Chapter 138 Vitex agnus castus (Chaste Tree)
Vitex agnus castus (family: Verbenaceae)
 General Description
General Description
Vitex agnus castus, also known as chaste tree, is a shrub with finger-shaped leaves and slender violet flowers. It grows in creek beds and on river banks in valleys and lower foothills in the Mediterranean and Central Asia. The plant blooms in high summer and, after pollination, develops dark-brown to black fruit the size of peppercorns. The fruit possesses a pepper-like aroma and flavor. The ripe, dried fruit of V. agnus castus is the part of the plant used in medicinal preparations today.1
 Chemical Composition
Chemical Composition
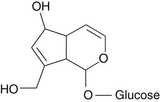
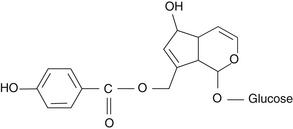
The fruit of Vitex contains essential oils, iridoid glycosides, and flavonoids.2 The essential oils include limonene, 1,8-cineole, and sabinene.3 The primary flavonoids are castican, orientin, and isovitexin. The two isolated iridoid glycosides are agnuside and aucubin (Figures 138-1 and 138-2).4 Agnuside serves as a reference material for quality control in the manufacture of Vitex extracts, although the flavonoid casticin appears to be the most active component.
 Pharmacology
Pharmacology
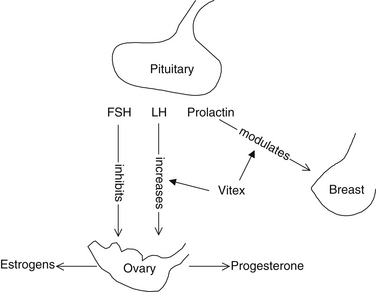
Vitex acts on the hypothalamic-pituitary axis. One observed effect is that it increases the production of luteinizing hormone (LH) (Figure 138-3), resulting in a corpus luteum–like hormonal effect that shifts the estrogen/progesterone ratio in favor of progesterone.5 The ability of Vitex to increase or modulate the body’s progesterone levels is therefore an indirect effect and not a direct hormonal action.6
Vitex also modulates the secretion of prolactin from the pituitary gland. In studies with rats, it was shown to inhibit prolactin release by the pituitary gland, particularly in conditions of stress. The mechanism of action appears to involve the ability of Vitex to directly bind dopamine receptors and subsequently inhibit prolactin release in the pituitary.7,8 The flavonoid casticin appears to be responsible for this antihyperprolactinemia effect.9 Vitex also possesses significant antioxidant effect.10,11
 Clinical Applications
Clinical Applications
Many of these conditions have been linked to corpus luteum insufficiency.
Corpus Luteum Insufficiency
Corpus luteum insufficiency (also referred to as luteal phase defect) is a manifestation of suboptimal ovarian function. In laboratory terms, corpus luteum insufficiency is usually defined as an abnormally low progesterone level 3 weeks after the onset of menstruation (serum progesterone below 10 to 12 ng/mL). This state is normal during puberty and at menopause; however, it is usually considered abnormal in women between the ages of 20 and 40 years.12
Corpus luteum insufficiency may give rise to a myriad of menstrual abnormalities. Table 138-1 lists the most common clinical conditions in 1592 women diagnosed with corpus luteum insufficiency. Foremost are hypermenorrhea (heavy periods), polymenorrhea (abnormally frequent periods), and persistent anovulatory bleeding. Secondary amenorrhea (lack of a period) may sometimes be observed in women with corpus luteum insufficiency.
TABLE 138-1 The Most Common Clinical Conditions in 1592 Women Diagnosed with Corpus Luteum Insufficiency
| DIAGNOSIS | NO. OF PATIENTS | PERCENTAGE OF TOTAL |
|---|---|---|
| Hypermenorrhea | 418 | 26.3 |
| Polymenorrhea | 369 | 23.2 |
| Persistent anovulatory bleeding | 216 | 13.6 |
| Secondary amenorrhea | 202 | 12.7 |
| Dysmenorrhea | 186 | 11.7 |
| Anovulatory cycles | 175 | 11.0 |
| Involuntary sterility | 145 | 9.1 |
| Oligomenorrhea | 69 | 4.3 |
| Menorrhagia/metrorrhagia | 66 | 4.1 |
| Irregular menstrual cycles | 32 | 2.0 |
| Primary amenorrhea | 1 | 0.1 |
Disturbances of other hormones may also be associated with corpus luteum insufficiency. One study found hyperprolactinemia in 70% of cases.13 Also noted is an exaggerated response to the thyroid-releasing hormone test, which is associated with manifest or latent hypothyroidism.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree