In order to perform a comprehensive neuromusculoskeletal examination, the clinician must have a clear understanding of the anatomy, physiology, and function of the various components of the nervous system, and be able to recognize those signs and symptoms that indicate a compromise of the nervous system. The nervous system is composed of only two principal types of cells—neurons and supporting cells. The neuron, which is classified according to structure or function, serves to store and process information, and is the functional unit of the nervous system. The supporting cell called the neuroglial cell, or simply glial, functions to provide structural and metabolic support for the neurons.1 Unlike many cells, neurons cannot divide by mitosis but they do have some capability to regenerate. In contrast, glial cells retain limited mitotic abilities.
CHAPTER 3
The Nervous System
OVERVIEW
Anatomy
The human nervous system can be subdivided into two anatomic divisions: the central nervous system (CNS), comprising the brain and the spinal cord, and the peripheral nervous system (PNS), formed by the nerves and ganglia (cluster of nerve cell bodies located outside of the CNS) outside of the brain and spinal cord. The PNS is further subdivided into somatic and autonomic divisions. The somatic division, which includes the cranial (with the exception of cranial II) and the spinal nerves, innervates the skin, the muscles, and the joints, while the autonomic division innervates the glands and the smooth muscle of the viscera and the blood vessels.2
Although neurons come in various sizes and shapes, there are four functional parts for each nerve fiber (Fig. 3-1):
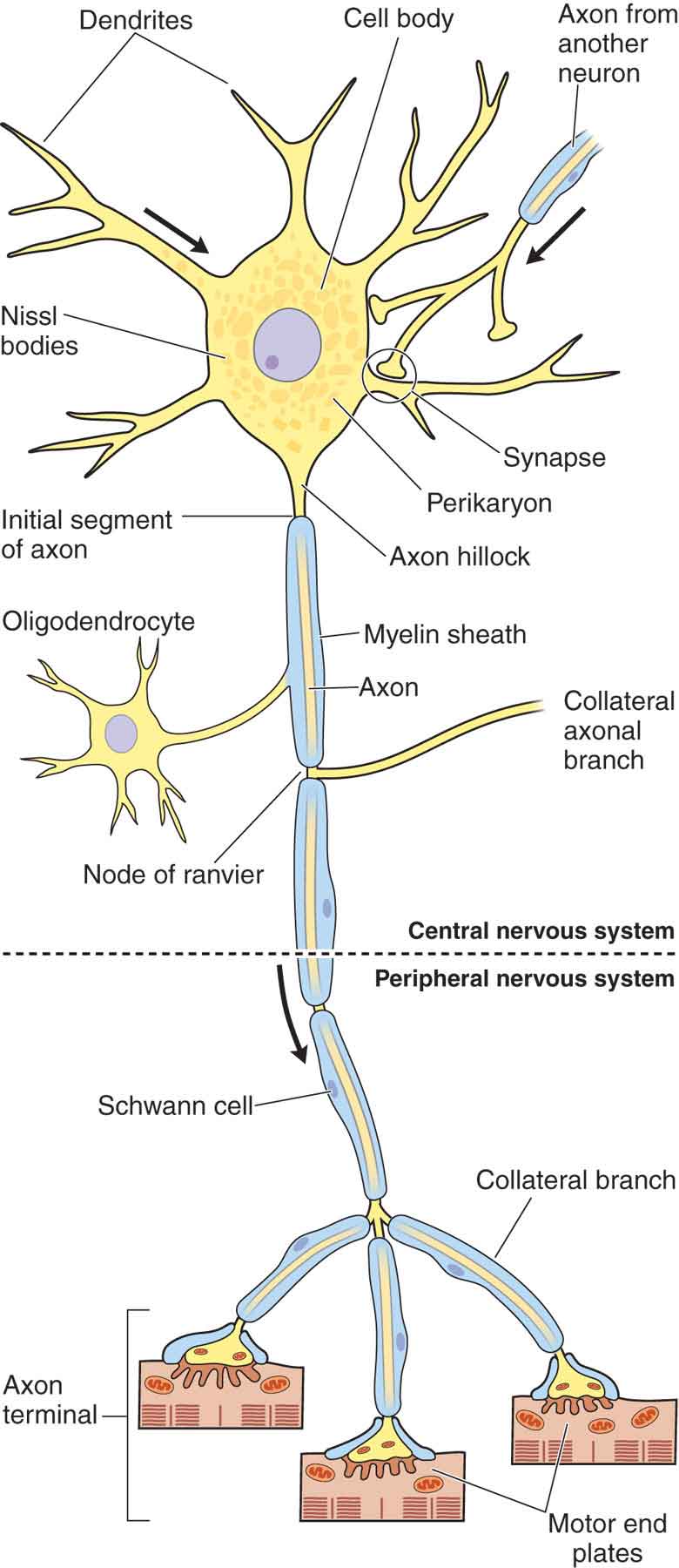
FIGURE 3-1 Schematic drawing of a neuron. (Reproduced, with permission, from Chapter 9. Nerve Tissue & the Nervous System. In: Mescher AL. eds. Junqueira’s Basic Histology: Text & Atlas, 13e. New York, NY: McGraw-Hill; 2013.)
 Dendrite. Dendrites serve a receptive function and receive information from other nerve cells, or the environment.
Dendrite. Dendrites serve a receptive function and receive information from other nerve cells, or the environment.
 Axon. The axon cylinder, in which there is a bidirectional flow of axoplasm, conducts information and nutrition to the nerve cells and the tissues that the nerve innervates. Many axons are covered by myelin, a lipid-rich membrane. In myelinated fibers, there is a direct proportional relationship between fiber diameter and conduction velocity.3 This membrane is divided into segments, approximately 1-mm long, by small gaps, called nodes of Ranvier, in which the myelin is absent.4 Myelin, which has a high electrical resistance and low capacitance, serves to increase the nerve conduction velocity of neural transmissions through a process called salutatory conduction. The Schwann cell is responsible for laying down myelin around axons.
Axon. The axon cylinder, in which there is a bidirectional flow of axoplasm, conducts information and nutrition to the nerve cells and the tissues that the nerve innervates. Many axons are covered by myelin, a lipid-rich membrane. In myelinated fibers, there is a direct proportional relationship between fiber diameter and conduction velocity.3 This membrane is divided into segments, approximately 1-mm long, by small gaps, called nodes of Ranvier, in which the myelin is absent.4 Myelin, which has a high electrical resistance and low capacitance, serves to increase the nerve conduction velocity of neural transmissions through a process called salutatory conduction. The Schwann cell is responsible for laying down myelin around axons.
 Cell body. The cell body contains the nucleus of the cell and has important integrative functions.
Cell body. The cell body contains the nucleus of the cell and has important integrative functions.
 Axon terminal. The axon terminal is the transmission site for action potentials, the messengers of the nerve cell.
Axon terminal. The axon terminal is the transmission site for action potentials, the messengers of the nerve cell.
Peripheral nerves are enclosed in three layers of tissue of differing character. From the inside outward, these are the endoneurium, perineurium, and epineurium.1 The nerve fibers embedded in endoneurium form a funiculus surrounded by perineurium, a thin but strong sheath of connective tissue. A fluid exists in the endoneurial spaces, which following nerve injury can produce intraneural edema, which in turn can play a major role in acute and chronic nerve lesions.3 The nerve bundles are embedded in a loose areolar connective tissue framework, called the epineurium. The epineurium that extends between the fascicles is termed the inner or the interfascicular epineurium, whereas that surrounding the entire nerve trunk is called the epifascicular epineurium.5 The connective tissue outside the epineurium is referred to as the adventitia of the nerve or the epineural tissue.5 Although the epineurium is continuous with the surrounding connective tissue, its attachment is loose, so that nerve trunks are relatively mobile, except where tethered by entering vessels or exiting nerve branches (see Chapter 11).6 There are no connective tissue components in the spinal nerves comparable to the epineurium and the perineurium of the peripheral nerve; at least they are not developed to the same degree.7 As a result, the spinal nerve roots are more sensitive to both tension and compression. The spinal nerve roots also are devoid of lymphatics and, thus, are predisposed to prolonged inflammation.8
The communication of information from one nerve cell to another occurs at junctions called synapses, where a chemical is released in the form of a neurotransmitter. A difference in concentration exists across the cell membrane of potassium, sodium, and chloride ions. These ions can selectively permeate ion channels in the membrane so that an unequal distribution of net charge occurs. The resting membrane potential results from an internal negativity resulting from the active transport of sodium from inside to outside the cell, and potassium from outside to inside the cell.3
Central Nervous System
The CNS consists of the brain and an elongated spinal cord. The spinal cord participates directly in the control of body movements, the processing and transmission of sensory information from the trunk and the limbs, and the regulation of visceral functions.2 The spinal cord also provides a conduit for the two-way transmission of messages between the brain and the body. These messages travel along the pathways, or tracts, that are fiber bundles of similar groups of neurons. These tracts may descend or ascend.
The spinal cord is normally 42–45-cm long in adults and is continuous with the medulla and brain stem at its upper end (Fig. 3-2A).4 The conus medullaris serves as the distal end of the cord, and, in adults, the conus ends at the L1 or L2 level of the vertebral column. A series of specializations, the filum terminales and the coccygeal ligament, anchor the spinal cord and the dural sac inferiorly and ensure that the tensile forces applied to the spinal cord are distributed throughout its entire length.9 The spinal cord has an external segmental organization. Each of the 31 pairs of spinal nerves that arise from the spinal cord has an anterior (ventral) root and a posterior (dorsal) root, with each root consisting of one to eight rootlets and bundles of nerve fibers.4 A spinal (sensory) ganglion (posterior [dorsal] root ganglion), a swelling that contains nerve cell bodies, is located in the posterior (dorsal) root (Fig. 3-2B) of a typical spinal nerve (Fig. 3-2B).4
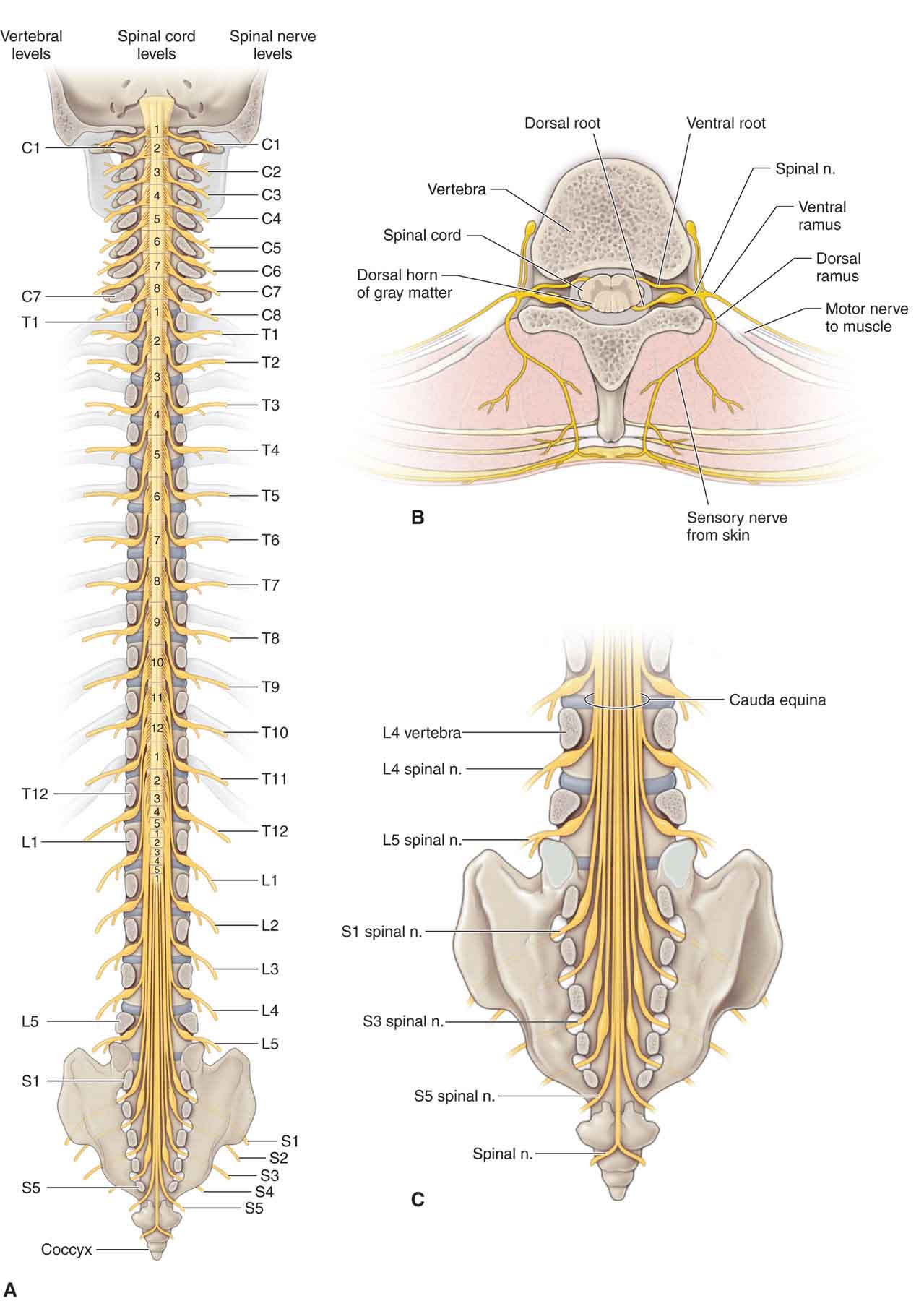
FIGURE 3-2 Schematic illustration of the spinal cord. (Reproduced, with permission, from Chapter 1. Back. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
Three membranes, or meninges, envelop the structures of the CNS: dura mater, arachnoid, and pia mater (Fig. 3-3). The meninges and related spaces are important to both the nutrition and the protection of the spinal cord. The cerebrospinal fluid that flows through the meningeal spaces, and within the ventricles of the brain, provides a cushion for the spinal cord. The meninges also form barriers that resist the entrance of various noxious organisms.
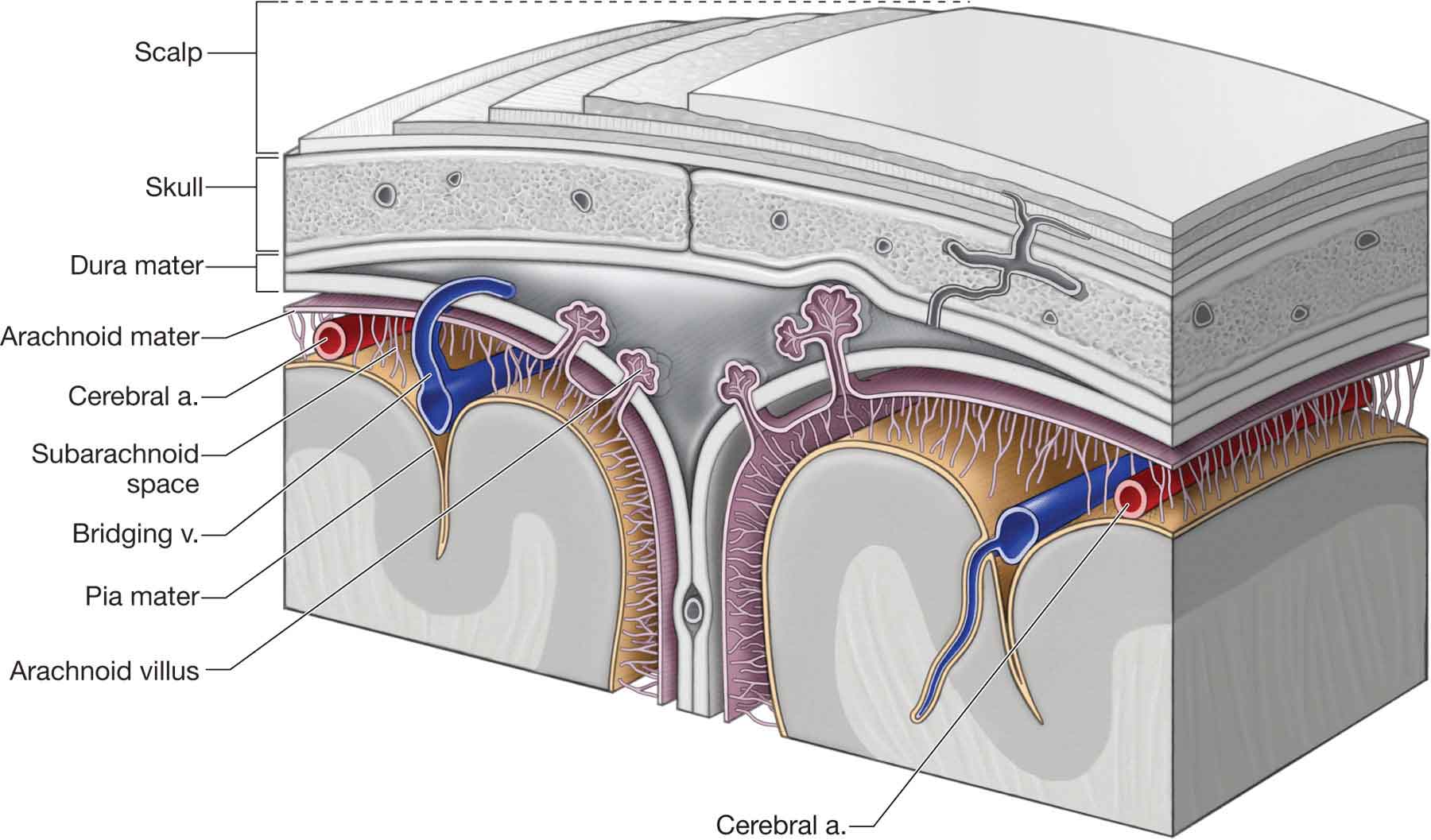
FIGURE 3-3 Schematic illustration of the relationship of the dura mater, arachnoid, and pia mater. (Reproduced, with permission, from Chapter 15. Scalp, Skull, and Meninges. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
Dura Mater
The dura mater (Latin, tough mother) (Fig. 3-3) is the outermost and the strongest of the membranes and is composed of an inner meningeal layer and an outermost periosteal layer. The dura runs uninterrupted from the interior of the cranium through the foramen magnum and surrounds the spinal cord throughout its distribution from the cranium to the coccyx at the second sacral level (S2).9 The dura also is attached to the posterior surfaces of C2 and C3.10
The dura forms a vertical sac (dural sac) around the spinal cord, and its short lateral projections blend with the epineurium of the spinal nerves. The dura is separated from the bones and the ligaments that form the walls of the vertebral canal by an epidural space, which can become partly calcified or even ossified with age.4
Arachnoid
The arachnoid is a thin and delicate avascular layer, coextensive with the dura mater and the pia mater (Fig. 3-3). Even though the arachnoid and the pia mater are interconnected by trabeculae, there is a space between them, called the subarachnoid space (Fig. 3-3), which contains the cerebrospinal fluid. The supposedly rhythmic flow of this cerebrospinal fluid is the rationale used by craniosacral therapists to explain their techniques, although there is no evidence of this finding in the literature.
Pia Mater
The pia mater (Fig. 3-3) is the deepest of the layers. It is intimately related and firmly attached, via connective tissue investments, to the outer surface of the spinal cord and the nerve roots. The pia mater conveys the blood vessels that supply the spinal cord and has a series of lateral specializations, the denticulate (dentate) ligaments, which anchor the spinal cord to the dura mater.9 These ligaments, which derive their name from their tooth-like appearance, extend the whole length of the spinal cord.
Peripheral Nervous System: Somatic Nerves
The somatic portion of the PNS consists of the cranial nerves (CNs) and the spinal nerves.
Cranial Nerves
The CNs, typically, are described as comprising 12 pairs, which are referred to by the Roman numerals I through XII (Fig. 3-4). The CN roots enter and exit the brain stem to provide sensory and motor innervation to the head and the muscles of the face. CN I (olfactory) and CN II (optic) are not true nerves but rather fiber tracts of the brain. The examination of the CN system is described later in this chapter (see section “Orthopaedic Neurologic Testing”).
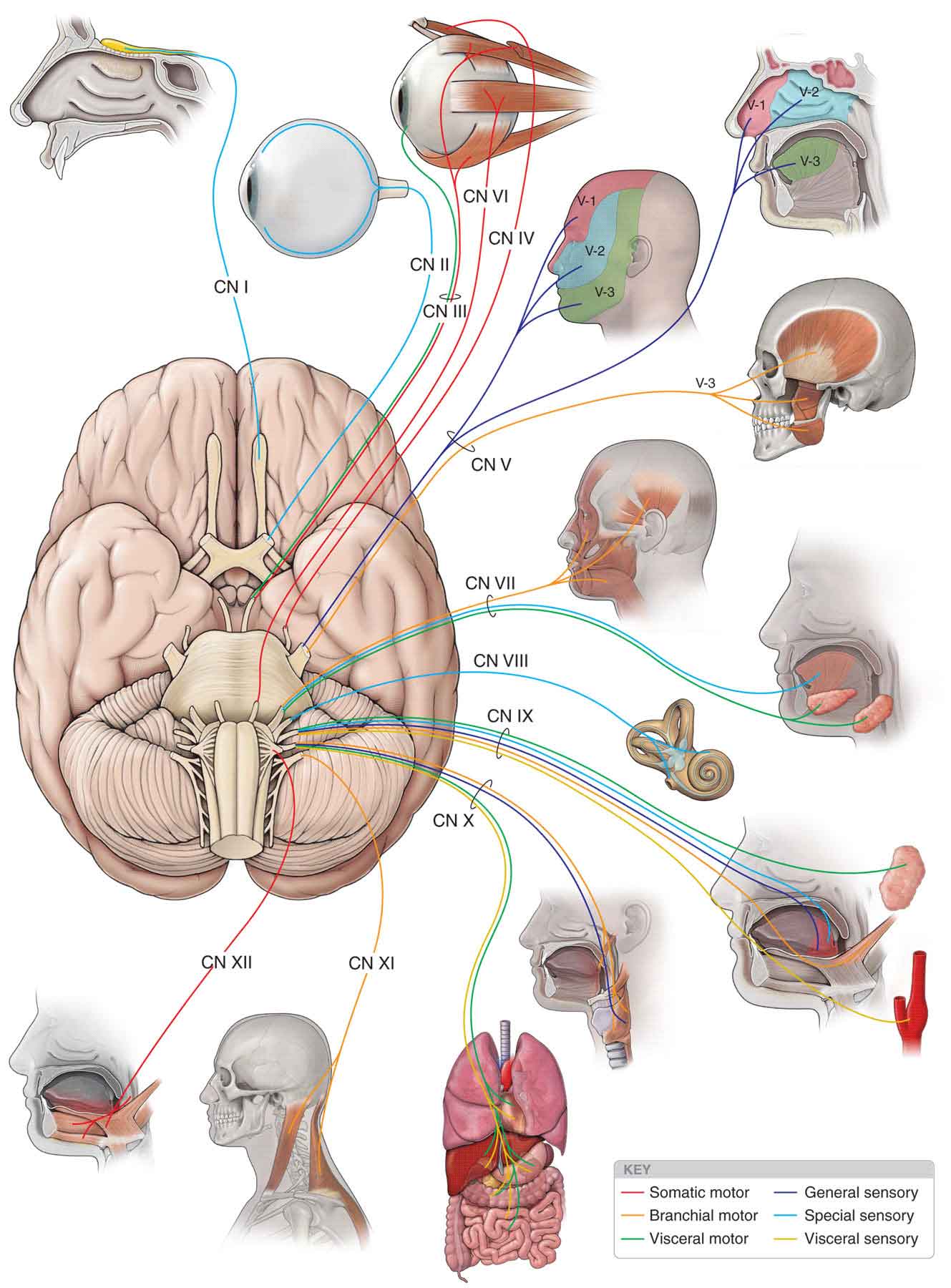
FIGURE 3-4 The CNs. (Reproduced, with permission, from Chapter 17. Cranial Nerves. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
 CN I (olfactory). The olfactory tract (Fig. 3-4) arises from the olfactory bulb on the inferior aspect of the frontal lobe, just above the cribriform plate. From here it continues posteriorly as the olfactory tract and terminates just lateral to the optic chiasm. The olfactory nerve handles the sense of smell.
CN I (olfactory). The olfactory tract (Fig. 3-4) arises from the olfactory bulb on the inferior aspect of the frontal lobe, just above the cribriform plate. From here it continues posteriorly as the olfactory tract and terminates just lateral to the optic chiasm. The olfactory nerve handles the sense of smell.
 CN II (optic). The fibers of the optic nerve arise from the inner layer of the retina and proceed posteriorly to enter the cranial cavity via the optic foramen, to form the optic chiasm (Fig. 3-4). The fibers from the nasal half of the retina decussate within the optic chiasm, whereas those from the lateral half do not. The optic nerve handles vision.
CN II (optic). The fibers of the optic nerve arise from the inner layer of the retina and proceed posteriorly to enter the cranial cavity via the optic foramen, to form the optic chiasm (Fig. 3-4). The fibers from the nasal half of the retina decussate within the optic chiasm, whereas those from the lateral half do not. The optic nerve handles vision.
 CN III (oculomotor). The oculomotor nerve arises in the oculomotor nucleus and leaves the brain on the medial aspect of the cerebral peduncle (Fig. 3-4). It then extends from the interpeduncular fossa and runs between the posterior cerebral artery and the superior cerebellar artery, before leaving the cranial cavity and entering the cavernous sinus by way of the superior orbital fissure. The somatic portion of the oculomotor nerve supplies the levator palpebrae superioris muscle; the superior, medial, and inferior rectus muscles; and the inferior oblique muscles (Fig. 3-4). These muscles handle some eye movements. The visceral efferent portion of this nerve innervates two smooth intraocular muscles: the ciliary and the constrictor pupillae. These muscles handle pupillary constriction.
CN III (oculomotor). The oculomotor nerve arises in the oculomotor nucleus and leaves the brain on the medial aspect of the cerebral peduncle (Fig. 3-4). It then extends from the interpeduncular fossa and runs between the posterior cerebral artery and the superior cerebellar artery, before leaving the cranial cavity and entering the cavernous sinus by way of the superior orbital fissure. The somatic portion of the oculomotor nerve supplies the levator palpebrae superioris muscle; the superior, medial, and inferior rectus muscles; and the inferior oblique muscles (Fig. 3-4). These muscles handle some eye movements. The visceral efferent portion of this nerve innervates two smooth intraocular muscles: the ciliary and the constrictor pupillae. These muscles handle pupillary constriction.
 CN IV (trochlear). The trochlear nerve arises from the trochlear nucleus, just caudal to the oculomotor nucleus at the anterior border of the periaqueductal gray (PAG) matter (Fig. 3-4). The fibers cross within the midbrain and then emerge contralaterally on the posterior surface of the brain stem, before entering the orbit via the superior orbital fissure, to supply the superior oblique muscle.
CN IV (trochlear). The trochlear nerve arises from the trochlear nucleus, just caudal to the oculomotor nucleus at the anterior border of the periaqueductal gray (PAG) matter (Fig. 3-4). The fibers cross within the midbrain and then emerge contralaterally on the posterior surface of the brain stem, before entering the orbit via the superior orbital fissure, to supply the superior oblique muscle.
Note: Because nerves III, IV, and VI are examined together, CN V is described after CN VI.
 CN VI (abducens). The abducens nerve originates from the abducens nucleus within the inferior aspect of the pons. Its long intracranial course to the superior orbital fissure makes it vulnerable to pathology in the posterior and middle cranial fossa. The nerve innervates the lateral rectus muscle.
CN VI (abducens). The abducens nerve originates from the abducens nucleus within the inferior aspect of the pons. Its long intracranial course to the superior orbital fissure makes it vulnerable to pathology in the posterior and middle cranial fossa. The nerve innervates the lateral rectus muscle.
 CN V (trigeminal). The trigeminal nerve is so named because of its tripartite division into the maxillary, ophthalmic, and mandibular branches (V-1, V-2, and V-3 respectively in Fig. 3-4). All three of these branches contain sensory cells, but the ophthalmic and the maxillary are exclusively sensory, the latter supplying the soft and hard palate, maxillary sinuses, upper teeth and upper lip, and mucous membrane of the pharynx. The mandibular branch carries sensory information but also represents the motor component of the nerve, supplying the muscles of mastication, both pterygoids, the anterior belly of digastric, tensor tympani, tensor veli palatini, and mylohyoid.
CN V (trigeminal). The trigeminal nerve is so named because of its tripartite division into the maxillary, ophthalmic, and mandibular branches (V-1, V-2, and V-3 respectively in Fig. 3-4). All three of these branches contain sensory cells, but the ophthalmic and the maxillary are exclusively sensory, the latter supplying the soft and hard palate, maxillary sinuses, upper teeth and upper lip, and mucous membrane of the pharynx. The mandibular branch carries sensory information but also represents the motor component of the nerve, supplying the muscles of mastication, both pterygoids, the anterior belly of digastric, tensor tympani, tensor veli palatini, and mylohyoid.
The spinal nucleus and the tract of the trigeminal nerve cannot be distinguished either histologically or on the basis of afferent reception from the cervical nerves. Consequently, the entire column can be viewed as a single nucleus and, legitimately, may be called the trigeminocervical nucleus.
 CN VII (facial). The facial nerve is made up of a sensory (intermediate) root, which conveys taste, and a motor root, the facial nerve proper, which supplies the muscles of facial expression, the platysma muscle, and the stapedius muscle of the inner ear (Fig. 3-4). The intermediate root, together with the motor nerve and CN VIII, travels through the internal acoustic meatus to enter the facial canal of the temporal bone. From here, the intermediate nerve swells to form the geniculate ganglion and gives off the greater superficial petrosal nerve, which eventually innervates the lacrimal and salivary glands via the pterygopalatine ganglion and the chorda tympani nerve, respectively. The facial nerve proper exits the skull through the stylomastoid foramen.
CN VII (facial). The facial nerve is made up of a sensory (intermediate) root, which conveys taste, and a motor root, the facial nerve proper, which supplies the muscles of facial expression, the platysma muscle, and the stapedius muscle of the inner ear (Fig. 3-4). The intermediate root, together with the motor nerve and CN VIII, travels through the internal acoustic meatus to enter the facial canal of the temporal bone. From here, the intermediate nerve swells to form the geniculate ganglion and gives off the greater superficial petrosal nerve, which eventually innervates the lacrimal and salivary glands via the pterygopalatine ganglion and the chorda tympani nerve, respectively. The facial nerve proper exits the skull through the stylomastoid foramen.
 CN VIII (vestibulocochlear). The vestibulocochlear nerve subserves two different senses: balance and hearing. The cochlear portion of the nerve arises from spiral ganglia, and the vestibular portion arises from the vestibular ganglia in the labyrinth of the inner ear (Fig. 3-4). The cochlear portion is concerned with the sense of hearing, whereas the vestibular portion is a part of the system of equilibrium, the vestibular system.
CN VIII (vestibulocochlear). The vestibulocochlear nerve subserves two different senses: balance and hearing. The cochlear portion of the nerve arises from spiral ganglia, and the vestibular portion arises from the vestibular ganglia in the labyrinth of the inner ear (Fig. 3-4). The cochlear portion is concerned with the sense of hearing, whereas the vestibular portion is a part of the system of equilibrium, the vestibular system.
The vestibular system includes the vestibular apparatus of the inner ear, the vestibular nuclei and their neural projections, and the exteroreceptors throughout the body, especially in the upper cervical spine and the eyes.11
The apparatus of the inner ear consists of the static labyrinth, which comprises three semicircular canals (SCC) (Fig. 3-5), each orientated at right angles to the other. The labyrinth includes specialized sensory areas that are located in the utricle and the saccule (Fig. 3-5), within which otoliths are located (Fig. 3-5).
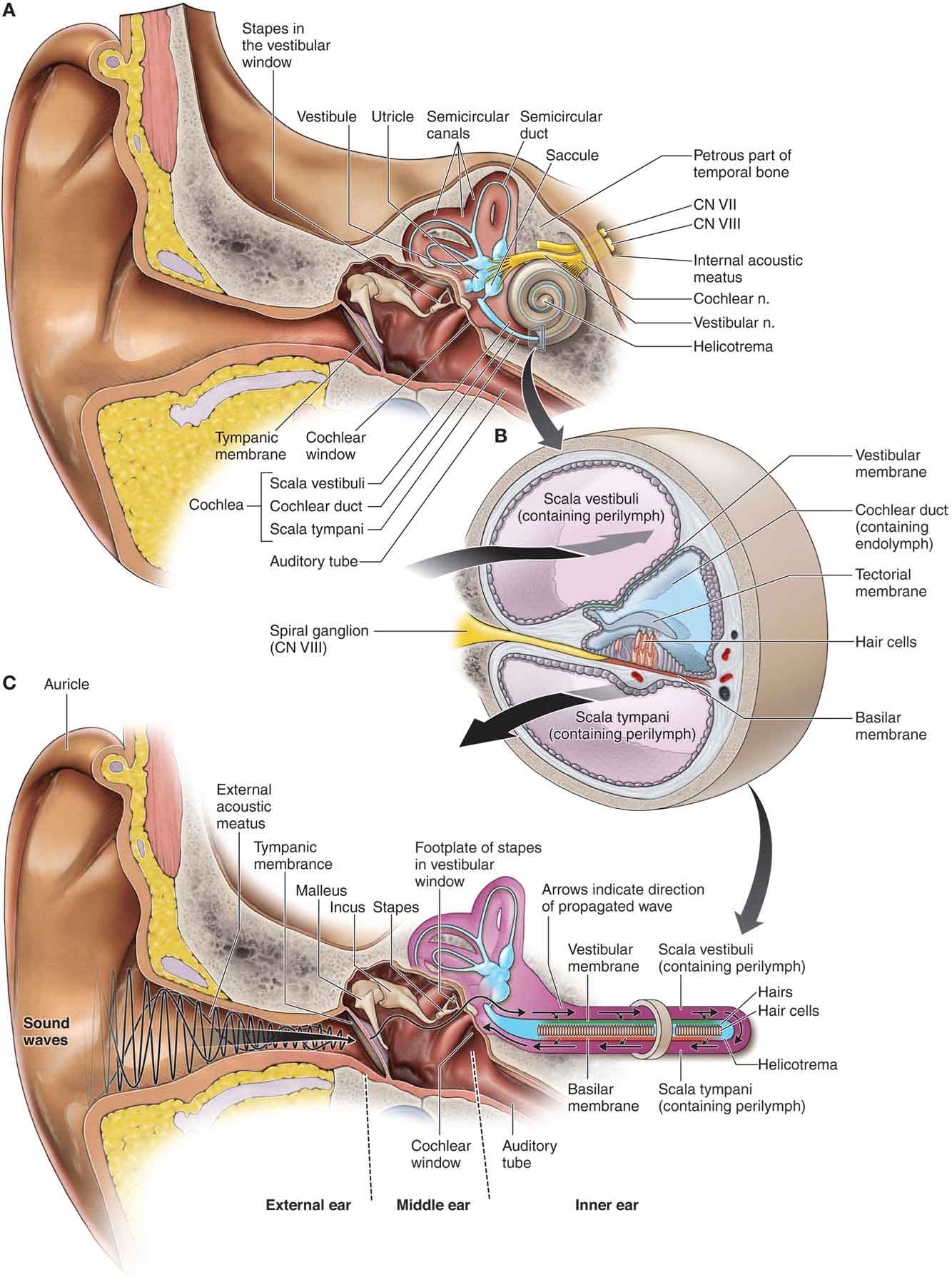
FIGURE 3-5 The apparatus of the inner ear. (Reproduced, with permission, from Chapter 19. Ear. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
A series of filaments line the basement membrane of the SCC and project into endolymph, which deforms these filaments when head motion occurs. This deformation is registered by receptor cells, and when sudden perturbations occur, the frequency of nerve impulses along the afferent nerve supply of the cell body is altered.
Unlike the filaments of the SCC, the filaments of the utricle and saccule do not project into endolymph but instead insert into a gelatinous mass, within which the otolith is embedded. Deformation of these filaments is produced by the weight of the otolith against the cilia, as the gelatinous mass is displaced during head movement.
The otoliths are responsible for providing information about gravitational forces, as well as vertical and horizontal motion. The filaments of the saccule also provide information about vertical motion. At rest, the endolymphatic fluid, or the gelatinous membrane, is stationary. When motion of the head occurs, the endolymphatic fluid, or the gelatinous membrane, initially remains stationary because of its inertia, while the canals move. This relative motion produces a dragging effect on the filaments and either increases or decreases the discharge rate, depending on the direction of shear. At the end of the head movement, the fluid and the membrane continue to move, and the cilia are now dragged in the opposite direction before coming to rest. In essence, the SCC receptors transmit a positive signal when movement begins, no signal when the motion has finished, and a normal level after the sensory cell has returned to its original position. As this occurs, other sensory cells orientated in the opposite direction react in the reverse fashion.
 CN IX (glossopharyngeal). The glossopharyngeal nerve (Fig. 3-4) contains a somatic motor, visceral efferent, visceral sensory, and somatic sensory fiber. The motor fibers originate in the nucleus ambiguous, leaving the lateral medulla to join the sensory nerve, which arises from cells in the superior and petrous ganglia. The glossopharyngeal nerve exits the skull through the jugular foramen and serves some functions, including supplying taste fibers for the posterior third of the tongue.
CN IX (glossopharyngeal). The glossopharyngeal nerve (Fig. 3-4) contains a somatic motor, visceral efferent, visceral sensory, and somatic sensory fiber. The motor fibers originate in the nucleus ambiguous, leaving the lateral medulla to join the sensory nerve, which arises from cells in the superior and petrous ganglia. The glossopharyngeal nerve exits the skull through the jugular foramen and serves some functions, including supplying taste fibers for the posterior third of the tongue.
 CN X (vagus). The functions of the vagus nerve (Fig. 3-4) are numerous and include the motor parasympathetic fibers to all the organs except the suprarenal (adrenal) glands, from its origin down to the second segment of the transverse colon. The vagus also controls some skeletal muscles, including:
CN X (vagus). The functions of the vagus nerve (Fig. 3-4) are numerous and include the motor parasympathetic fibers to all the organs except the suprarenal (adrenal) glands, from its origin down to the second segment of the transverse colon. The vagus also controls some skeletal muscles, including:
 Cricothyroid muscle
Cricothyroid muscle
 Levator veli palatini muscle
Levator veli palatini muscle
 Salpingopharyngeus muscle
Salpingopharyngeus muscle
 Palatoglossus muscle
Palatoglossus muscle
 Palatopharyngeus muscle
Palatopharyngeus muscle
 Superior, middle, and inferior pharyngeal constrictors
Superior, middle, and inferior pharyngeal constrictors
 Muscles of the larynx
Muscles of the larynx
The vagus nerve is thus responsible for such varied tasks as heart rate, gastrointestinal peristalsis, sweating, speech, and breathing. It also has some afferent fibers that innervate the inner (canal) portion of the outer ear.
 CN XI (accessory). The accessory nerve consists of a cranial component and a spinal component. The cranial root originates in the nucleus ambiguous and is often viewed as an aberrant portion of the vagus nerve. The spinal portion of the nerve arises from the lateral parts of the anterior horns of the first five or six cervical cord segments and ascends through the foramen magnum. The spinal portion of the accessory nerve supplies the sternocleidomastoid (SCM) and the trapezius muscles (Fig. 3-4).
CN XI (accessory). The accessory nerve consists of a cranial component and a spinal component. The cranial root originates in the nucleus ambiguous and is often viewed as an aberrant portion of the vagus nerve. The spinal portion of the nerve arises from the lateral parts of the anterior horns of the first five or six cervical cord segments and ascends through the foramen magnum. The spinal portion of the accessory nerve supplies the sternocleidomastoid (SCM) and the trapezius muscles (Fig. 3-4).
 CN XII (hypoglossal). The hypoglossal nerve is the motor nerve of the tongue, innervating the ipsilateral side of the tongue (Fig. 3-4) as well as forming the descendens hypoglossi, which anastomoses with other cervical branches to form the ansa hypoglossi. The latter, in turn, innervates the infrahyoid muscles.
CN XII (hypoglossal). The hypoglossal nerve is the motor nerve of the tongue, innervating the ipsilateral side of the tongue (Fig. 3-4) as well as forming the descendens hypoglossi, which anastomoses with other cervical branches to form the ansa hypoglossi. The latter, in turn, innervates the infrahyoid muscles.
Spinal Nerves
There are a total of 31 symmetrically arranged pairs of spinal nerves, each derived from the spinal cord.14 The spinal nerves are divided topographically into eight cervical pairs (C1–8), 12 thoracic pairs (T1–12), five lumbar pairs (L1–5), five sacral pairs (S1–5), and a coccygeal pair (Fig. 3-2A).
The posterior (dorsal) and anterior (ventral) roots of the spinal nerves are located within the vertebral canal (Fig. 3-2B). The portion of the spinal nerve that is not within the vertebral canal, and that usually occupies the intervertebral foramen, is referred to as a peripheral nerve. As the nerve roots begin to exit the vertebral canal, they must penetrate the dura mater before passing through dural sleeves within the intervertebral foramen. The dural sleeves are continuous with the epineurium of the nerves.
Essentially, there are four branches, or rami, of spinal nerves:4
 Posterior (dorsal) rami (Fig. 3-2B). This type usually consists of a medial sensory branch and a lateral motor branch.
Posterior (dorsal) rami (Fig. 3-2B). This type usually consists of a medial sensory branch and a lateral motor branch.
 Anterior (ventral) rami (Fig. 3-3B). Except in the thoracic nerves T2–12, the anterior (ventral) rami of the spinal nerves combine and then split again into networks of nerves referred to as nerve plexuses. There are four plexuses of spinal nerves: the cervical, brachial, lumbar, and sacral.
Anterior (ventral) rami (Fig. 3-3B). Except in the thoracic nerves T2–12, the anterior (ventral) rami of the spinal nerves combine and then split again into networks of nerves referred to as nerve plexuses. There are four plexuses of spinal nerves: the cervical, brachial, lumbar, and sacral.
 Communicating rami. The rami serve as a connection between the spinal nerves and the sympathetic trunk. Only the thoracic and upper lumbar nerves contain a white ramus communicans, but the gray ramus is present in all spinal nerves.
Communicating rami. The rami serve as a connection between the spinal nerves and the sympathetic trunk. Only the thoracic and upper lumbar nerves contain a white ramus communicans, but the gray ramus is present in all spinal nerves.
 Meningeal or recurrent meningeal (also known as sinuvertebral). These nerves carry sensory and vasomotor innervation to the meninges.
Meningeal or recurrent meningeal (also known as sinuvertebral). These nerves carry sensory and vasomotor innervation to the meninges.
There are three functional types of nerve fibers in the major nerve trunks, which vary in quantity depending on the particular nerve: afferent (sensory), autonomic (visceral efferent) (see PNS: Autonomic Nervous System [ANS]), and motor (somatic efferent) (Table 3-1). The faster nerve fibers such as A delta fibers are more concerned with speed and quality of human movement whereas the C fibers conduct far more slowly and are more involved with nociception and, by the compounds they release, the health of surrounding tissue.3
TABLE 3-1 | Classification of Afferent, Cutaneous, and Efferents |
Type | Conduction Velocity (m/s) | Function |
Afferents | ||
I | 70–120 | Provide input from muscle and tendon receptors |
II | 36–72 | Afferents from muscle spindles |
III | 27–68 | Pressure/nociceptive afferents from joints and aponeuroses |
IV | 1–4 | Pain |
Cutaneous | ||
Aα, β | 30–70 | Tactile receptors |
Aδ | 12–30 | Cold; fast nociception |
C | 0.5–1.0 | Warmth; tissue damage nociception |
Efferents | ||
α | 60–100 | Extrafusal muscle fibers |
γ | 10–30 | Intrafusal muscle fibers |
B | 3–30 | Preganglionic autonomic |
C | 0.5–2.0 | Postganglionic autonomic |
Afferent (sensory) Nerves. The sensory nerves carry afferents (a nerve conveying impulses from the periphery to the CNS) from a portion of the skin. They also carry efferents (a nerve conveying impulses from the CNS to the periphery) to the skin structures. When a sensory nerve is compressed, symptoms occur in the area of the nerve distribution. This area of distribution, called a dermatome, is a well-defined portion of the skin and follows the segmental distribution of the underlying muscle innervation. Figure 3-6 is an approximate representation of the various dermatomes on the anterior trunk for the cervical and thoracic nerves. However, there is no current, diagnostically accurate dermatome illustration, and so it is important to remember that textbooks commonly used in medical and allied health programs contain multiple, conflicting dermatome maps, most of which rely on research from the 1940s.15 For example, the majority show the cutaneous distribution of the fourth lumbar spinal nerve (L4 dermatome) either running from the lateral aspect of the thigh to the medial side of the great toe, or confined to the medial portion of the leg distal to the knee.15 Consequently, any diagnosis related to the integrity of the nervous system cannot be based on sensory testing alone.
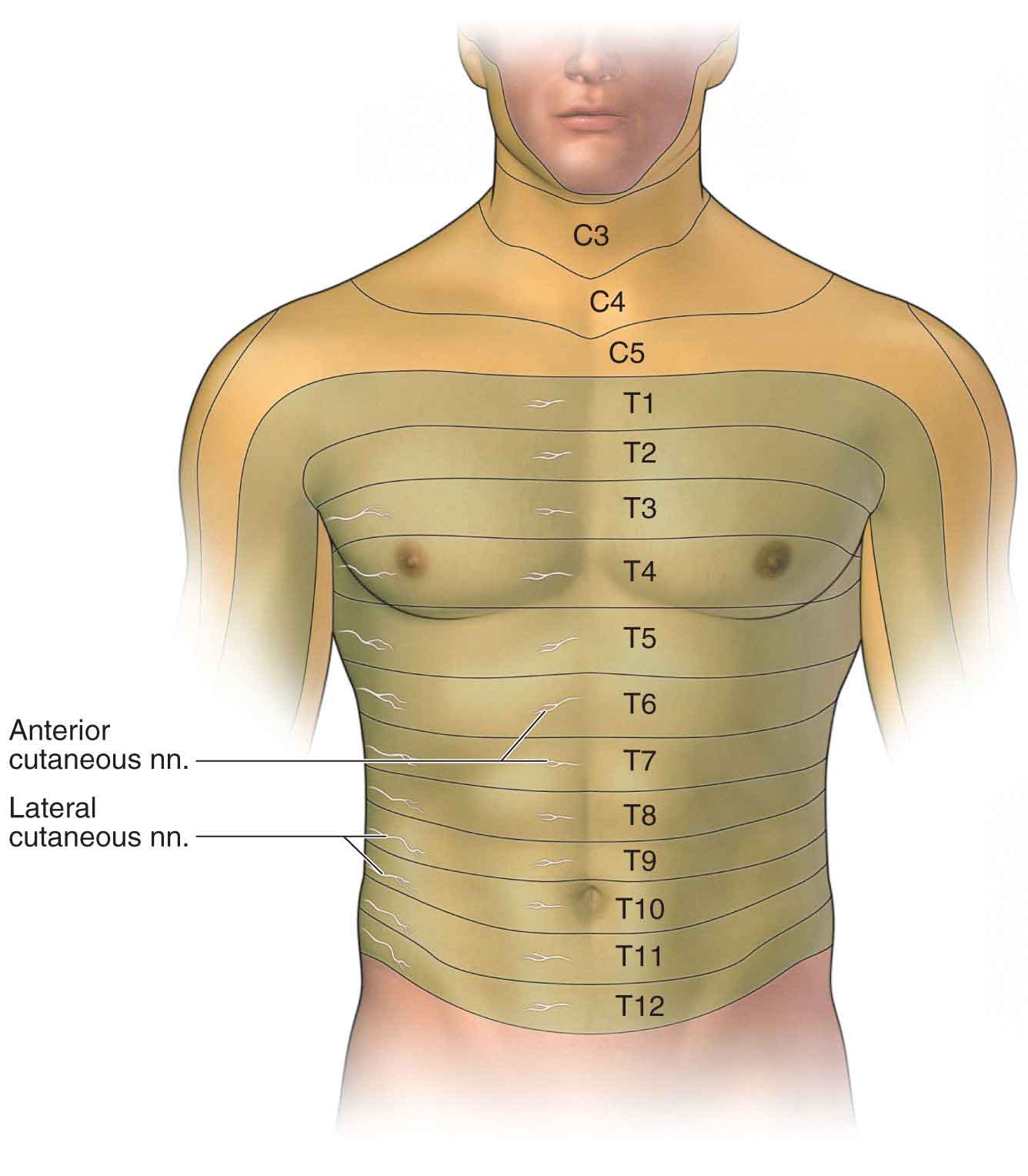
FIGURE 3-6 Segmental distribution of the body. (Reproduced, with permission, from Chapter 2. Anterior Thoracic Wall. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
Examples of sensory nerves in the body are the lateral (femoral) cutaneous nerve (LCN) of the thigh (Fig. 3-7), the saphenous nerve, and the interdigital nerves (see later).
FIGURE 3-7 LCN of the thigh.
Motor (somatic efferent). The motor nerves carry efferents to muscles and return sensation from muscles and associated ligamentous structures. Any nerve that innervates a muscle also mediates the sensation from the joint upon which that muscle acts. Examples of a motor nerve include the suprascapular nerve and the posterior (dorsal) scapular nerve. A hierarchical recruitment pattern exists in the nervous system for muscle recruitment called the law of parsimony.18 The law of parsimony states that the nervous system tends to activate the fewest muscles or muscle fibers possible for the control of a given joint action. This hierarchical pattern of muscle recruitment makes practical sense from an energy perspective.18
Cervical Plexus
The eight pairs of cervical nerves are derived from cord segments between the level of the foramen magnum and the middle of the seventh cervical vertebra.19 The spinal nerves from C3 to C7, exiting from the intervertebral foramen, divide into a larger anterior (ventral) ramus and a smaller posterior (dorsal) ramus. The anterior (ventral) ramus of the cervical spinal nerve travels on the transverse process in an anterior–lateral direction to form the cervical plexus and brachial plexus. The posterior (dorsal) ramus of the spinal nerve runs posteriorly around the superior articular process, supplying the facet (zygapophyseal) joint, ligaments, deep muscles, and skin of the posterior aspect of the neck.9
Each nerve joins with a gray communicating ramus from the sympathetic trunk and sends a small, recurrent meningeal branch back into the spinal canal to supply the dura with sensory and vasomotor innervation. It also branches into anterior and posterior primary divisions, which are mixed nerves that pass to their respective peripheral distributions. The motor branches carry a few sensory fibers that convey proprioceptive impulses from the neck muscles.
 Posterior primary divisions. The C1 (suboccipital) nerve is the only branch of the first posterior primary divisions. It is a motor nerve, serving the muscles of the suboccipital triangle, with very few, if any, sensory fibers.19
Posterior primary divisions. The C1 (suboccipital) nerve is the only branch of the first posterior primary divisions. It is a motor nerve, serving the muscles of the suboccipital triangle, with very few, if any, sensory fibers.19
 Anterior primary divisions. The cervical plexus is formed by the anterior primary divisions of the first four cervical nerves (C1–4) (Fig. 3-8).
Anterior primary divisions. The cervical plexus is formed by the anterior primary divisions of the first four cervical nerves (C1–4) (Fig. 3-8).
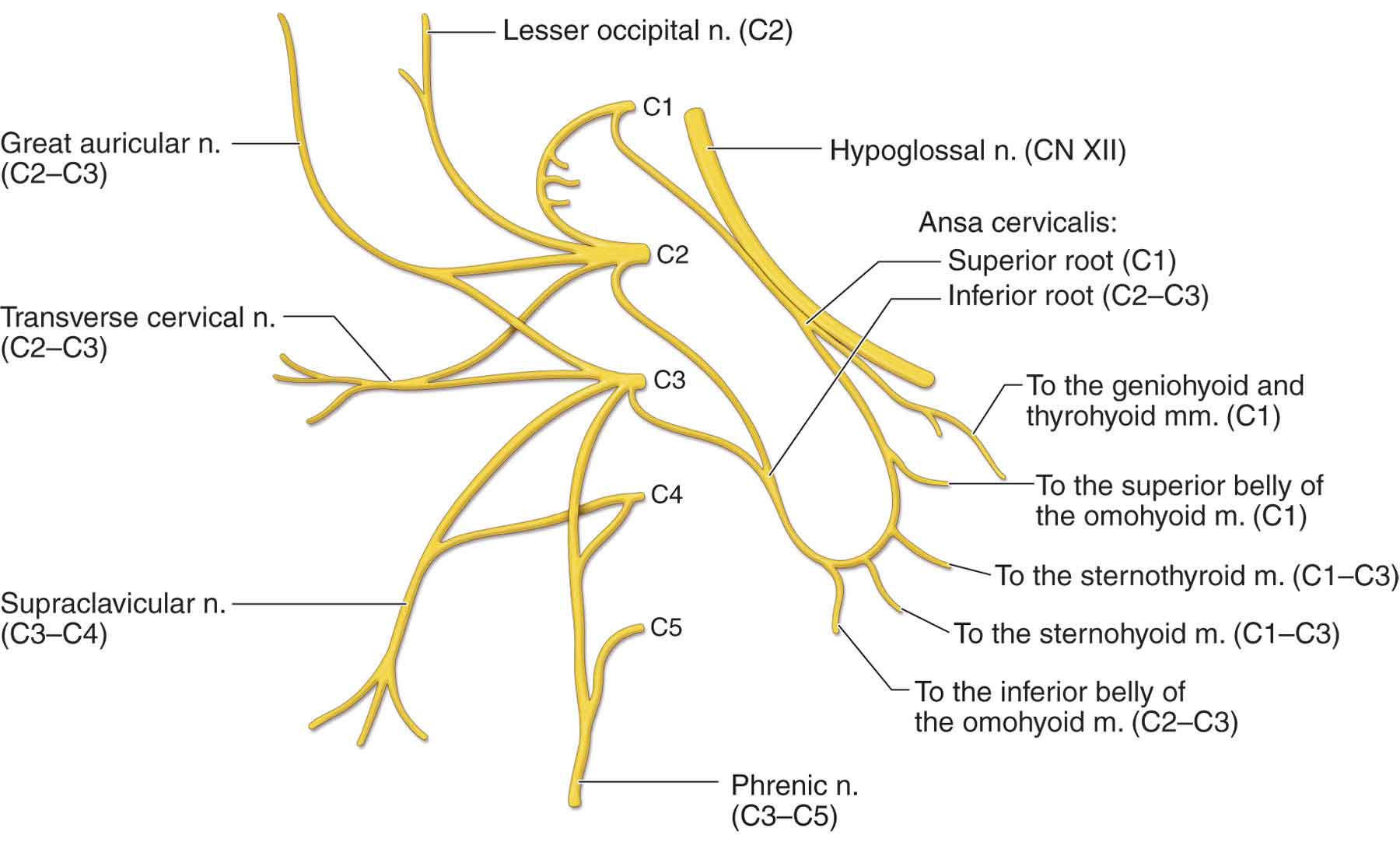
FIGURE 3-8 The cervical plexus. (Reproduced, with permission, from Chapter 25. Overview of the Neck. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
 Cervical plexus (C1–4).
Cervical plexus (C1–4).
 Sensory branches (see Fig. 3-6).
Sensory branches (see Fig. 3-6).
 Small occipital nerve (C2, 3). This nerve (Fig. 3-8) supplies the skin of the lateral occipital portion of the scalp, the upper median part of the auricle, and the area over the mastoid process.19
Small occipital nerve (C2, 3). This nerve (Fig. 3-8) supplies the skin of the lateral occipital portion of the scalp, the upper median part of the auricle, and the area over the mastoid process.19
 Great auricular nerve (C2, 3). This nerve (Fig. 3-8) supplies sensation to the ear and the face via the ascending ramus of the mandible. The nerve lies on or just below the deep layer of the investing fascia of the neck. It arises from the anterior rami of the second and third cervical nerves and emerges from behind the sternocleidomastoid (SCM) muscle, before ascending on it to cross over the parotid gland.
Great auricular nerve (C2, 3). This nerve (Fig. 3-8) supplies sensation to the ear and the face via the ascending ramus of the mandible. The nerve lies on or just below the deep layer of the investing fascia of the neck. It arises from the anterior rami of the second and third cervical nerves and emerges from behind the sternocleidomastoid (SCM) muscle, before ascending on it to cross over the parotid gland.
 Cervical cutaneous nerve (cutaneous coli) (C2, 3). This nerve supplies the skin over the anterior portion of the neck.
Cervical cutaneous nerve (cutaneous coli) (C2, 3). This nerve supplies the skin over the anterior portion of the neck.
 Supraclavicular branches (C3, 4). These nerves supply the skin over the clavicle and the upper deltoid and pectoral regions, as low as the third rib.
Supraclavicular branches (C3, 4). These nerves supply the skin over the clavicle and the upper deltoid and pectoral regions, as low as the third rib.
 Communicating branches. The ansa cervicalis nerve (Fig. 3-8) is formed by the junction of two main nerve roots, derived entirely from anterior (ventral) cervical rami. A loop is formed at the point of their anastomosis, and sensory fibers are carried to the dura of the posterior fossa of the skull via the recurrent meningeal branch of the hypoglossal nerve. The communication with the vagus nerve from C1 is of undetermined function.
Communicating branches. The ansa cervicalis nerve (Fig. 3-8) is formed by the junction of two main nerve roots, derived entirely from anterior (ventral) cervical rami. A loop is formed at the point of their anastomosis, and sensory fibers are carried to the dura of the posterior fossa of the skull via the recurrent meningeal branch of the hypoglossal nerve. The communication with the vagus nerve from C1 is of undetermined function.
 Muscular branches. Communication with the hypoglossal nerve from C1 to C2 (Fig. 3-8) carries motor fibers to the geniohyoid and thyrohyoid muscles and to the sternohyoid and sternothyroid muscles by way of the superior root of the ansa cervicalis (Fig. 3-8). The nerve to the thyrohyoid branches from the hypoglossal nerve and runs obliquely across the hyoid bone to innervate the thyrohyoid. The nerve to the superior belly of the omohyoid branches from the superior root (Fig. 3-8) and enters the muscle at a level between the thyroid notch, and a horizontal plane 2-cm inferior to the notch. The nerves to the sternohyoid and sternothyroid share a common trunk, which branches from the loop (Fig. 3-8). The nerve to the inferior belly of the omohyoid also branches from the loop (Fig. 3-8). The loop is most frequently located just deep to the site where the superior belly (or tendon) of the omohyoid muscle crosses the internal jugular vein. There is a branch to the SCM muscle from C2, and there are branches to the trapezius muscle (C3–4) via the subtrapezial plexus.
Muscular branches. Communication with the hypoglossal nerve from C1 to C2 (Fig. 3-8) carries motor fibers to the geniohyoid and thyrohyoid muscles and to the sternohyoid and sternothyroid muscles by way of the superior root of the ansa cervicalis (Fig. 3-8). The nerve to the thyrohyoid branches from the hypoglossal nerve and runs obliquely across the hyoid bone to innervate the thyrohyoid. The nerve to the superior belly of the omohyoid branches from the superior root (Fig. 3-8) and enters the muscle at a level between the thyroid notch, and a horizontal plane 2-cm inferior to the notch. The nerves to the sternohyoid and sternothyroid share a common trunk, which branches from the loop (Fig. 3-8). The nerve to the inferior belly of the omohyoid also branches from the loop (Fig. 3-8). The loop is most frequently located just deep to the site where the superior belly (or tendon) of the omohyoid muscle crosses the internal jugular vein. There is a branch to the SCM muscle from C2, and there are branches to the trapezius muscle (C3–4) via the subtrapezial plexus.
Smaller branches to the adjacent vertebral musculature supply the rectus capitis lateralis and rectus capitis anterior (C1), the longus capitis (C2, 4) and longus colli (C1–4), the scalenus medius (C3, 4) and scalenus anterior (C4), and the levator scapulae (C3–5).
The phrenic nerve (C3–5) (see Fig. 3-8) passes obliquely over the scalenus anterior muscle and between the subclavian artery and the vein to enter the thorax behind the sternoclavicular (S-C) joint, where it descends vertically through the superior and middle mediastinum to the diaphragm.19 Motor branches supply the diaphragm. Sensory branches supply the pericardium, the diaphragm, and part of the costal and mediastinal pleurae.
Phrenic nerve involvement has been described in several neuropathies, including critical illness, polyneuropathy, Guillain–Barré syndrome, brachial neuritis, and hereditary motor and sensory neuropathy type 1.26,27 The symptoms depend largely on the degree of involvement, and whether one or both of the nerves are involved.19
 Unilateral paralysis of the diaphragm causes few or no symptoms except with heavy exertion.
Unilateral paralysis of the diaphragm causes few or no symptoms except with heavy exertion.
 Bilateral paralysis of the diaphragm is characterized by dyspnea upon the slightest exertion and difficulty with coughing and sneezing.26,27
Bilateral paralysis of the diaphragm is characterized by dyspnea upon the slightest exertion and difficulty with coughing and sneezing.26,27
 Phrenic neuralgia, which can result from neck tumors, aortic aneurysm, and pericardial or other mediastinal infections, is characterized by pain near the free border of the ribs, beneath the clavicle, and deep in the neck.26,27
Phrenic neuralgia, which can result from neck tumors, aortic aneurysm, and pericardial or other mediastinal infections, is characterized by pain near the free border of the ribs, beneath the clavicle, and deep in the neck.26,27
Brachial Plexus
The brachial plexus (Fig. 3-9) arises from the anterior divisions of the fifth cervical through the first thoracic nerve roots, with occasional contributions from the fourth cervical and the second thoracic roots. The roots of the plexus, which consist of C5 and C6, join to form the superior (upper) trunk; C7 becomes the middle trunk, and C8 and T1 join to form the inferior (lower) trunks. Each of the trunks divides into anterior and posterior divisions, which then form cords (Fig. 3-9). The anterior divisions of the upper and middle trunk form the lateral cord, the anterior division of the inferior (lower) trunk forms the medial cord, and all three posterior divisions unite to form the posterior cord. The three cords, named for their relationship to the axillary artery, split to form the main branches of the plexus. These branches give rise to the peripheral nerves: musculocutaneous (lateral cord), axillary and radial (posterior cord), ulnar (medial cord), and median (medial and lateral cords).28 Numerous smaller nerves arise from the roots, the trunks, and the cords of the plexus. Peripheral nerve injuries of the upper extremity and their respective clinical findings are listed in Table 3-2.
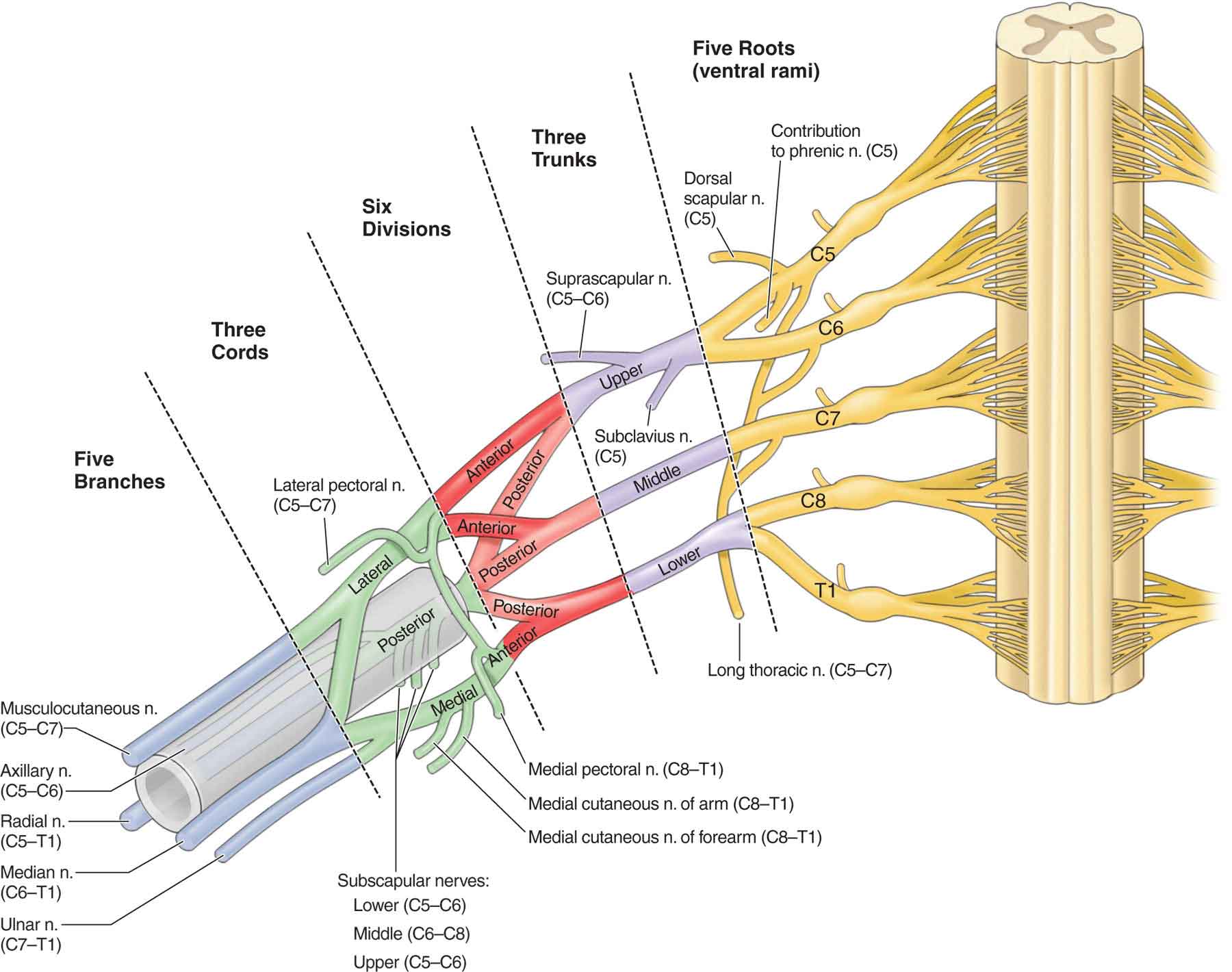
FIGURE 3-9 The brachial plexus. (Reproduced, with permission, from Chapter 29. Overview of the Upper Limb. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.)
TABLE 3-2 | Peripheral Nerve Injury Related to the Cords of the Brachial Plexus |
Cord and Nerve | Level of Injury | Motor Loss | Cutaneous Loss | |
Posterior cord: radial (C5–T1) | Plexus—proximal to axillary nerve Axilla (brachioaxillary angle) Spiral groove Proximal to lateral epicondyle Arcade of Frohse | All muscles innervated by radial nerve All muscles innervated by axillary nerve Triceps (medial and lateral heads) and anconeus All muscles innervated by radial nerve except medial head of triceps Brachialis, brachioradialis, ECRL, and ECRB Supinator, all muscles innervated by posterior interosseous nerve | Throughout radial and axillary distribution Posterior brachial cutaneous Posterior antebrachial cutaneous Superficial radial (Wartenberg syndrome) | |
Posterior cord: axillary nerve (C5–6) | Axilla (quadrangular space) | Teres minor and deltoid | Lateral arm | |
Medial and lateral cord: median nerve (C5–T1) | Plexus (proximal to the joining of the medial and lateral cords)—thoracic outlet syndrome Ligament of Struthers—proximal to medial epicondyle Cubital fossa exit—between two heads of the pronator teres Forearm | All muscles innervated by median, musculocutaneous, and ulnar nerves Pronator teres Pronator teres, FCR, FDS, PL, and lumbricales I and II Anterior interosseous: FDP (I and II), FPL, and PQ median muscular branch: Thenar muscles (APB, FPB, and OP) and lumbricales I and II | Throughout median, musculocutaneous, and ulnar distributions Palmar branch: radial half of thumb Digital branch: posterior (dorsal) tips of thumb, index, and middle finger and radial half of ring finger | |
Lateral: musculocutaneous nerve (C5–7) | Coracobrachialis Elbow Cubital tunnel Between the two heads of the FCU Proximal to wrist Guyon canal | Coracobrachialis Biceps Brachialis FCU, FDP, adductor pollicis, lumbricales, and interossei FDP and FCU Deep branch: all hand muscles innervated by the ulnar nerve Superficial branch: Palmaris brevis Muscles of the hypothenar eminence (hand of benediction) and interossei | Lateral antebrachial cutaneous nerve: Lateral forearm Posterior and anterior aspects on the ulnar side of the hand Medial: ulnar nerve (C8–T1) Ulnar aspect of the hand | |
ECRL, extensor carpi radialis longus; ECRB, extensor carpi radialis brevis; FCR, flexor carpi radialis; FDS, flexor digitorum superficials; PL, palmaris longus; FDP, flexor digitorum profundus; FPL, flexor pollicis longus; PQ, pronator quadratus; APB, abductor pollicis brevis; FPB, flexor pollicis brevis; OP, opponens pollicis; FCU, flexor carpi ulnaris; DIP, distal interphalangeal. | ||||
From the Roots
 The origin of the posterior (dorsal) scapular nerve (C5) frequently shares a common trunk with the long thoracic nerve (Fig. 3-9). The former passes through the scalenus medius anterior internally, and scalenus posterior laterally, with the presence of some tendinous tissues. Leaving the long thoracic nerve, it often gives branches to the shoulder and the subaxillary region, before the branches join the long thoracic nerve again. The posterior (dorsal) scapular nerve supplies the rhomboids and the levator scapulae muscles. Posterior (dorsal) scapular nerve lesions can result from a forward posture of the head and the neck as this position increases tension in the anterior cervical spine, producing the potential for hypertonicity and hypertrophy of the medial scalene.29 The chief complaint is usually one of scapular pain radiating to the lateral shoulder and arm.
The origin of the posterior (dorsal) scapular nerve (C5) frequently shares a common trunk with the long thoracic nerve (Fig. 3-9). The former passes through the scalenus medius anterior internally, and scalenus posterior laterally, with the presence of some tendinous tissues. Leaving the long thoracic nerve, it often gives branches to the shoulder and the subaxillary region, before the branches join the long thoracic nerve again. The posterior (dorsal) scapular nerve supplies the rhomboids and the levator scapulae muscles. Posterior (dorsal) scapular nerve lesions can result from a forward posture of the head and the neck as this position increases tension in the anterior cervical spine, producing the potential for hypertonicity and hypertrophy of the medial scalene.29 The chief complaint is usually one of scapular pain radiating to the lateral shoulder and arm.
 A small branch from C5 passes to the phrenic nerve.
A small branch from C5 passes to the phrenic nerve.
 Smaller branches from C6 to C8 extend to the scaleni and longus colli muscles.
Smaller branches from C6 to C8 extend to the scaleni and longus colli muscles.
 The first intercostal nerve extends from T1.
The first intercostal nerve extends from T1.
 The long thoracic nerve (C5–7) is purely a motor nerve that originates from the anterior (ventral) rami of the fifth, the sixth, and the seventh cervical roots (Fig. 3-9). It is the sole innervation to the serratus anterior muscle. The fifth and the sixth cervical roots, along with the posterior (dorsal) scapular nerve, pass through the scalenus medius muscle, whereas the seventh cervical root passes anterior to it.30 The nerve then travels beneath the brachial plexus and clavicle to pass over the first rib. From there, it descends along the lateral aspect of the chest wall, where it innervates the serratus anterior muscle. The long thoracic nerve extends as far inferior as the eighth or the ninth rib. Its long and relatively superficial course makes it susceptible to injury from any of the following causes.10,31–33
The long thoracic nerve (C5–7) is purely a motor nerve that originates from the anterior (ventral) rami of the fifth, the sixth, and the seventh cervical roots (Fig. 3-9). It is the sole innervation to the serratus anterior muscle. The fifth and the sixth cervical roots, along with the posterior (dorsal) scapular nerve, pass through the scalenus medius muscle, whereas the seventh cervical root passes anterior to it.30 The nerve then travels beneath the brachial plexus and clavicle to pass over the first rib. From there, it descends along the lateral aspect of the chest wall, where it innervates the serratus anterior muscle. The long thoracic nerve extends as far inferior as the eighth or the ninth rib. Its long and relatively superficial course makes it susceptible to injury from any of the following causes.10,31–33
 Entrapment of the fifth and sixth cervical roots, as they pass through the scalenus medius muscle.
Entrapment of the fifth and sixth cervical roots, as they pass through the scalenus medius muscle.
 Compression of the nerve during traction to the upper extremity by the undersurface of the scapula, as the nerve crosses over the second rib.
Compression of the nerve during traction to the upper extremity by the undersurface of the scapula, as the nerve crosses over the second rib.
 Compression and traction to the nerve by the inferior angle of the scapula during general anesthesia, or with vigorous passive abduction of the arm.
Compression and traction to the nerve by the inferior angle of the scapula during general anesthesia, or with vigorous passive abduction of the arm.
 Lesions of the long thoracic nerve are common and are the single most common peripheral nerve lesion at the shoulder. The most common cause of long thoracic nerve injury results from carrying a heavy object on the shoulder. Other causes include postinfection, postinjection, postpartum, and postoperative origins.34 Similar to other peripheral nerve injuries, trauma to the nerve can be caused by a direct blow or a traction force to the nerve. The traction injury can occur when concurrent head rotation away, side bending away, and neck flexion, are coupled with the arm positioned overhead.35–37 Other mechanisms that have been attributed to long thoracic nerve dysfunction include lifting weights overhead, driving a golf ball, and serving a tennis ball.38
Lesions of the long thoracic nerve are common and are the single most common peripheral nerve lesion at the shoulder. The most common cause of long thoracic nerve injury results from carrying a heavy object on the shoulder. Other causes include postinfection, postinjection, postpartum, and postoperative origins.34 Similar to other peripheral nerve injuries, trauma to the nerve can be caused by a direct blow or a traction force to the nerve. The traction injury can occur when concurrent head rotation away, side bending away, and neck flexion, are coupled with the arm positioned overhead.35–37 Other mechanisms that have been attributed to long thoracic nerve dysfunction include lifting weights overhead, driving a golf ball, and serving a tennis ball.38
The typical clinical presentation includes the following:
 Vague pain in the neck and the scapula region.
Vague pain in the neck and the scapula region.
 An inability to fully elevate the arm overhead.
An inability to fully elevate the arm overhead.
 Shoulder flexion and abduction are weak and limited in AROM due to the loss of the trapezius–serratus anterior force couple (see Chapter 16). The clinician should note the presence of winging of the scapula when testing the serratus anterior.
Shoulder flexion and abduction are weak and limited in AROM due to the loss of the trapezius–serratus anterior force couple (see Chapter 16). The clinician should note the presence of winging of the scapula when testing the serratus anterior.
Conservative intervention includes protection of the serratus anterior with a brace or restraint,34,37,39 galvanic stimulation to the serratus anterior, muscle taping,40 strengthening exercises for the rhomboids, pectoralis, trapezius, and serratus anterior muscles.35,38,41 The average rate of return ranges from 3–7 months30,34 to 2 years.38
From the Trunks
A nerve extends to the subclavius muscle (C5–6) from the superior (upper) trunk, or fifth root. The subclavius muscle acts mainly on the stability of the S-C joint, with more or less intensity, according to the degree of the clavicular interaction with the movements of the peripheral parts of the superior limb, and seems to act as a substitute for the ligaments of the S-C joint.42
The suprascapular nerve originates from the superior (upper) trunk of the brachial plexus formed by the roots of C5 and C6 (see Fig. 3-9) at Erb’s point.
From the Cords
 The medial and lateral pectoral nerves extend from the medial and lateral cords, respectively (see Fig. 3-9). They supply the pectoralis major and pectoralis minor muscles. The pectoralis major muscle has dual innervation.43 The lateral pectoral nerve (C5–7), which is more medial in the muscle, travels with the thoracoacromial vessels, and innervates the clavicular and sternal heads. The medial pectoral nerve (C8–T1) shares a course with the lateral thoracic vessels and provides innervation to the sternal and costal heads.44 The main trunk of these nerves can be found near the origin of the vascular supply of the muscle.
The medial and lateral pectoral nerves extend from the medial and lateral cords, respectively (see Fig. 3-9). They supply the pectoralis major and pectoralis minor muscles. The pectoralis major muscle has dual innervation.43 The lateral pectoral nerve (C5–7), which is more medial in the muscle, travels with the thoracoacromial vessels, and innervates the clavicular and sternal heads. The medial pectoral nerve (C8–T1) shares a course with the lateral thoracic vessels and provides innervation to the sternal and costal heads.44 The main trunk of these nerves can be found near the origin of the vascular supply of the muscle.
 The three subscapular nerves from the posterior cord consist of:
The three subscapular nerves from the posterior cord consist of:
 The upper subscapular nerve (C5–6), which supplies the subscapularis muscle (see Fig. 3-9).
The upper subscapular nerve (C5–6), which supplies the subscapularis muscle (see Fig. 3-9).
 The thoracodorsal nerve, or middle subscapular nerve, which arises from the posterior cord of the brachial plexus with its motor fiber contributions from C6, C7, and C8 (see Fig. 3-9). This nerve courses along the posterior–lateral chest wall, along the surface of the serratus anterior, and deep to the subscapularis, giving rise to branches that supply the latissimus dorsi.
The thoracodorsal nerve, or middle subscapular nerve, which arises from the posterior cord of the brachial plexus with its motor fiber contributions from C6, C7, and C8 (see Fig. 3-9). This nerve courses along the posterior–lateral chest wall, along the surface of the serratus anterior, and deep to the subscapularis, giving rise to branches that supply the latissimus dorsi.
 The lower subscapular nerve (C5–6) to the teres major and part of the subscapularis muscle (see Fig. 3-9).
The lower subscapular nerve (C5–6) to the teres major and part of the subscapularis muscle (see Fig. 3-9).
 Sensory branches of the medial cord (C8–T145,46 or T1 alone46) comprise the medial cutaneous (antebrachial) nerve to the medial surface of the forearm and the medial cutaneous (brachial) nerve to the medial surface of the arm (see Fig. 3-9).
Sensory branches of the medial cord (C8–T145,46 or T1 alone46) comprise the medial cutaneous (antebrachial) nerve to the medial surface of the forearm and the medial cutaneous (brachial) nerve to the medial surface of the arm (see Fig. 3-9).
Obstetric Brachial Plexus Lesions
The pathomorphologic spectrum of traumatic brachial plexus impairments most often includes combinations of various types of injuries: compression of spinal nerves, traction injuries of spinal roots and nerves, and avulsions of spinal roots.47 If the rootlets are traumatically disconnected from the spinal cord, they normally exit the intradural space; in rare cases, however, they may also remain within the dural space.
Brachial plexus injuries are most commonly seen in children and usually are caused by birth injuries, although more advanced birthing techniques and an increase in the number of Caesarian births in the United States are producing a decline in incidence. However, difficult deliveries continue to cause such lesions.48 Although multiple potential risk factors have been cited, including maternal demographic variables, diabetes, hypertension, prior Caesarian delivery, uterine abnormalities, induction of labor, prolonged second stage (adjusted by parity and epidural use), assisted vaginal delivery, and neonatal birth weight, no specific risk factor has been identified.49 Stretch (neurapraxia or axonotmesis) and incomplete rupture are more common in obstetric brachial plexus palsy than complete rupture or avulsion.
Obstetrical brachial plexus palsy is classified into upper (involving C5, C6, and usually C7 roots), lower (predominantly C8 and T1), and total (C5–C8 and T1) plexus palsies.50,51 Upper brachial plexus palsy, although described first by Duchenne,52 ironically bears the name Erb’s palsy.53 Most cases of obstetric brachial plexus palsy involve Erb’s palsy, and the lesion is always supraclavicular. Lower brachial plexus palsy is extremely rare in birth injuries46,54 and is referred to as Klumpke’s palsy.55
The infant with Erb’s palsy typically shows the classic “waiter’stip” posture of the paralyzed limb.56,57 The arm lies internally rotated at the side of the chest, the elbow is extended (paralysis of C5, 6) or slightly flexed (paralysis of C5–C7), the forearm is pronated, and the wrist and the fingers are flexed. This posture occurs because of paralysis and atrophy of the deltoid, biceps, brachialis, and brachioradialis muscles.58
Klumpke’s paralysis is characterized by paralysis and atrophy of the small hand muscles and flexors of the wrist (the so-called claw-hand). Prognosis of this type is more favorable. If the sympathetic rami of T1 are involved, Horner syndrome (ptosis, enophthalmos, facial reddening, and anhydrosis) may be present.
