Legg-Calvé-Perthes Disease
Harry Kim
Key Points
• A permanent deformity of the femoral head is the most important sequela of Perthes disease.
• Biological and mechanical factors contribute to the pathogenesis of the femoral head deformity.
• Treatment should be guided by age at onset of the disease and early prognostic factors.
Introduction
Legg-Calvé-Perthes disease (LCPD) is a complex pediatric hip disorder that affects children over a wide age range and produces variable outcomes even with treatment. Since the original reports of the disease in 1910 by Legg from the United States, Calvé from France, and Perthes from Germany, controversies regarding its causes, pathogenesis, natural history, and treatment have persisted. Various theories on its origin have been proposed, but none has yet to be conclusively substantiated. With limited understanding of the disease process, it is difficult at this time to reconcile various clinical features of the disease with one etiologic factor. Recent studies on the causes of Legg-Calvé-Perthes disease have described type II collagen mutation as a potential cause of the disease in a limited number of inherited, bilateral cases in Asian families. Although the cause of disruption of the blood supply to the femoral head remains unknown, further insight into the pathogenesis of a femoral head deformity has been gained through experimental studies using animal models of ischemic osteonecrosis. These studies reveal that mechanical and biological factors contribute to the pathogenesis of the femoral head deformity following ischemic necrosis. Long-term studies suggest that although femoral head deformity is relatively well tolerated over short and intermediate terms, 50% of patients develop disabling arthritis in the sixth decade of life. Thus the overall goal of treatment should focus on preventing or minimizing the femoral head deformity. Along with recent large retrospective studies, two multicenter prospective cohort studies provide some guidance on treating this condition in patients of different age groups.
Epidemiology and Risk Factors
The annual incidence of Perthes disease shows regional variability. Overall, the lowest incidences have been reported from Asian countries, with 1 case per 450,000 in South China and from 0.4 to 14.4 per 100,000 in South India.1 In North America, an incidence of 5.1 per 100,000 was found in British Columbia and 5.7 per 100,000 in Massachusetts.2,3 The cumulative incidence of Perthes disease appearing in children up to 15 years of age was 1 in 740 males and 1 in 3700 females in Massachusetts.3 In Europe, reported incidences have ranged from 7.9 per 100,000 in Trent, England, to 16.9 per 100,000 in Liverpool. In contrast to these data from Liverpool obtained in the period from 1976 to 1981, subsequent study has shown a decrease in incidence to 8.7 in 1990 to 1995, indicating a significant decline in incidence within the region.4 Improvements in socioeconomic status and nutritional deprivation have been proposed as factors associated with the decline in incidence. In other European countries, rates of 9.2 per 100,000 in Norway and 8.5 per 100,000 in Sweden have been reported.1 A comparison of incidence from one region to another requires caution because some studies were performed more recently and others were performed more than 30 years ago. The older studies may or may not truly reflect the current incidence of the disease in the region because the demographics may have changed over time.
Perthes disease can affect children younger than 15 years, but it is most commonly seen in children between the ages of 4 and 9 years. The male-to-female ratio ranges from 3.3 : 1 in Norway to 5 : 1 in Massachusetts.1,3 Bilateral disease occurs in about 10% to 15% of patients, and the presentation tends to be asynchronous in timing. In most studies, the reported incidence of a positive family history is less than 5%.5,6
Studies have reported an increased association between Perthes disease and congenital abnormalities (odds ratio = 2.0), including congenital hip dislocation, Down syndrome, undescended testicle, clubfoot, hypospadias, atrial septal defect (ASD), ventricular septal defect (VSD), and anomalies of skull and facial bones or upper extremities. Studies have also reported an increased association between very low birth weight or short body length at birth and Perthes disease, suggesting that genetic or early developmental factors may be linked to the disease. An increased association has also been reported with maternal smoking and second-hand smoke exposure.7–9
Since the original description of the condition was put forth in 1910, numerous theories on the origin of Perthes disease have been proposed (Box 41-1). A conceptual challenge has been to explain various clinical features, such as unilateral presentation in most patients, predilection for boys, delayed bone age, patient hyperactivity, and association with congenital anomalies in some patients, using a single etiologic theory. The prevailing view at this time is that Perthes disease is a multifactorial disease that is caused by a combination of genetic and environmental factors. It is postulated that genetic factors impart “susceptibility” to the disruption of blood supply to the femoral head, and environmental factors, such as repeated subclinical trauma or mechanical overloading, trigger the disease.
Of proposed etiologic factors, the hypothesis postulating alteration of the insulin-like growth factor 1 (IGF-1) pathway as a cause of Perthes disease merits attention because IGF-1 is known to be expressed in many tissues, including brain and skeleton, and affects postnatal development of these tissues. Because IGF-1 pathway dysfunction can influence brain and skeletal development, it can potentially explain the delayed skeletal maturity, hyperactive behavior, and minor congenital abnormalities seen in patients with Perthes disease. Low levels of serum IGF-1 have been reported in patients during the first 2 years after the diagnosis of Perthes disease in a cohort consisting of 59 consecutive patients.10 In one study, the serum level of IGF-1 was reported to be normal and the level of its major binding protein, IGF-1 binding protein 3, was decreased.11 These results conflict with those of another study, which reported normal IGF-1 binding protein levels.12 Thus, the significance of the IGF-1 pathway in the origin of Perthes disease remains unclear.
Recently, advances in our understanding of the origin of Perthes disease have come from genetic studies of Asian families with inherited bilateral avascular necrosis of the femoral head. Taiwanese, Japanese, and Chinese families with multiple members affected by femoral head osteonecrosis in an autosomal dominant fashion were found to have a missense mutation in the type II collagen gene (replacement of glycine with serine at codon 1170 of COL2A1).13–15 What is noteworthy is that in contrast to skeletal dysplasias and other type II collagenopathies, affected individuals with this mutation in general did not appear to have skeletal abnormalities outside the hips. In affected individuals with open growth plates, radiographic changes typical of Perthes disease were observed14,15 (Fig. 41-1). Although secondary structural consequences due to the mutation appeared minor in nature, it is speculated that the mutation may cause weakening of the cartilage matrix.15 Given that the femoral head is a major weight-bearing structure, it is proposed that cartilage matrix weakening may compromise blood vessels within the cartilage as they traverse from the femoral neck to the bony epiphysis through the cartilage. Although these findings provide new insight into the pathophysiology of femoral head osteonecrosis, the type II collagen mutation may account for only a small number of patients with LCPD because it has yet to be reported in sporadic unilateral or nonfamilial bilateral cases of LCPD.16 It also is not completely clear at this time whether this small number of cases in fact represents an ischemic condition, or whether it represents a subtle form of skeletal dysplasia.
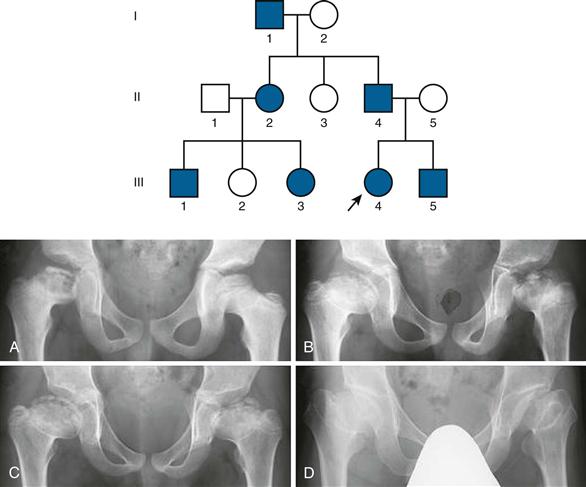
Figure 41-1 Pedigree of a Japanese family with Legg-Calvé-Perthes disease, and radiographs of subject II-4 at (A) age 7, (B) age 9, (C) age 11, and (D) age 45. (From Miyamoto Y, Matsuda T, Kitoh H, et al: A recurrent mutation in type II collagen gene causes Legg-Calvé-Perthes disease in a Japanese family. Hum Genet 121:625–629, 2007.)
Thrombophilia leading to thrombotic venous occlusion of the femoral head has been proposed as a cause of Perthes disease, but this theory remains controversial. In a case control study that reported on the association between Perthes disease and coagulation abnormalities, Glueck and associates found that 75% of the patients in the study had a coagulation abnormality.17 Although some studies have reported an increased rate of coagulation abnormalities in patients with Perthes disease, others have not found any association at all. A prospective study of a random series of 50 consecutive patients with Perthes disease also did not find a difference in the prevalence of protein C, protein S, or antithrombin III deficiencies, or factor V Leiden mutation, between the study group and the estimated population frequency.18 The authors of the study proposed that the high prevalence of coagulation abnormalities found in the study by Glueck and colleagues may be attributed to the less stringent reference range used to define the coagulation abnormalities. A recent case control study that included some of the investigators from the original study by Glueck and coworkers also found no increase in the prevalences of protein C, protein S, and antithrombin III deficiencies, hyperhomocysteinemia, and elevated plasminogen activator inhibitor-1 activity between 72 nonselected, consecutive series of patients with Perthes disease and 197 healthy controls.19 However, this study did find a higher prevalence of factor V Leiden (8 of 72 in the study group compared with 7 of 197 in the control group) and anticardiolipin antibodies (19 of 72 in the study group compared with 22 of 197 in the control group) among patients with Perthes disease. Because thrombotic events are uncommon during childhood, even in those patients with inherited thrombophilia,20 the significance of inherited or acquired thrombophilia in the pathogenesis of Perthes disease remains unclear at this time.
Pathophysiology
Although the origin of Perthes disease remains unknown, clinical and experimental evidence supports the hypothesis that disruption of the blood supply to the femoral head is a key pathogenic event associated with the disease process. Selective angiography,21–23 bone scintigraphy,24 perfusion magnetic resonance imaging (MRI),25 and biopsy studies26 from early stages of the disease show evidence of disruption of perfusion and tissue damage consistent with ischemic necrosis. Furthermore, disruption of the blood supply to the femoral head in large animal models produced the histopathologic and radiographic changes observed in Perthes disease, including a fragmented appearance of the bony epiphysis and the coxa plana.27
It is debatable whether a single episode or multiple episodes of ischemia are necessary to produce Perthes disease. The second or multiple infarction theory is based on observations from an immature canine model in which a single surgical attempt at inducing femoral head infarction did not produce the femoral head deformity and histologic features of Perthes disease.28,29 These findings were observed only after a second infarction surgery in some animals, leading to speculation that Perthes disease may be due to more than one episode of infarction.28 In contrast, a single infarction surgery produced a femoral head deformity and histologic changes resembling Perthes disease in a piglet model of ischemic necrosis.27 The finding of thickened trabeculae with multiple cement lines from the specimens of patients with Perthes disease has been proposed as supportive evidence for the multiple infarction theory.30 An alternative interpretation of the finding, however, is that a single episode of ischemia produces the disease, but subsequent reinjury or injuries of the revascularized region occur as the result of repeated mechanical overloading and/or further collapse of the femoral head during a vascular repair phase. One implication of the latter theory is that femoral head overloading and deformation during the healing phase may hinder revascularization of the necrotic femoral head and may prolong the healing process.
A report describing histopathologic findings from six whole femoral heads constitutes the largest assembly of whole head samples of Perthes disease studied to date.31 The small number underscores the major obstacle in attempts to understand the pathophysiology of a condition with very limited availability of tissue samples for research investigation. Findings from this study, a few isolated necropsy reports, and some studies based on surgical biopsy specimens26,30-37 are all that are available at this time to enhance our understanding of the disease process. From these studies, it can be summarized that pathologic processes in Perthes disease affect the articular cartilage (also called the epiphyseal cartilage), bony epiphysis, physis, and metaphysis.
Articular cartilage changes are found mainly in the middle and deep layers of the cartilage. Necrosis in the deep layer of the cartilage, cessation of endochondral ossification, separation of cartilage from underlying subchondral bone, vascular invasion of the cartilage, and new accessory ossification are the observed changes (Fig. 41-2). In the bony epiphysis, necrosis of the marrow space and of trabecular bone, compression fracture of trabeculae, osteoclastic resorption, fibrovascular granulation tissue invasion of the necrotic head, and thickened trabeculae have been reported. Physeal changes are seen most often in the anterior part of the femoral head, with focal areas of growth cartilage columns extending below the endochondral ossification line. Premature growth arrest of the growth plate is seen in only 30% of patients with Perthes disease, suggesting that in most patients, the growth plate continues to function. Findings of a recent experimental study are consistent with clinical findings indicating that in most patients with Perthes disease, the growth plate remains functional.38 Metaphyseal changes are commonly seen during the early stages of Perthes disease and usually are found subjacent to or below the growth plate in the anterior aspect of the femoral head. The mechanisms responsible for the appearance of these lesions are unclear. Various tissue types have been reported, including columns of normal or degenerated cartilage extending down to the metaphysis, fibrocartilage, fat necrosis, vascular proliferation, and focal fibrosis.31 Some have found an association between the presence of radiolucent metaphyseal changes and poor prognosis, but others have not.
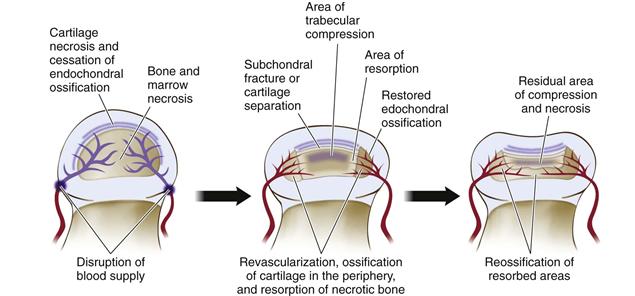
Figure 41-2 An illustration combining the described histopathologic changes from the necropsy and biopsy studies of Legg-Calvé-Perthes disease. Important pathologic processes include cartilage and bone necrosis, subchondral fracture and compaction of necrotic bone, revascularization from the periphery of the epiphysis with associated resorption of necrotic bone and further collapse, and asymmetrical restoration of endochondral ossification at the periphery, further contributing to the deformed, ovoid shape.
The lack of availability of clinical samples for research has prompted alternative approaches, such as the use of animal models, to investigate the pathogenesis of Perthes disease. In particular, a piglet model of ischemic osteonecrosis has allowed more systematic and in-depth investigation of ischemic tissue damage, the repair process, and the pathogenesis of femoral head deformity following disruption of the epiphyseal blood supply. Key findings from this line of investigation are that the induction of ischemia produces a decrease in the mechanical stiffness of the necrotic femoral head, making it relatively soft in comparison with the normal femoral head, from the early avascular necrotic phase to the latter vascular repair phase of the model.39 The mechanical properties of the two components of the femoral head—articular cartilage and trabecular bone from the bony epiphysis—were found to be decreased.40 The mechanical compromise observed in the avascular necrotic phase may be due to necrosis of the deep layer of the articular cartilage,41 changes in the material properties of calcified cartilage and trabecular bone in the infarcted head,42 and possible accumulation of microfractures in the necrotic bone. Repetitive loading is known to produce microfractures or microcracks in the bone, which are detected and repaired by bone cells.43 However, in necrotic bone, no cells are available to detect and repair the microdamage related to repetitive “wear-and-tear.” Furthermore, it is proposed that a significant increase in the mineral content of necrotic trabecular bone makes it more brittle and prone to microdamage.42 As a result, a subchondral fracture (crescent sign) or a compaction fracture may occur in the necrotic femoral head with repetitive overloading of the hip joint.
Vascular invasion and subsequent resorption of necrotic bone further compromise the mechanical properties of the infarcted head in the vascular repair phase.39 The mismatch of bone resorption and formation produces a net bone loss, seen radiographically as a radiolucent area within the necrotic bone.27 The predominance of bone resorption during the early vascular phase of the repair weakens the femoral head, predisposing it to flattening. It is postulated that the weakened femoral head begins to deform when its ability to resist deformation falls below a critical level surpassed by hip joint loading (Fig. 41-3). Inhibition of bone resorption using antiresorptive agents, such as bisphosphonates and receptor activator of nuclear factor (NF)κB ligand (RANKL) inhibitor, has been shown to decrease the deformity in animal studies, indicating that the resorptive process is an important component of the pathogenesis of femoral head deformity in these models.44,45 Clinical studies are required to validate the significance of bone resorption and the efficacy of antiresorptive agents in decreasing femoral head deformity in Perthes disease. Further discussion on the development of medical treatment for Perthes disease is found in the treatment section.
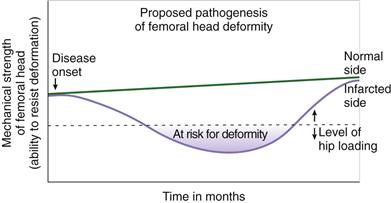
Figure 41-3 Line drawing depicting the proposed pathogenesis of the femoral head deformity following ischemic necrosis based on piglet mechanical studies. With the onset of ischemic osteonecrosis, the mechanical properties of the femoral head decrease over time owing to factors described in the text. It is proposed that when hip joint loading surpasses the mechanical strength of the femoral head, the deformity is initiated and progresses.
Because the hip joint is a major load-bearing joint, it is important to consider the development of the femoral head deformity in the context of loading of the joint. This is one of the areas of Perthes disease research that has a paucity of data. The relationship between the magnitude and frequency of hip joint loading and the development of the femoral head deformity remains unclear. Basic data such as hip contact pressures associated with various activities of daily living are not available in children. In adults, a sophisticated femoral head prosthesis equipped with a strain gauge and telemetric data transmission capability has allowed real-time collection of these valuable data following total hip replacement, with patients performing various activities and positioning of the leg.46 The measurements indicate that significant forces act on the femoral head with daily activities. Walking was associated with hip contact pressure reaching about 2.5 times the body weight. Running on a treadmill at a rate of 8 kilometers per hour increased the contact pressure to about 4.5 times the body weight. Some supine and prone activities were also associated with hip contact pressures elevated above the body weight. These measurements have provided insight into the magnitude of loading associated with various activities and leg positioning. A complete loading history and magnitude for an individual will likely depend on the list of activities the person performs, the frequency of each activity, and the body weight. In a disease where femoral head deformity is produced as the result of mechanical weakening, avoidance of activities that generate a significant increase in hip contact pressure would seem reasonable. At this time it is unknown what “significant” loading is, and what effect restricting activities has on preventing the deformity.
In contrast to femoral head weakening and hip joint loading, which promote the development of femoral head deformity, the healing or remodeling potential related to age at onset of the disease appears to offset the deformity. Clinical studies consistently show a better outcome in terms of femoral head shape in patients with early onset of Perthes disease.47 It is unclear what factors determine the healing and remodeling potentials. Important biological factors to keep in mind are that Perthes disease affects a wide age range of children from preschool years to early teenage years, and that the age range represents a growth period when significant changes are taking place in terms of femoral head anatomy, size, and vasculature48–51 (Fig. 41-4). The size of the bony epiphysis increases while the thickness of the articular cartilage and the growth potential of the bony epiphysis decrease with age. In addition, more subtle changes, such as regression of cartilage vascularity (presence of cartilage vascular canals) and changes in the vascular anatomy of the proximal femur, are taking place. Given these changes, onset of the disease at different ages implies that the disease is affecting a femoral head that may have significantly different growth and remodeling potentials. These factors are likely to affect the final outcome of the femoral head.
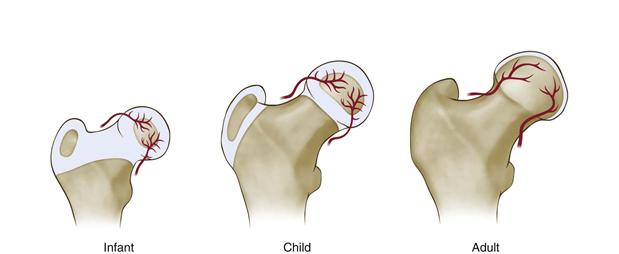
Figure 41-4 A drawing showing significant developmental changes occurring in the proximal femur from infancy to maturity. Notable changes included decreased cartilage thickness and cartilage vascularity, increased size of the bony epiphysis, and regression of the growth plate. It is postulated that the onset of ischemic necrosis at different stages of femoral head development will have a significant implication in terms of the healing and remodeling potentials of the femoral head owing to these changes.
Clinical Features and Diagnosis
Patients with Perthes disease generally present with pain, a mild limp, and limited hip motion (Box 41-2). In general, the pain is mild and does not restrict the child from daily activities. Often, the pain is not localized to the hip joint, misleading the unsuspecting physician to obtain radiographs or an MRI of the knee. In a study of 425 patients with Perthes disease, 50% of children had pain in the hip and thigh, 18% in the thigh and knee, 14% in the knee only, 8% in the hip, thigh, and knee, and 1% in other areas; 9% had no pain.1 It is important to recognize that what is perceived as thigh or knee pain can be due to hip disease in a child. Occasionally, a patient may become symptomatic after a minor trauma or a fall during a sports activity.
Physical examination often reveals a mild limp at the time of presentation. The limitation of hip range of motion depends on the stage of the disease. Hip motion may be good in the early stage, before the development of fragmentation or resorptive changes in the epiphysis. In those patients with marked synovitis, however, the motion may become restricted early. Gentle rotation of the affected hip with the leg extended often demonstrates restriction to the rolling maneuver, so-called irritability of the hip. During the stage of fragmentation, hip motion can decrease further and become severely limited. Hip abduction and internal rotation are the earliest motions to become restricted. When assessing passive hip abduction, it is important to stabilize or feel the pelvis to confirm that the leg abduction is coming from the hip joint, not from the lateral rotation of the pelvis (Fig. 41-5). Hip motion improves during the stage of reossification unless significant flattening or deformity of the femoral head that mechanically restricts hip abduction and rotation is present. Depending on the duration of the disease, hip adductor contracture, atrophy of thigh and calf muscles, and limb length discrepancy (0.5 to 1.5 cm) are observed in some patients. The presence of hip adduction contracture or complete absence of passive abduction during the fragmentation stage of the disease should alert the examiner to the possibility of hinge abduction.
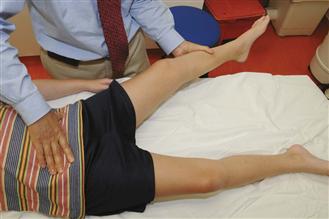
Figure 41-5 To assess hip abduction, a hand should be placed on the pelvis (anterior superior iliac spines) to ensure that the abduction is coming from the hip joint and not from the lateral pelvic rotation.
Imaging Studies
Plain radiography remains the main diagnostic and clinical assessment tool for Perthes disease. Anteroposterior (AP) and frog-leg lateral x-rays of the pelvis are used to determine the radiographic stage of the disease, the extent of head involvement, serial progression of the disease, and the development of head-at-risk signs such as lateral subluxation, metaphyseal reaction, Gage’s sign, lateral calcification, and horizontal alignment of the growth plate52 (Fig. 41-6). Some have found that lateral subluxation has greater prognostic importance than the other signs53; others have found that two or more head-at-risk signs are associated with a poor outcome.54 The Catterall, Salter-Thompson, or lateral pillar classification can be applied to assist with treatment decision making when patients reach the stage of fragmentation. These classification systems are further discussed later in the natural history and treatment sections.
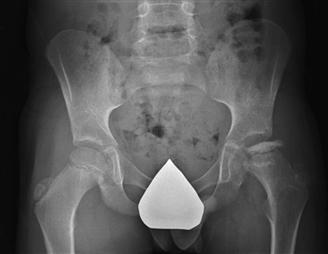
Figure 41-6 A radiograph demonstrating some Catterall’s at-risk signs: lateral subluxation with a break in Shenton’s line, metaphyseal reaction, lateral calcification, and horizontal alignment of the growth plate.
Waldenström (1922) described four radiographic stages during the active phase of the disease: the initial stage or the stage of increased radiodensity, the stage of fragmentation, the stage of reossification, and the healed stage, according to characteristic radiographic features of each stage (Fig. 41-7). The duration of each stage is variable from one patient to another. What determines the duration of each stage and the total duration of the active phase remains unknown. In general, older patients appear to have a longer duration than younger patients. According to one study, fragmentation staging lasts about 1 year and the reossification stage lasts from 3 to 5 years.55 It is during the initial and fragmentation stages that femoral head deformity develops and progresses. During the reossification stage, the femoral head shape can improve, worsen, or remain unchanged. Herring and associates observed that the femoral head was more likely to undergo progressive flattening in older patients, in those with more severe lateral pillar involvement, and in those with prolonged ossification.55 Although plain radiographs are useful in assessing the progression of the disease, they lack the sensitivity and specificity needed to reveal vascular and repair changes occurring within the bony epiphysis.
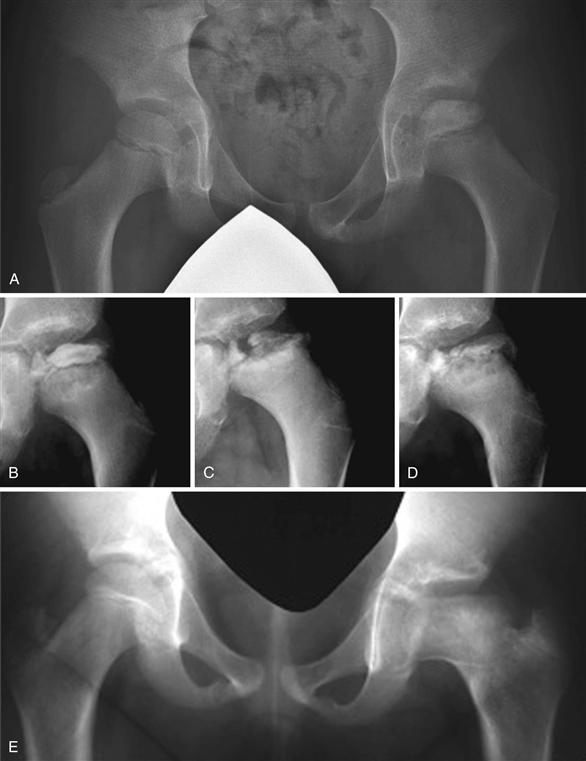
Figure 41-7 Radiographs demonstrating Waldenström’s radiographic stages. A, Initial stage. This stage is also called the stage of increased radiodensity. Characteristic features of this stage include a smaller epiphysis compared with the unaffected side, increased radiodensity, and minimal flattening. A subchondral fracture (crescent sign) may or may not be present. B and C, Fragmentational stage. This stage is also called the resorptive stage. During this stage, resorptive and fragmented changes are seen in the epiphysis. This is the stage when most deformity occurs. D, Stage of reossification. Radiolucent areas in the epiphysis are filled in with new bone. Along with reossification of the epiphysis, remodeling of the epiphysis takes place, making the radiodensity of the epiphysis more homogeneous. E, Healed stage.
Bone scintigraphy can detect changes in bone perfusion in the early stage of Perthes disease, when radiographic changes are not apparent. On the basis of scintigraphic findings, Conway proposed that two distinct processes are involved with revascularization (i.e., recanalization and neovascularization), and that they carry different prognostic significance.24 Bone scintigraphy has been used to assess femoral head revascularization with good correlation with MRI findings.25 However, exposure to ionizing radiation and an inability to provide cross-sectional imaging are significant barriers to wide use of this technique.
The role of MRI in the management of Perthes disease is evolving. Like bone scintigraphy, MRI is a sensitive tool for detecting femoral head avascularity. As the techniques, resolution, and scanning time of MRI continue to improve, gadolinium-enhanced MRI may serve as a useful imaging tool to quantify the extent of femoral head avascularity at early stages of the disease and to quantify and compare the extent of revascularization of the femoral head that occurs over time25 (Fig. 41-8). Because MRI is much more sensitive than plain radiography in visualizing vascular and repair changes that occur in the bony epiphysis, the possibility of using this technique as an early prognostic indicator before the development of deformity to guide treatment decision making requires investigation. Another future application of MRI in assessing Perthes disease consists of exploiting its ability to visualize the outline of the femoral head cartilage to assess the femoral head shape change. A three-dimensional MRI reconstruction technique has been shown to be able to quantify the extent of loss of femoral head sphericity in patients with Perthes disease.56 Whether such a quantitative method is superior to a qualitative radiographic outcome system such as the Stulberg classification remains to be studied.
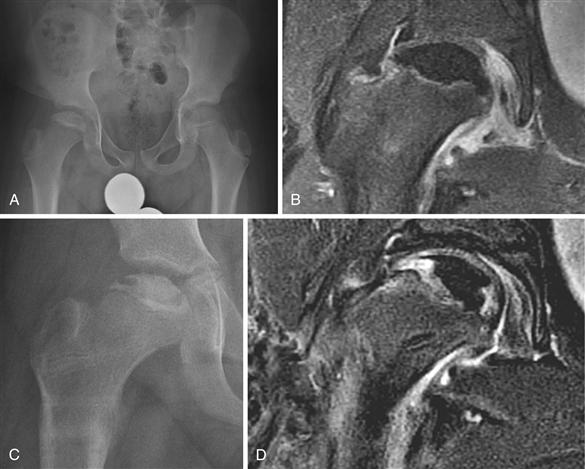
Figure 41-8 An 11-year-old male with right Legg-Calvé-Perthes disease. A, An anteroposterior (AP) pelvis radiograph showing the initial stage of the disease. Based on the radiograph, it is difficult to determine the extent of the femoral head necrosis. B, Dynamic subtraction magnetic resonance imaging (MRI) following an intravenous gadolinium administration showing complete absence of perfusion in the epiphysis, indicating a total disruption of perfusion. C, A radiograph obtained 7 months after a femoral varus osteotomy and subsequent removal of the fixation device showing maintenance of the femoral head shape with the presence of resorptive changes. D, A repeat gadolinium-enhanced MRI showing partial revascularization of the femoral head in the medial and lateral aspects of the epiphysis with the central region remaining avascular. Based on this information, the patient was maintained on non–weight-bearing status on the affected side.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








