Primary Malignant Bone Tumors
Peter S. Rose
Key Points
• Primary malignant tumors about the hip are rare.
• Early recognition is critical in curative treatment.
Introduction
Primary malignant processes frequently present in the hip region. Prompt recognition of these patients is critical, as a window for cure often exists. These lesions are treated with resection with or without adjuvant therapies, depending on histology. With few exceptions, wide surgical resection of all disease is required for long-term survival. Limb salvage procedures are possible in 85% to 90% of cases, and modern techniques give durable prosthetic reconstructions. This chapter will address the treatment of primary bone and soft tissue sarcomas about the hip.
Epidemiology
Although sarcomas can present in any age group, classic patterns have been observed and documented. Osteosarcoma is the most common bone sarcoma with the best characterized epidemiology. Approximately 500 cases per year arise in the United States. Although mutations in the retinoblastoma gene can lead to osteosarcoma, most cases arise sporadically.1
Data from more than 2000 cases of osteosarcoma treated over a 100 year period highlight the presentation of this tumor (Fig. 51-1). Males account for a modest majority of cases, and most patients are in the second or third decade of life. The hip region (including proximal femur and pelvis) is the second most common site of disease after the knee. The skeletal distribution of osteosarcoma roughly parallels the amount of skeletal growth at various sites.
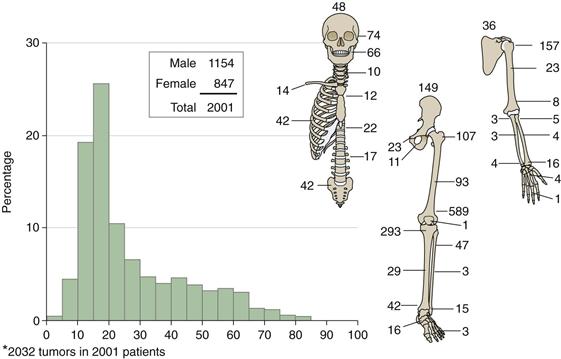
Figure 51-1 Age, sex, and skeletal distribution of osteosarcoma in 2001 patients.
Other sarcomas have similar distributions. Chondrosarcoma and Ewing’s sarcoma are the next most common primary bone tumors about the hip. Chondrosarcoma arises in an older patient population than osteosarcoma and most commonly involves the pelvis or proximal femur region. Ewing’s sarcoma shows a similar age distribution to osteosarcoma but has more tumors in the hip and pelvis region. Extra-abdominal soft tissue sarcomas present in middle-aged to older adult patients, with the thigh and buttock regions being the most frequent sites of disease. Although true bony invasion is rare, these tumors may encase the sciatic nerve or femoral artery and often abut the bone directly.
General Considerations
Staging and Biopsy
Patients presenting with a primary malignant tumor about the hip joint require accurate diagnosis and staging for proper management. Staging involves defining the local and systemic extent of disease. Local imaging begins with plain radiographs of the affected area (even for patients with soft tissue sarcomas). Patients with bony tumors should undergo imaging of the entire femur and pelvis to identify any discontiguous sites of disease (skip lesions). Magnetic resonance imaging is used to characterize marrow involvement and soft tissue masses or extensions. Select tumors may require computed tomography (CT) angiography to define vascular invasion or encasement.
The systemic extent of disease is evaluated with computed tomography of the chest and bone scintigraphy (for bony tumors) to complete oncologic staging. Although CT of the abdomen and pelvis is not part of routine oncologic staging for sarcomas, we employ this approach in any patient whose primary tumor involves the pelvis or extends above the inguinal ligament, to detect any regional metastases that might be missed with imaging of the chest and tumor site alone. The role of positron emission tomography (PET) is currently evolving in the staging evaluation of primary tumors. Select histologies (e.g., myxoid liposarcoma) may have specific additional staging protocols.2
Although many lesions have a characteristic appearance on imaging studies, biopsy is generally needed to establish diagnosis before malignant conditions are treated. We generally use image-guided (fluoroscopic, CT, or ultrasound) biopsy as the first-line invasive sampling method for tumors. High diagnostic accuracy is seen in the hands of experienced pathologists.3 Open biopsy is reserved for unsuccessful core needle biopsy. The biopsy approach, whether open or percutaneous, should be carefully planned to target high-yield areas of the lesion and to allow excision of the biopsy tract and any contaminated tissues at the time of definitive resection. Poorly planned biopsy can severely compromise ultimate patient outcomes.4 In the case of open biopsy, transverse incisions are never used, and care is taken to split individual muscles rather than open classic planes to minimize the zone of biopsy site contamination.
Adjuvant Therapy
Adjuvant chemotherapy and/or radiation therapy is the standard of care in high-grade primary bony malignancies. Chemotherapy improves the survival of patients with localized Ewing’s sarcoma and osteosarcoma from approximately 15% to approaching 70% or greater. Additionally, long-term disease-free survival is seen even in patients with limited metastatic disease treated with aggressive surgery and chemotherapy. Essentially all standard regimens for these tumors employ two to four cycles of chemotherapy preoperatively. Surgical resection is accomplished with a minimal delay in therapy, followed by multiple postoperative cycles.1,5
The critical nature of chemotherapy in these diseases highlights the importance of an accurate initial diagnosis before therapy commences. The chemotherapy response as assessed by the necrosis seen in the resected tumor specimen is predictive of overall survival. That said, up-front surgery followed by an equivalent total number of postoperative chemotherapy cycles yielded identical clinical outcomes in the only study that directly examined this.6
Radiotherapy may be used in the treatment of Ewing’s sarcoma and is commonly used in the treatment of soft tissue sarcoma. High-dose radiation can be used in place of surgery for local control in Ewing’s sarcoma, when resection is not possible or practical. Although no prospective randomized studies have compared surgery versus radiotherapy for local control in Ewing’s sarcoma, available data appear to indicate that surgery or surgery combined with radiation may be superior to radiation alone.7,8 Additionally, long-term complications are seen following radiation for Ewing’s sarcoma.9
Local control of soft tissue sarcomas is improved with the use of radiotherapy. Radiation for soft tissue sarcomas may be administered preoperatively or postoperatively and has been examined in a prospective randomized study. Preoperative therapy engenders a higher wound complication rate but allows a lower dose to be administered to a smaller volume of tissue.10 Although initial wound complications are more frequent with preoperative therapy, long-term limb function appears better as the result of less ultimate edema and fibrosis.11 The use of preoperative versus postoperative radiotherapy is often influenced by institutional practice patterns and specific patient situations.
Use of chemotherapy for soft tissue tumors is best considered investigational. Multiple studies (most retrospective) have found conflicting results, and a high-quality meta-analysis revealed modest survival benefits in patients with high-grade tumors.12 Relatively few studies of chemotherapy in soft tissue sarcoma are “clean.” Most mix patients with different histologies and use varying treatment regimens. Just as the results of chemotherapy for bone sarcoma would be difficult to interpret if osteosarcoma, chondrosarcoma, and Ewing’s sarcoma were lumped together, interpretation of chemotherapy results for soft tissue sarcoma is nuanced and complex. Adjuvant chemotherapy for these conditions is best given under the advice and management of an experienced sarcoma oncologist in a clinical trial until further data are available.13
Patients in whom soft tissue tumors are excised with a periosteal margin following radiation have a high risk of subsequent femur fracture and may benefit from prophylactic fixation.14 This usually is not done at the time of tumor resection to avoid increasing the magnitude of an already complex procedure. Patients can be allowed to recover from the initial surgery with final margin status assessed and flap healing accomplished before prophylactic nailing is done.
Medical Considerations
Patients requiring hip surgery for oncologic indications are at elevated risk of postoperative complications compared with general orthopedic patients. Preoperative chemotherapy can leave patients malnourished and immunosuppressed. Patients treated with doxorubicin (Adriamycin)-containing regimens (which are commonly used for sarcoma chemotherapy) are at risk for cardiomyopathy and arrhythmia. Patients are at risk for colonization with resistant bacteria from their frequent interaction with the health care system.
It is invaluable to meet with the patient and his/her medical/pediatric oncologist immediately before planned resection for patients on chemotherapy to assess fitness for surgery. One primary consideration is that the patient should not be neutropenic when entering a large surgery. As well, inferior survival is seen in patients with a delay to chemotherapy resumption longer than 21 days postoperatively in high-grade bone sarcoma (Fig. 51-2).15 Meeting with the medical/pediatric oncology team immediately preoperatively allows for planned resumption of chemotherapy following surgical resection.
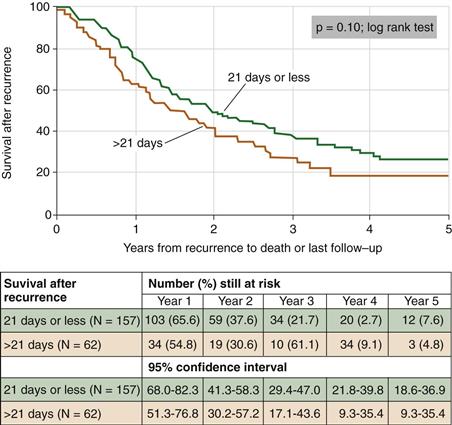
Figure 51-2 Kaplan-Meier curves comparing survival after recurrence according to whether the delay in the resumption of chemotherapy after definitive surgery was longer than 21 days as opposed to within 21 days. (Redrawn from Imran H, Enders F, Krailo M, et al: Effect of time to resumption of chemotherapy after definitive surgery on prognosis for non-metastatic osteosarcoma. J Bone Joint Surg Am 91:604–612, 2009.)
Thromboembolic disease is seen more frequently in patients with sarcoma resection than metastatic disease or standard joint arthroplasty.16 Patients with soft tissue sarcoma may present with venous thrombosis from vascular compression. Surgical site infection is seen in ≈10% of patients.17 The use of radiotherapy increases this risk further, particularly if an endoprosthesis is placed.18 These frequent complications highlight the complexity of medical care in an oncologic population.
Surgical Approach
The first critical assessment in approaching patients with primary malignancies around the hip is suitability for limb salvage. The two central tenets of limb salvage surgery are that (1) oncologic outcome should be the same as amputation, and (2) function should be equal to or better than amputation. Close scrutiny is given to involvement of the acetabulum, external iliac/femoral vessels, and sciatic nerve by tumor. These areas are often the places where critical margins can occur in primary tumors about the hip.
Surgical resection of the acetabulum, external iliac vessels, or sciatic nerve can be accomplished with good functional outcomes. However, if two or three of these structures require resection, patient function significantly falls while complication rates increase. Resection of the sciatic nerve, acetabulum, and external iliac vessels implies such a large tumor and engenders such a high likelihood of complications that patients generally will have better oncologic and functional outcomes with an amputation. Decisions when two of these three structures are involved are individualized, but very strong consideration should be given to amputation in such cases as a way to likely provide better ultimate functional and oncologic outcomes. When amputation is contemplated, preoperative meetings between the patient and a prosthetist are valuable.
When limb salvage is appropriate, the location and extent of the primary tumor dictate the ultimate surgical approach. Our most common approach to tumors about the hip is a direct lateral approach. All bony and nearly all relevant soft tissue structures can be accessed through this method, and the approach can be extended to access the entire femur if needed. The incision can be extended up around the iliac crest to access the pelvis and “T’d” to provide extension into an ilioinguinal approach. A poorly placed biopsy site may require alteration of this approach or a separate incision to resect the biopsy tract in continuity with the tumor.
Large oncologic resections around the hip and pelvis may be associated with significant hemorrhage. Use of autologous blood salvage (“cell-saver”) is contraindicated in the setting of malignancy. We administer prophylactic antibiotics around the time of surgery only and are liberal in our use of drains; these are often left for low wall suction for the first 2 to 3 days after surgery to minimize any fluid collections. A 12 French pediatric chest tube can be used in this manner and connected to a pleurovac to maintain a sterile circuit. When drains are near implants, we try to remove them by day 3. Given the amount of dissection and the often large areas of tissue “dead space” following resection, we often use vacuum-assisted wound therapy to provide negative pressure across an incision after primary closure. It has been our experience that we avoid the persistent weeping surgical wound at hip or hemipelvectomy incisions by using this technique.19
Oncologic resection may lead to extensive soft tissue defects. Similarly, radiated wounds present significant healing challenges. Healthy muscle and fascia should be used to cover implants whenever possible. This hip region often lends itself to local soft tissue transfer (e.g., gracilis flap) or regional flap coverage (vertical rectus abdominis turndown flap). If free flap coverage is needed, the femoral or profundus system provides ready sites of anastomosis.
When vascular structures are encased by tumor, resection and reconstruction of involved blood vessels can be performed (Fig. 51-3). The complication rate from these procedures is formidable, however.20 Direct vascular invasion often heralds metastatic disease.
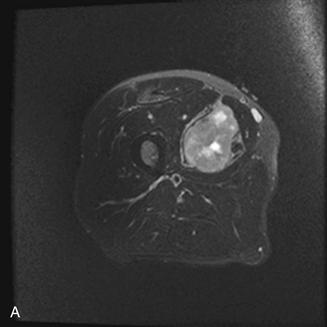
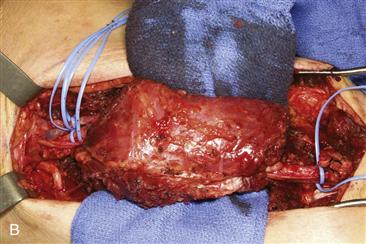
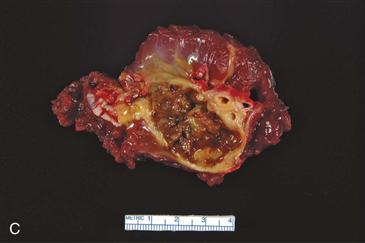
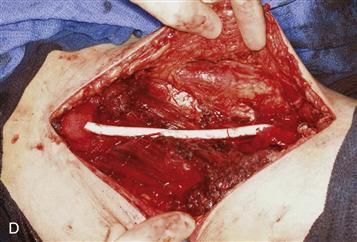
Figure 51-3 Wide excision of high-grade soft tissue sarcoma of the thigh. A, Magnetic resonance (MR) demonstrating tumor encasement of femoral vessels. B, Specimen immediately before removal, with vessels entering and exiting the lesion. C, Cut tumor demonstrating vessel encasement. D, Vascular reconstruction.
Reconstructive Options
Soft Tissue Tumors
Reconstructive options are individualized to the needs of the patient. Many soft tissue resections require little formal reconstruction. In the event that significant skin is excised, split-thickness skin grafting may be needed. Local muscle transfer may serve to cover vascular or nervous structures or to recentralize the extensor mechanism if an asymmetrical portion of the quadriceps is resected.
Resection of the sciatic nerve is often required with soft tissue sarcomas of the buttock region. Ultimate function for these patients is surprisingly good, although they require bracing and physical therapy to achieve these goals.21
If tumor extends to but does not infiltrate bone, resection may be accomplished with a periosteal margin. Our practice is to treat these patients with preoperative radiation and surgical resection using the periosteum as a margin, and then to provide a boost dose with intraoperative radiotherapy. Postoperatively, patients are at risk for insufficiency fractures that are difficult to heal.14 We maintain them in a protected weight-bearing stance until all wounds heal, and then proceed with prophylactic intramedullary fixation 8 to 12 weeks postoperatively. This delay allows an initial magnetic resonance (MR) scan to be obtained after postoperative changes have regressed and before susceptibility artifact from an implant is present.
Malignant Bone Tumors
Multiple reconstructive options following wide bony resections about the hip are available, depending on which combination of acetabulum, proximal femur, and femoral diaphysis is resected. Reconstructions use some combination of vascularized autograft, allograft, and prosthetics.
Resection of the femoral diaphysis alone is generally treated with intercalary allografts to preserve the native hip joint (Fig. 51-4). Grafts may be fixed with plates or intramedullary devices. Although union rates are similar with the two approaches, fracture rates may be lower with the use of intramedullary fixation.22 Vascularized free fibula or periosteal graft transfer can be used to augment healing of the host-allograft junction.23
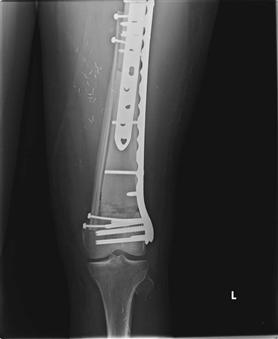
Figure 51-4 Intercalary allograft reconstruction augmented with vascularized fibula following resection of a diaphyseal osteosarcoma (similar techniques are used for more proximal tumors).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







