CHAPTER 23 Advanced Evaluation and Management of Glenohumeral Arthritis in the Cuff-Deficient Shoulder
The arthritic shoulder with irreparable massive cuff deficiency is one of the most difficult and challenging issues in shoulder practice, due to the combination of severe articular and periarticular soft tissue damage. This entity is the common end-stage result of several disease processes such as rheumatoid arthritis, rotator cuff tear arthropathy, and the Milwaukee shoulder syndrome. Characterized as a substantial defect in the rotator cuff tendons, these disease processes lead to destabilization of the glenohumeral joint with subsequent superior migration of the humeral head and secondary severe damage to both the intra-articular and extra-articular elements. The result is a painful, dysfunctional shoulder that necessitates, in many cases, a surgical solution.
Inadequate stability and insufficient bone stock make the task of replacing the damaged joint with a stable construct very difficult. Neer called this one of the most challenging issues in shoulder arthroplasty1 and, because of the minor functional gain with conventional shoulder arthroplasty, he introduced the term limited-goal operation. In many series, conventional hemiarthroplasty did not achieve good functional results.2,3 In general, only a certain amount of pain relief was achieved. The functional improvement was not very predictable, and whether more or less functional improvement is gained depends mainly on whether the cuff-deficient shoulder is biomechanically stable or unstable. This means that the functional outcome of the joint mainly depends on the amount of biomechanical compensation available from the concavity compression of the remaining rotator cuff.
HISTORY AND BIOMECHANICS OF REVERSE ARTHROPLASTY
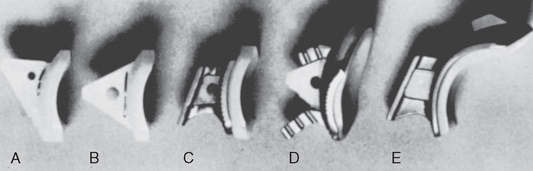
To counteract the superior migration of the humeral head, a superior hood was added to the glenoid component (Fig. 23-1).1–4 This would restore the length of the deltoid muscle and decrease the instability of the humeral head. Initial results were encouraging and pain relief was good, but further follow-up showed a high failure rate, with loosening of the glenoid component. In 2000, Nwakama and colleagues5 reported on seven shoulders treated with a hooded glenoid component. All but one was unsatisfactory at a mean follow-up of 69 months. This design is rarely used today.
Several constrained total shoulders have been tried. Some of these have been reverse designs. The goals were to restore deltoid length and to create a fixed center of rotation. Initial results showed significant improvement in function, but due to high failure rate on the glenoid side, these designs were abandoned.6–10 A common design feature of these early constrained arthroplasties was that the center of rotation was lateralized and outside the glenoid bone (Fig. 23-2). This created a high stress on the fixation of the glenoid component, resulting in aseptic loosening.
However, Prof. Paul Grammont in Dijon continued to work on an arthroplasty that could restore function in the rotator cuff–deficient arthritic shoulder. In the early 1980s, he advocated a medialized center of rotation to improve the biomechanics of the deltoid muscle by restoring length and increasing the lever arm. The Ovoid arthroplasty was tried in 1983 (Fig. 23-3). It had an egg-shaped head on the humeral side. The center of rotation was medialized but, due to the ovoid shape, the deltoid muscle was maintained in the lateral position to improve abduction. Range of motion was improved, but instability was a concern.
Grammont therefore moved to a reverse design with a large hemisphere on the glenoid side to place the center of rotation at the bone–implant interface. His first reverse design was the trumpet prosthesis in 1985 (Fig. 23-4). The humeral component was all polyethylene, and the glenoid component was a ball made of metal or ceramic and two thirds of a sphere with a 42-mm diameter. Both components were cemented. Preliminary results of eight cases were published 1987.11 Four of these were revisions of failed anatomic arthroplasties (mean follow-up, 6 months). Results varied, but in three patients the elevation was more than 100 degrees.
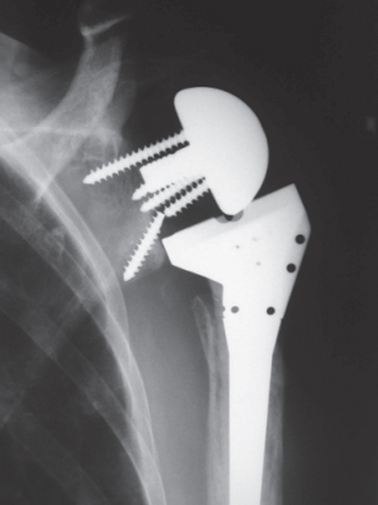
The implant was redesigned, and Grammont decided to use uncemented fixation of the glenoid component and to have half a sphere instead of two thirds of a sphere on the glenoid side. The first version of this first Delta reverse arthroplasty was used in the late 1980s.12,13 The glenoid component consisted of a metaglene plate that had a central peg and was fixed with four additional screws. A large hemisphere (36 or 42 mm) was then attached to this metal plate. This created a fixed center of rotation positioned at the bone–implant interface. The humeral component was uncemented and had a corresponding polyethylene cup with various depths to give the surgeon tools to create a stable joint. It was further redesigned into the Delta 3 arthroplasty (DePuy International, Ltd), available in 1991 (Fig. 23-5).
This implant has been further redesigned into the Delta Xtend (2007). This revised design is based on the proven features of Grammont’s ideas, and new features have been incorporated to reduce the risk of complications such as scapula notching and to facilitate the correct insertion of the implant. This reverse ball-and-socket design by Grammont has two major innovations compared to previous designs.14 It has a large glenoid hemisphere with no neck and a small humeral cup with a nonanatomic inclination angle of 155 degrees, which covers less than half of the glenosphere. This design, with a large hemisphere and no neck on the glenoid side, medializes the center of rotation and places the center of rotation at the surface of the glenoid bone. This reduces the torque forces on the component and decreases the risk of loosening. It provides a fixed center of rotation, preventing superior migration of the humeral head, and restores the length of the deltoid muscle. The large diameter also allows greater range of motion before impingement occurs. Because the center of rotation is medialized, more deltoid muscle fibers are lateral of the center of rotation, allowing more strength in abduction. The nonanatomic inclination of the humeral component places the humeral component in a low position, making it easier to restore the length of the deltoid muscle or to slightly overstretch the muscle.
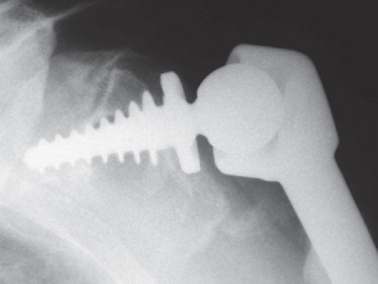
Today there are several reverse ball-and-socket designs that more or less follow Grammont’s basic principles, and published series have showed good outcome. However, external rotation cannot be restored, and impingement inferiorly between the humeral component and scapula, resulting in a notch, is a concern (Fig. 23-6). To reduce notching and to improve the longevity of the implant, new designs are available; based on research and clinical experience, the surgical technique is evolving. It has been shown that a low placement of the glenoid component reduces the risk of mechanical impingement between the humeral component and the scapula.15,16 Glenoid components with polyaxial screws make it easier for the surgeon to place the component low and still have the ability to place the inferior screw into the inferior pillar of scapula.
CLASSIFICATION OF CUFF TEAR ARTHROPATHY
Cuff tear arthropathy (CTA) or rotator cuff–deficient arthritic shoulder (RCDA) is not one unique pathologic entity but consists of different underlying pathologies. It is a common end stage of several disease processes with a unique clinical feature: Painful arthritic shoulder with a massive, irreparable rotator cuff defect. Often this is combined—secondary or primary—with a significant bone erosion on the glenoid and also sometimes at the humeral epiphysis.
The glenohumeral joint lacks significant intrinsic bony stability, thus it relies, to a great extent, on its soft tissue components. The rotator cuff tendons provide a major contribution to the dynamic stabilization of the glenohumeral joint by increasing the concavity–compression force in the joint.17–20 A massive tear, consisting of the supraspinatus and at least one of the other rotator cuff tendons21 (in most cases the infraspinatus) can render the rotator cuff’s anterior and posterior force couple ineffective in both the vertical and the transverse planes. The result is a diminution of joint reaction force and a change in the overall direction of the joint force that leads to the destabilization of the glenohumeral joint.22
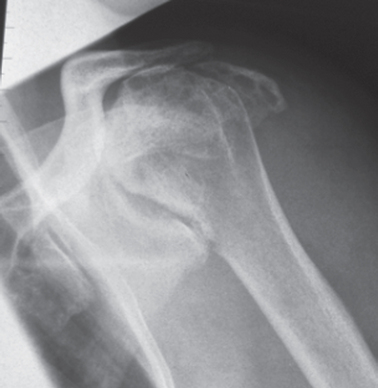
Whether the presence of the long head of the biceps tendon is still contributing resistance against superior translation is still under discussion. However, it is unlikely that a severely damaged tendon could add stability by active tensioning of the muscle–tendon unit.23 Due to an unopposed proximal pull of the deltoid, the humeral head migrates superiorly. The fulcrum of the deltoid’s action evaporates with superior migration of the head and the mechanics of the deltoid deteriorate; therefore, it must generate more force to perform its function.24,25 The humeral head then articulates with the coracoacromial arch and the superior glenoid rim. With longer periods of time, this leads to deformation of the humeral head (femoralization), rounding and thinning of the coracoacromial arch (acetabularization), and, subsequently, to the destruction of the anterior-superior glenoid (Fig. 23-7).26
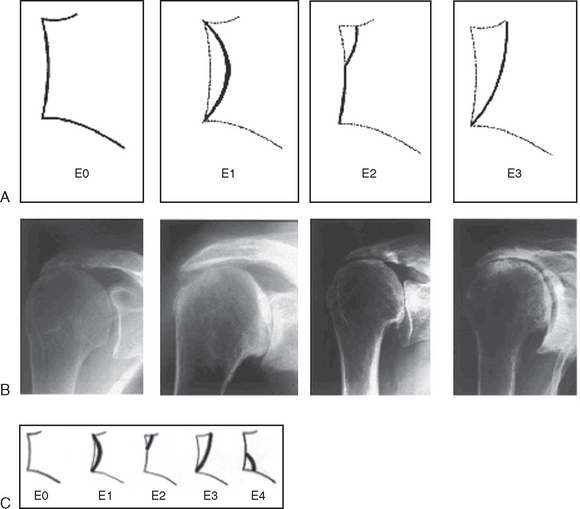
FIGURE 23-7 Favard classification of glenoid defects.
(A and B, From Favard L, Lautmann S, Clement P: Osteoarthritis with massive rotator cuff-tear. In Walch G, Boileau P [eds]: Shoulder Arthroplasty. Berlin: Springer-Verlag, 1999, pp 261-266. C, From Sirveaux F, Favard L, Oudet D, et al: Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Joint Surg Br 86:388-395, 2004.)
The acromioclavicular joint is also commonly involved in the process, joining its cavity with that of the now-joined synovial intra-articular and subacromial bursal spaces. Cases of secondary stress fractures of the thinned acromion have also been published.27,28 The end result is an incongruous, unstable joint with an unstable and superiorly malpositioned center of rotation and secondary glenoid erosion (Fig. 23-8).
Classification of Glenoid Defects
There are different types of classification of glenoid defect. The classification by Antuna, Sperling, and Cofield is more appropriate for glenoid defects seen in revision surgery of failed conventional total shoulder arthroplasty.29 Glenoid defects that arise superiorly due to the superior migration of the head are better described in the classification of Farvard (see Fig. 23-7).30 Because there are not only superior defects but also anterior, superior, and inferior defects, we developed a classification that distinguishes between centric and eccentric defects. In eccentric defects, not only the extension but also the position of the defect is classified (Fig. 23-9 and Box 23-1).
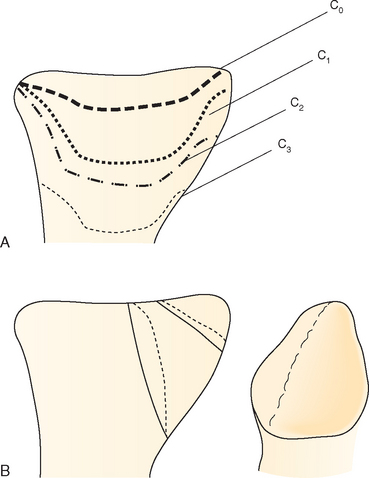
FIGURE 23-9 Authors’ classification of glenoid defects. A, Centric or contained defects. B, Eccentric defects.
Biomechanical Classification of Cuff Tear Arthropathy
The extent of anterior-superior displacement of the humeral head depends on the number and locations of tendons affected,31–33 their degree of involvement, the extent of atrophy of the muscles,34,35 the structural integrity of the coracoacromial arch, and the extent and direction of the accompanying glenoid destruction. The consecutive anterosuperior displacement and instability of the humeral head and the change in the center of rotation cause an insufficiency of the deltoid muscle.25 Biomechanical investigations by Grammont11 and De Wilde36 have shown that a caudal and medial displacement of the glenohumeral center of rotation causes a significant increase in the moment of rotation of the deltoid muscle. Conversely, it could be assumed that the superior and lateral displacement of the center of rotation deteriorates the biomechanics of the deltoid.
The previous classifications of Hamada and Fukuda37 or Favard38 had no therapeutic impact. They purely described the natural course and explained the pathomorphologic consequences of large and massive cuff tears. We intended to establish a more functional and biomechanical classification of cuff tear arthropathies into four types, focusing on the position and stability of the center of rotation on static (plain x-ray) and dynamic (fluoroscopy) radiologic investigation. The major intention was to develop a classification that offers therapeutic guidelines and is highly independent of the underlying etiology.
Pathomechanics
The rotator cuff tendons provide a major contribution to the dynamic stabilization of the glenohumeral joint. By increasing the concavity-compression force in the joint by their synchronous action, they oppose the displacing effect of the strong deltoid muscle, keeping the humeral head centered in the glenoid fossa throughout its movement. The coupled work of the infraspinatus and subscapularis has been shown to be a major factor in superior glenohumeral stability; the contribution of the supraspinatus is less significant.39–41 A massive tear consisting of the supraspinatus and at least one of the other rotator cuff tendons (in most cases the infraspinatus) makes the rotator cuff’s anterior and posterior force couple ineffective in both the vertical and the transverse planes. The result is a diminution of joint reaction force and a destabilization of the glenohumeral joint.42 In cases in which the long head of biceps is still functional, it might oppose, to some extent, the superior migration of the humeral head.23
Nové-Josserand and Walch31 demonstrated in a retrospective analysis of 246 patients with large and massive cuff tears that the additional involvement of the subscapularis leads to a significant decrease of the acromiohumeral distance (an indicator of the superior migration of the center of rotation) in comparison to two-tendon tears of the supra- and infraspinatus that did not involve the subscapularis. The location of the defect, whether it is more a posterior-superior or an anterior-superior large or massive defect, is therefore important for the amount of superior displacement. Posterior-superior defects have a bony buttress provided by the osseous arch of the acromion, and therefore the superior displacement has structural barriers and limits.
Gagey32 described a biomechanically important tight fibrous frame consisting of collagen fiber bundles in the anterior part of the supraspinatus and in the superior part of the subscapularis, the biceps tendon, and the coracohumeral ligament. This fibrous frame acts as a passive restraint against anterior-superior translation; therefore, rotator cuff defects located in the anterior-superior section of the cuff lead to a greater amount of superior translation than posterior-superior defects of the same size. If the proximal pull of the deltoid is left unopposed, the humeral head migrates superiorly toward the coracoacromial arch. The humeral head then articulates with the coracoacromial arch superiorly and the superior glenoid rim inferiorly, leading to flattening of the superior part of the humeral head and tuberosities (femoralization), rounding and thinning of the coracoacromial arch (acetabularization), and destruction of the superior glenoid region (see Fig. 23-8). The occurrence, expression, and presentation of the single morphologic features are multifactorial and mainly depend on the underlying pathology and the pathomechanics of the rotator cuff tear. Otherwise, the pathomechanics of the rotator cuff tear depend on the size and location of the tear, the number of tendons involved, the integrity of the coracoacromial arch, and the bony geometry of the glenoid.
Previous Classifications of Cuff Tear Arthropathy
Hamada–Fukuda Classification
The Hamada–Fukuda classification37 is more or less a morphologic description of the natural course of massive rotator cuff tear and therefore only focuses on the group of (primary) cuff tear arthropathy according to Neer.43 Using the arthrographic confirmation of a massive cuff tear, Neer defined five types:
Favard Classification of Cuff Tear Arthropathy
Favard38 described three different types.
As with the Hamada–Fukuda classification, this one is merely a pathophysiologic and pathogenetic explanation for cuff tear arthropathy but without any therapeutic impact. Type I develops slowly out of old tears (cuff tear arthropathy according to Neer), and some develop into a destructive type of arthropathy. Type II is an osteoarthritic shoulder with secondary massive cuff tear. Type III is seen in rheumatoid shoulders with a concomitant cuff tear prior to bony destruction.
Burkhart Classification
Burkhart40 developed a classification of cuff tears based on their biomechanical behavior. Under dynamic radiologic examination (fluoroscopy), Burkhart found three biomechanically different types of rotator cuff tears; his investigations focused on the position of the fulcrum of the glenohumeral joint. The fulcrum of the glenohumeral joint could be stable, unstable, or captured. He assumed that the pathomechanics of rotator cuff tears are highly dependent on the balance of the force couple of the subscapularis in the front and the infraspinatus in the back as counterparts of the superior displacing effect of the strong deltoid muscle.
Seebauer Classification
The specific problems and features of the single etiologies are extensively described later in this chapter. Besides the specific problems there is a common feature for all pathologies, which is characterized by progressive superior migration and instability of the humeral head. We have developed a more biomechanical, functional, and morphologic classification that focuses on the position and stability of the center of rotation of the glenohumeral joint (Fig. 23-10).44,45
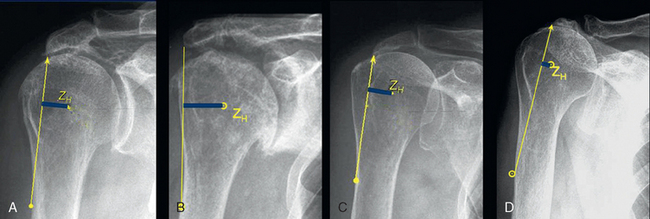
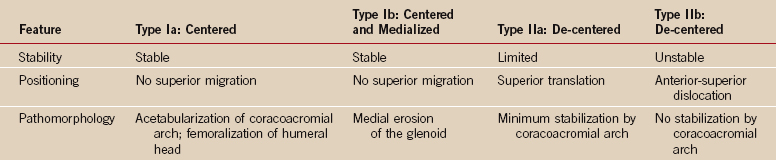
In developing our classification as a tool in decision making for prosthetic therapy of cuff tear arthropathy we aimed specifically for one that is not a simple pathomorphologic description, is biomechanically oriented, describes the static and dynamic amount of anterior-superior instability, considers the position and stability of center of rotation as decisive parameters, is oriented toward therapy for the pathology, is independent of underlying pathology, and that can be used as an additional tool for decision making in prosthetic treatment besides the clinical parameters. Therefore, we established a mainly functional and biomechanical classification of cuff tear arthropathies into four types focusing on the position and stability of the center of rotation on static (plain x-ray) and dynamic (fluoroscopy) radiologic investigations. In types Ia and Ib the center of rotation is not displaced; in types IIa and IIb it is significantly cranially displaced. Type IIb is characterized by a completely static or dynamic anterosuperior instability (Table 23-1).
Sometimes the distinction between type IIa and IIb is difficult on a simple static AP x-ray. To distinguish between these types, the clinical examination under loaded conditions (active abduction or elevation against resistance) shows an increased superior displacement in type IIb patients. This could be also proved by a radiologic investigation under fluoroscopy (Fig. 23-11).
The four types are markedly different in respect to preoperative function after elimination of pain and to the results after conventional shoulder hemiarthroplasty.46 The amount of anterior-superior displacement of rotator cuff–deficient shoulders depends on several factors. The functional impairment is directly correlated with the amount of superior translation of the center of rotation and with the extent of static or dynamic superior instability of the joint.
DIFFERENTIAL DIAGNOSIS
It is important to recognize the various disease processes leading to glenohumeral RCDA.
Rheumatoid arthritis (RA) is the most common cause of RCDA. Between 48% and 65% of RA patients have significant glenohumeral joint involvement. About 24% of those having glenohumeral arthritis have a simultaneous rotator cuff tear.47,48 Superimposed on the aforementioned changes are severe osteopenia, erosions of the entire glenoid without osteophyte formation, and medialization of the glenohumeral joint.
CTA is the extreme end result of a massive rotator cuff tear. The term cuff tear arthropathy, coined by Neer in 1983,43 refers to a primary massive rotator cuff tear that by virtue of mechanical superior instability and nutritional effects leads to a secondary glenohumeral joint destruction. It is believed that up to 25% of massive rotator cuff tears will end up as CTA, but it is difficult, if not impossible, to predict which massive tears will progress to this result.
The Milwaukee shoulder syndrome was originally described by McCarty in 1981.49 This is an uncommon entity affecting shoulders of elderly persons, predominantly women. It consists of a massive rotator cuff tear, joint instability, bony destruction, and large blood-stained joint effusion containing basic calcium phosphate crystals, detectable protease activity, and minimal inflammatory elements. Its relation to rotator cuff arthropathy is not clear, and it might represent one of the previously discussed syndromes. The role of the basic calcium phosphate crystals in producing this syndrome is still controversial.
Primary glenohumeral osteoarthritis is the most common reason for shoulder joint replacement. However, it is associated with rotator cuff tear in only 5% of patients, and most of these tears are reparable. It is therefore uncommon for primary osteoarthritis to end up as RCDA.
EVALUATION
Patients with rotator cuff deficiency have various degrees of pain, recurrent swelling and functional loss.43 The etiology may be trauma or various diseases affecting the articular surfaces and soft tissues around the joint, such as rheumatoid arthritis. To determine optimal treatment, a careful clinical evaluation is necessary. It is important to take the patient’s age and functional demands into account.
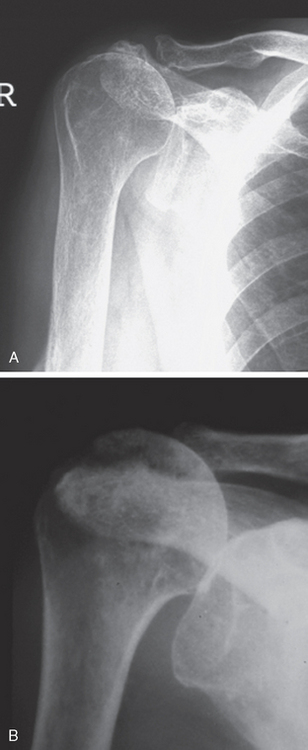
Active and passive range of motion is evaluated. If there is severe pain, injection of local anesthesia into the joint can be helpful in evaluating the true active range of motion. Patients with preserved fixed fulcrum kinematics have good active range of motion, whereas patients with biomechanical deficiency (unbalanced shoulder) have poor active range of motion (pseudoparalysis) (Fig. 23-12).
TREATMENT
Arthroscopic Surgery
Arthroscopic débridement and biceps tenotomy has been reported to improve function in patients with cuff tear arthopathy.50,51 This can be tried, particularly in elderly patients with low demands and in patients not willing to undergo major surgery. It might relieve pain but does not restore function in pseudoparalytic shoulders.
Arthrodesis
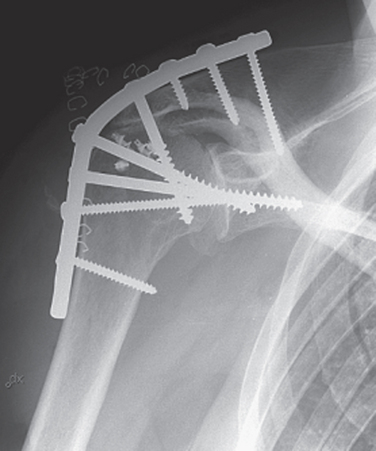
Good pain relief can be achieved by performing an arthrodesis in patients with cuff tear arthropathy (Fig. 23-13). In patients with pseudoparalysis, an arthrodesis can also improve functional range of motion. However, increased load on the thoracoscapular articulation and the acromioclavicular joint can cause pain from these locations. In 1991, Arntz and colleagues reported 10 patients who underwent arthrodesis for irreparable cuff tears.52 The arthrodesis improved active forward elevation by a mean of 15 degrees. They concluded that arthrodesis could be indicated in patients with massive irreparable cuff tears and significant deltoid deficiency or in young patients with significant strength demands at low angles of flexion.
Anatomic Unconstrained Arthroplasty
Total Shoulder Arthroplasty
The superior migration of the humeral head in patients with cuff tear arthropathy creates eccentric load on the glenoid component. This has been called the rocking horse effect; it results in increased rates of loosening.53 In 1991, Lohr and Cofield reported better pain relief in total shoulder arthroplasty compared to hemiarthroplasty in 22 patients with cuff deficiency, but an increased incidence of radiographic and clinical loosening.54 Similar findings were reported by Franklin and colleagues in a study where three of nine patients with cuff deficiency needed early revision after total shoulder arthroplasty.53 Due to this increased risk of glenoid loosening, anatomic total shoulder replacement is not recommended in patients with significant cuff deficiency.
Hemiarthroplasty
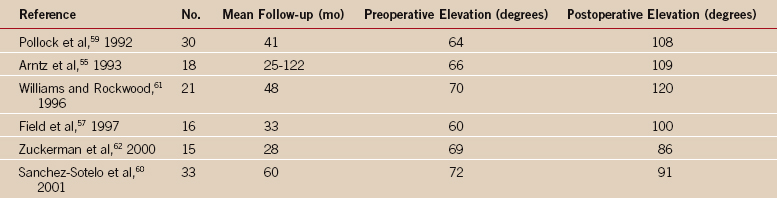
Hemiarthroplasty has been the treatment of choice for the arthritic shoulder with cuff deficiency. Due to the clinical success of the Grammont reverse ball-and-socket arthroplasty, the indication for using a hemiarthroplasty is more limited. The results after hemiarthroplasty are unpredictable, and Neer proposed the term limited-goal rehabilitation for these patients.1 Persistent pain has been reported in 6% to 53% of the patients after hemiarthroplasty; active elevation is reported at 60 to 70 degrees preoperatively improving to 90 to 120 degrees postoperatively (Table 23-2).3,55–62
An oversized humeral head has been suggested to create a better articulation with the acromion and the coracoacromial arch. However, Sanchez and colleagues could not find a correlation between head size and function, and there are several disadvantages. An oversized humeral head can increase the joint reaction forces and possibly more-rapid bone resorption.60 A large head makes it more difficult to partially repair the rotator cuff. A larger head puts the center of rotation more lateral than inferior, which contributes a biomechanical disadvantage to the deltoid by shortening the lever arm. Any attempt to even partially repair the deficient rotator cuff becomes much more difficult with a larger humeral head.
Several techniques have been proposed and used for this purpose. Pollock,59 Cantrell and Burkhead,63 and DiGiovanni3 advocated the use of subscapularis tendon transfer to a more superior position as a method for superior stabilization of the humeral head. The subscapularis tendon can then be reinserted into the top of the greater tuberosity. The use of anatomic, or slightly smaller, humeral head size facilitates this transfer. This option is limited to patients who have a functional unshortened subscapularis tendon.
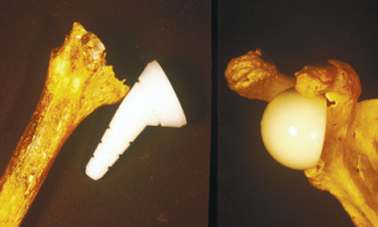
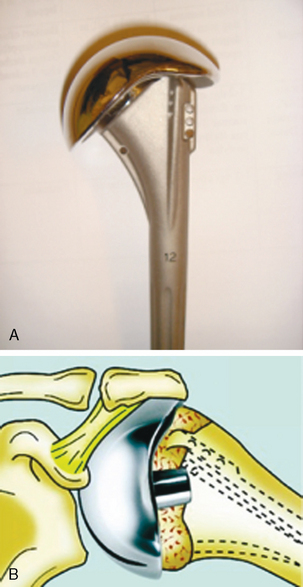
In an attempt to improve results of hemiarthroplasty in patients with cuff tear arthropathy, a specific head has been designed: the CTA head (Fig. 23-14). It has a lateral extension over the greater tuberosity, producing a greater arc for articulation against the glenoid fossa and the undersurface of the acromion and coracoacromial arch. The head size is anatomic, thus there is no overstuffing of the joint.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree