once the data forms have been verified, the data go to a secure password-protected SQL server database. This server is backed up nightly with archival storage maintained at two alternate off-site locations. Once data have been committed to the data tables, the data are subjected to logic and edit checks as well as cross-checks between different form types. A separate file tracks each batch of data entered through the scanning system. Additionally, a correction database has been developed to document data change or data transactions that result from logic/error checks or updates received from clinical centers. Computer programs have also been written to generate one-page patient narrative reports. Export procedures were developed to convert data to Statistical Analysis System (SAS) and State formats to facilitate statistical analyses. Programs have been developed to tabulate summary statistics and generate reports. All steps from data logging, entry, committing to SQL and MS Access, editing, reporting, and summary analysis have been closely documented and are known by multiple members at the DMC. This redundancy and shared responsibility reduces the turnaround time from receipt of forms to generation of data-editing reports sent back to the clinics. As patients are enrolled in the registry, the quality of data on key measures is evaluated and, when necessary, measures are refined or clarified in the DMC Manual of Operations. A protocol was written that stated the goals of the registry, the inclusion/exclusion enrollment criteria, the process of data collection, and the precautions to ensure patient protection and the security of the data. An Informed Consent Form (ICF) and an Explanatory Brochure for patients were written. Institutional review board (IRB) Approval of the Protocol and the ICF was obtained from the institutional review boards of the centers, and from the Human Research Protection Office (HRPO), Department of Defense (DOD). Two workshops were held for the investigators, nurse clinicians, physiatrists, and coordinators at which they received training in the ASIA examination and in the data collection methodologies. The registry has enrolled 414 patients with ASIA grades A to E, from October 2005 to October 2010. A Phase 1 Study of the Safety and Pharmacokinetics of Riluzole in Patients with Traumatic Acute Spinal Cord Injury was undertaken to investigate its therapeutic effect in SCI. Riluzole is a neuroprotective drug that blocks glutamate-mediated sodium and calcium ion entry into neurons and glia and blocks glutamate release from presynaptic terminals. It has been shown to be effective in limiting traumatic damage to the spinal cord in laboratory studies.13 Riluzole presented several advantages as a therapy in NACTN’s first trial, in which the working of the network was being tested. Riluzole is used clinically in the treatment of amyotrophic lateral sclerosis (ALS).14 It is relatively inexpensive, it can be given orally, it has a favorable safety profile in ALS patients, it can be measured in blood and cerebrospinal fluid, and costs are reasonable for laboratory studies that are necessary for monitoring its use. The initial phase of the study is a multisite, single-arm active treatment pilot study involving 36 subjects. If the rate of adverse effects in the initial trial group is no greater than that in the NACTN database, a phase 2 study of a larger number of patients will be undertaken as a comparative efficacy trial. Features of the riluzole protocol are the detail in which possible adverse effects of the therapy are being studied, and the use of pharmacokinetic and pharmacodynamic data and its correlation with adverse effects and efficacy. Previous studies of drug therapy for SCI have not measured blood and cerebrospinal fluid levels of the therapeutic drugs to see if effective or toxic levels of drug were reaching the spinal cord and brain. Multiple steps were involved in planning and in implementing the riluzole trial, which enrolled its first patient on April 12, 2010. 1. Selection of riluzole after discussion by the investigators of candidate therapies 2. Writing the research protocol 3. Creation of a schedule of events, a day-by-day hourly schedule of tests, procedures, laboratory work, rules for drug administration 4. Modifying elements of the registry case report forms for the requirements of the trial 5. Designing case report forms (CRFs) 6. Writing a Manual of Operations and patient brochure 7. Writing the Informed Consent Form 8. Setting up the study database at the DMC 9. Developing new methods of measuring riluzole in plasma and cerebrospinal fluid 1. Approval of the research protocol by the HRPO, DOD 2. Harmonization of the IRB requirements of each center with requirements of the HRPO; final approval of harmonized protocol and ICF by each IRB 3. Appointment of a central trial monitor, a distinguished physiatrist at a university not affiliated with any of the centers. For a phase 2 study, a data monitoring and safety committee will be established. 4. Appointment of a local trial monitor at each clinical center 5. Trial initiation meeting of all investigators and coordinators; intensive 2 day review of the protocol, the schedule of events, the rules and procedures for reporting adverse events, interim analysis, and stopping rules 6. Writing and signing of site agreements between the trial sponsor and the network centers. The site agreements are a contract setting forth the statement of work, the responsibilities of the center, and deadlines for completion of work. 1. Creation of patient data binders 2. Creation of regulatory binders 3. At the Coordinating Center, files of all regulatory documents and files of adverse events An individual trained in monitoring clinical trials who has been conducting on-site monitoring at each clinical center and reviewing CRFs, regulatory documents, source documentation, adherence to protocol, and drug accountability in accordance with federal regulations and Good Clinical Practices (GCPs) 1. Writing a Governance Manual 2. Forming committees: Executive; Selection of Therapies; Publications; Data Management 3. Forming Neurological Outcomes Assessment Task Force, led by Dr. Susan Harkema, to develop improved quantitative outcome measures 4. Conducting monthly conference calls by the investigators and coordinators, the committees, and the Neurological Outcome Assessment (NOA) Task Force 5. Posting of NACTN documents and communications on an FTP Web site Pearls
North American Clinical Trials Network: Building a Clinical Trials Network for Spinal Cord Injury
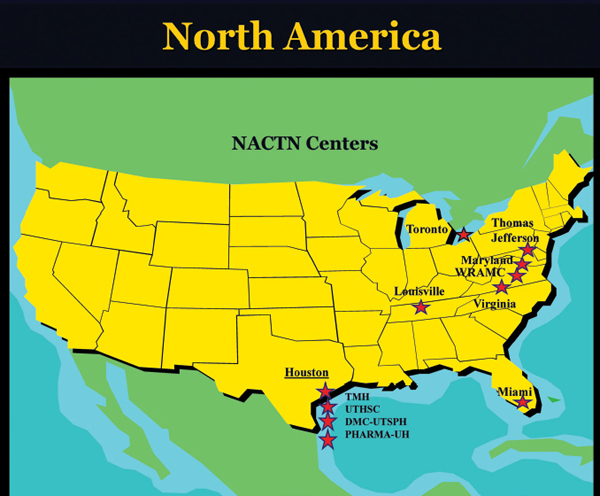
 NACTN Centers and Investigators
NACTN Centers and Investigators
Clinical Centers (Fig. 29.1)
 The Methodist Hospital, Houston–Coordinating Center?
The Methodist Hospital, Houston–Coordinating Center?
 Principal Investigator, Robert G. Grossman, MD?
Principal Investigator, Robert G. Grossman, MD?
 Clinical Trials Manager, Elizabeth Toups, MS, RN, CCRP
Clinical Trials Manager, Elizabeth Toups, MS, RN, CCRP
 The University of Texas–Memorial Hermann Hospital, Houston
The University of Texas–Memorial Hermann Hospital, Houston
 Investigator, Michele Johnson, MD
Investigator, Michele Johnson, MD
 The University of Virginia Hospital, Charlottesville
The University of Virginia Hospital, Charlottesville
 Investigators, Christopher I. Shaffrey, MD, John Jane, Sr., MD, PhD
Investigators, Christopher I. Shaffrey, MD, John Jane, Sr., MD, PhD
 The University of Toronto, Toronto
The University of Toronto, Toronto
 Investigators, Michael Fehlings, MD, PhD, Charles Tator, MD, PhD
Investigators, Michael Fehlings, MD, PhD, Charles Tator, MD, PhD
 The University of Louisville, Louisville
The University of Louisville, Louisville
 Investigators, Susan Harkema, PhD, Jonathan Hodes, MD
Investigators, Susan Harkema, PhD, Jonathan Hodes, MD
 University of Maryland, Baltimore
University of Maryland, Baltimore
 Investigator, Bizhan Aarabi, MD
Investigator, Bizhan Aarabi, MD
 Walter Reed Army Medical Center
Walter Reed Army Medical Center
 Investigator, Michael Rosner, MD
Investigator, Michael Rosner, MD
 University of Miami, Miami
University of Miami, Miami
 Investigator, James Guest, MD, PhD
Investigator, James Guest, MD, PhD
 Thomas Jefferson University, Philadelphia
Thomas Jefferson University, Philadelphia
 Investigator, James Harrop, MD
Investigator, James Harrop, MD
Data Management Center
 The University of Texas School of Public Health, Houston
The University of Texas School of Public Health, Houston
 Investigators, Ralph Frankowski, PhD, Keith Burau, PhD
Investigators, Ralph Frankowski, PhD, Keith Burau, PhD
Pharmacological Center
 University of Houston, College of Pharmacy, Houston
University of Houston, College of Pharmacy, Houston
 Investigator, Diana Chow, PhD
Investigator, Diana Chow, PhD
 Background
Background
 Stages in the Development of NACTN
Stages in the Development of NACTN
 NACTN has developed in three stages:
NACTN has developed in three stages:
Stage 1: Recruitment of Network Components
 Clinical centers were sought that met the following criteria:
Clinical centers were sought that met the following criteria:
 A Data Management and Statistical Center (DMC) was sought that had experience with large clinical trials, experimental design, and familiarity with medical and governmental regulatory processes.
A Data Management and Statistical Center (DMC) was sought that had experience with large clinical trials, experimental design, and familiarity with medical and governmental regulatory processes.
 A Pharmacological Center was sought that had experience in pharmacokinetic and pharmacodynamic measurements of medications, including metabolism in animal and human studies.
A Pharmacological Center was sought that had experience in pharmacokinetic and pharmacodynamic measurements of medications, including metabolism in animal and human studies.
Stage 2: Steps in the Creation of the Data Registry
Acute Hospitalization Forms
 Contact Sheet: Hospital Name, ID Number, Patient and Family Contact
Contact Sheet: Hospital Name, ID Number, Patient and Family Contact
 Page 1 Demographic Data; Medical History
Page 1 Demographic Data; Medical History
 Page 2 Circumstances of Injury, Evacuation Details
Page 2 Circumstances of Injury, Evacuation Details
 Page 3 Initial Clinical Status; Glasgow Coma Scale (GCS) score; Abbreviated Injury Score (AIS)
Page 3 Initial Clinical Status; Glasgow Coma Scale (GCS) score; Abbreviated Injury Score (AIS)
 Page 4A,B ASIA (American Spinal Injury Association) Motor, Sensory Scales at < 72 h and 2 wk
Page 4A,B ASIA (American Spinal Injury Association) Motor, Sensory Scales at < 72 h and 2 wk
 Page 5 Type of Neurological Injury; Type of Bony Injury
Page 5 Type of Neurological Injury; Type of Bony Injury
 Page 6 Imaging Cord and Canal Diameters: CT; MRI; CT/Myelogram
Page 6 Imaging Cord and Canal Diameters: CT; MRI; CT/Myelogram
 Page 7A,B Nonoperative Treatment: Medical; Traction–Reduction
Page 7A,B Nonoperative Treatment: Medical; Traction–Reduction
 Page 8A,B Posterior Surgical Treatment: Procedural Details; Levels
Page 8A,B Posterior Surgical Treatment: Procedural Details; Levels
 Page 9A,B Anterior Surgical Treatment: Procedural Details; Levels
Page 9A,B Anterior Surgical Treatment: Procedural Details; Levels
 Page 10A–D Complications: Cardiac; Pulmonary; Hematological; GI/GU; Infection; Skin; Psychiatric
Page 10A–D Complications: Cardiac; Pulmonary; Hematological; GI/GU; Infection; Skin; Psychiatric
 Page 11 Acute Hospitalization Outcome Summary
Page 11 Acute Hospitalization Outcome Summary
Rehabilitation and Long-Term Follow-Up Forms
 ASIA Scale (American Spinal Injury Association) at 3, 6, and 12 months after injury
ASIA Scale (American Spinal Injury Association) at 3, 6, and 12 months after injury
 FIM (Functional Independence Measure) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
FIM (Functional Independence Measure) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
 SCIM (Spinal Cord Independence Measure) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
SCIM (Spinal Cord Independence Measure) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
 WISCI-II (Walking Index for Spinal Cord Injury) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
WISCI-II (Walking Index for Spinal Cord Injury) at Acute Discharge, Rehabilitation Discharge, 3, 6, 12 months
Data Quality Control: The NACTN Manual of Operations
Data Form Design and Transmission of Data from the Centers to the Data Management and Statistical Center
Steps in the Initiation of the Data Registry
Protocol Development
An Informed Consent Form and an Explanatory Brochure
Institutional Review Board Approval of the Protocol and the Informed Consent Form
Training in Data Collection
Data in the Registry
Stage 3: NACTN’s Initial Clinical Trial
Research Design and Methods
Selection of a Therapy and Development of the Protocol
Trial Initiation: Compliance with Regulatory Requirements
Trial Conduct at Each Center
Appointment of a Site Monitor
Ensuring Communication between Network Centers
Acknowledgments
 Dr. Branko Kopjar, Associate Professor, Department of Health Services, University of Washington, contributed to the development of the riluzole protocol.
Dr. Branko Kopjar, Associate Professor, Department of Health Services, University of Washington, contributed to the development of the riluzole protocol.
 The study sponsor was the Christopher Reeve Foundation, Short Hills, New Jersey.
The study sponsor was the Christopher Reeve Foundation, Short Hills, New Jersey.
 Support was provided by the Telemedicine and Advanced Technology Research Center (TATRC) US Army Medical Research and Materiel Command.
Support was provided by the Telemedicine and Advanced Technology Research Center (TATRC) US Army Medical Research and Materiel Command.
 Additional support was provided by Mission Connect, a project of the TIRR Foundation.
Additional support was provided by Mission Connect, a project of the TIRR Foundation.
 Clinical trial organization and implementation require a fusion of many disparate elements in our complex, nonintegrated medical system.
Clinical trial organization and implementation require a fusion of many disparate elements in our complex, nonintegrated medical system.
 The most important elements in building a clinical trials network is the dedication of the investigators and the clinical and scientific staff and the willing participation of patients, to whom we owe our gratitude.
The most important elements in building a clinical trials network is the dedication of the investigators and the clinical and scientific staff and the willing participation of patients, to whom we owe our gratitude.
 Current thinking is that a trial of a new therapy for SCI will require a cohort of 200 to 250 patients to achieve statistical significance. Depending on the therapy, the cost of such a study can be in the millions of dollars. An urgent question is how to apportion these costs between governmental and private funding agencies, pharmaceutical companies, voluntary health organizations, not-for-profit hospitals, philanthropy, and health insurers. NACTN has combined support from multiple sources to develop a clinical trial network. We believe that a parallel effort to develop a consortium of funding sources is required to support clinical trials of new therapy to improve the outcome of SCI.
Current thinking is that a trial of a new therapy for SCI will require a cohort of 200 to 250 patients to achieve statistical significance. Depending on the therapy, the cost of such a study can be in the millions of dollars. An urgent question is how to apportion these costs between governmental and private funding agencies, pharmaceutical companies, voluntary health organizations, not-for-profit hospitals, philanthropy, and health insurers. NACTN has combined support from multiple sources to develop a clinical trial network. We believe that a parallel effort to develop a consortium of funding sources is required to support clinical trials of new therapy to improve the outcome of SCI.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


