Chapter 119 Ruscus aculeatus (Butcher’s Broom)
Ruscus aculeatus (family: Liliaceae)
 General Description
General Description
Ruscus aculeatus (butcher’s broom) is an evergreen shrub native to Mediterranean Europe and Africa.1 It has rigid leaves that terminate in a single sharp spine. Its berries remain attached to the plant through the winter. The root, the part used medicinally, is thick, typically 2 to 4 inches long, with many woody rootlets growing on its lower surface.
 Chemical Composition
Chemical Composition
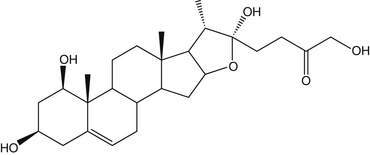
The primary active ingredients of butcher’s broom are the steroidal saponins ruscogenin and neoruscogenin (up to 6%). The plant also contains other steroidal sapogenins and saponins (such as ruscin and ruscoside), fatty acids, sterols, flavonoids, coumarins, sparteine, tyramine, and glycolic acid1–5 (see Figure 119-1).
 History and Folk Use
History and Folk Use
In ancient Greece, butcher’s broom was used as a laxative and to treat kidney stones.6 In Europe, a wine decoction of the root was used as a diuretic to treat urinary obstructions, kidney stones, and gravel. It was also used to regulate menses, ameliorate jaundice, and treat headaches. In South America, the roasted root was made into a beverage for prostate tumors.7
 Pharmacology
Pharmacology
Anticancer Effect
Butcher’s broom saponins have a cytostatic effect on leukemia HL-60 cells in vitro.8
Antielastase Effect
Ruscinogens isolated from butcher’s broom have a remarkable antielastase effect in vitro.9
Antihypoxic Effect
Hypoxia-induced activation of endothelial cells may be a cause of venous disease. A proprietary extract of butcher’s broom known as Cyclo 3 Fort dose-dependently protected human endothelial cells from induced hypoxia in vitro.9
Antiinflammatory Effect
Butcher’s broom extract and its isolated steroidal saponins were antiinflammatory in several animal models.10 Injected intravenously, it inhibits the permeability increasing effects of bradykinin, leukotriene B4, and histamine in hamster cheek pouch preparations.11
Fibrinolytic Effect
Thrombosis in chronic venous insufficiency (CVI) is associated with reduced levels of tissue plasminogen activator in varicose veins. In a small study, 20 patients scheduled for vein stripping surgery took butcher’s broom (150 mg) and methyl hesperidin chalcone (150 mg) or placebo three times daily for 14 days before surgery.13 Segments of the great saphenous vein removed from patients in the active group had enhanced levels of tissue plasminogen activator and enhanced fibrinolytic activity compared with placebo.
Lymphatic Flow Effect
Butcher’s broom dose dependently enhanced the duration and flow intensity of the lymphatic vessels in dogs.14 This effect did not change when the calcium channel antagonist nifedipine was injected, indicating that butcher’s broom’s lymphatic effect does not involve the opening of calcium channels but appears to act directly on peripheral lymphatics.
Vasoconstrictive Effect
Topical administration of butcher’s broom extract in a hamster cheek preparation dose dependently inhibited histamine-induced increases in permeability.15 Research suggested that butcher’s broom’s vasoconstrictive effect is mediated by calcium and α-1 adrenergic receptors. Heat increases, and cold decreases, butcher’s broom’s ability to increase the release of norepinephrine from the adrenoreceptors.15
 Clinical Applications
Clinical Applications
Chronic Venous Insufficiency
Butcher’s broom is most commonly used to alleviate the symptoms of CVI, including ankle edema, itching, tension, and cramping of the legs. Both animal and clinical studies support its effectiveness in CVI, and the German Commission E approved butcher’s broom as an adjunct treatment for this condition.1 A meta-analysis evaluated 20 placebo-controlled, double-blind studies and 5 randomized trials against a comparator drug along with 6 single-arm studies without a placebo. The review included information on 10,246 subjects. Although data quality varied, Cyclo 3 Fort significantly reduced CVI symptoms compared with placebo.15a
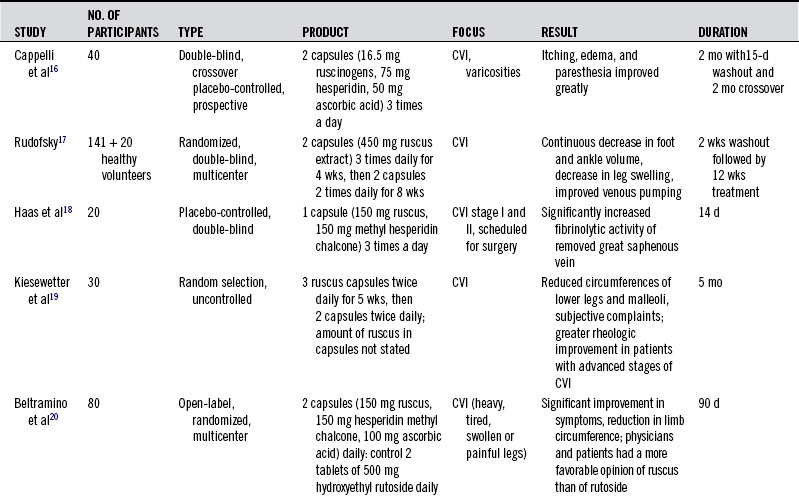
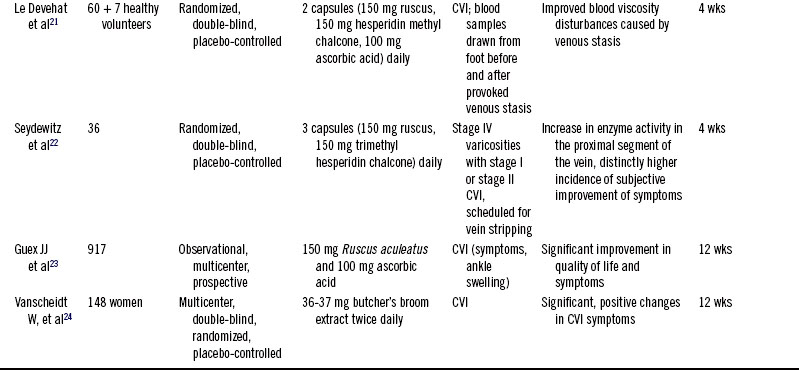
Nine of these studies are summarized in Table 119-1.16–24 All but two were randomized double-blind trials. In one large multicenter trial, 81.6% of treating physicians rated its efficacy as excellent, and the remaining physicians rated it as good.20 Butcher’s broom acts most strongly when circulation is impaired, but some researchers caution that it is less effective when the disease has compromised venous wall receptor activity.18
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree