Chapter 49 Neoplasms Around the Elbow
Introduction
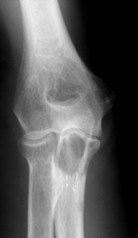
The elbow appears to be an area which is rarely afflicted by primary bone tumours. Although it is difficult to give a true incidence of the conditions, a review of 2900 cases referred to the London Bone Tumour Service reveals only 39 cases involving the elbow joint. The pathological diagnoses of these cases are shown in the Table 49.1. Although this table may represent the true incidence of primary malignant lesions in the southern half of the UK, it does not reflect the true incidence of benign lesions, many of which will be treated without referral to a supra-specialist centre. Finally the most frequent lesion seen around the elbow joint will in fact be a metastasis, particularly in a patient over 40, the primary lesion being either in the breast, prostate, bronchus, kidney or thyroid (Fig. 49.1). Pathological fracture is not an unusual mode of presentation (Fig. 49.2).
Table 49.1 London Bone Tumour Service: pathological diagnoses of cases involving the elbow
| Eventual diagnosis | No |
|---|---|
| Ewing’s/malignant round cell tumour | 8 |
| Aneurysmal bone cyst | 6 |
| Osteoblastoma | 2 |
| Giant cell tumour | 1 |
| Chondroblastoma | 1 |
| Metastasis | 6 |
| Others | 15 |
| Total (elbow) | 39 |
| Total cases (all sites) | 2900 |
Background/aetiology
The most common benign lesion of bone is undoubtedly the solitary osteocartilaginous exostosis (osteochondroma). This condition was first described by Sir Astley Cooper in 1818.1 A review of 323 solitary lesions throughout the skeleton revealed only five around the distal humerus.2 In multiple exostoses (diaphyseal aclasis) the distal humerus, in common with other long tubular bones, may be affected and unequal growth of the bones about the elbow may cause radiohumeral subluxation.3 Diaphyseal aclasis must be distinguished from Ollier’s disease or multiple enchondromatosis. This is a cartilage dysplasia which rarely affects the elbow region, although involvement of a bone close to an epiphysis may lead to forearm distortion.4 The association of soft tissue haemangiomas with chondromatosis is known as Maffucci’s syndrome.5,6 Any bone or area may be affected and the rate of malignant transformation of the enchondritic element is said to be greater than in Ollier’s disease.
Sir Astley Cooper is also credited with the first description of a giant cell tumour and emphasized its benign nature, although the French surgeon, Nelaton, was the first to give a good account of the varied clinical course and histological features.7 A review of 265 cases of giant cell tumour finds only three occurring around the distal humerus.
None of the four common primary bone malignancies – osteosarcoma, chondrosarcoma, Ewing’s sarcoma and malignant fibrous histiocytoma – are commonly found around the elbow joint. Sir James Paget first described osteosarcoma in 1854.8 Until the mid-1970s, treatment had always been by ablation, but with modern chemotherapy, limb-sparing surgical techniques may be attempted. Ewing first described his eponymous disease in 19219 and further classified telangiectatic osteosarcoma in 1922.10 The disease which bears his name is now thought to have been originally recognized by Lucke in 1866.11 It is considered to be one of a spectrum of small round cell tumours to afflict bone. Considerable skill and immunocytochemistry may be required for a histopathologist to differentiate Ewing’s sarcoma from neuroblastoma, primitive neuroectodermal tumour of bone, small cell osteosarcoma, embryonal rhabdomyosarcoma and lymphoma of bone.
Presentation, investigation and treatment options
Bone-forming tumours
Benign
Osteoid osteoma
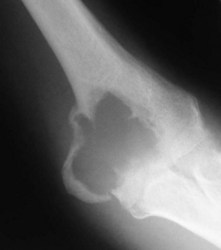
This condition usually presents as vague pain, often nocturnal characteristically relieved by aspirin or other non-steroidal antiinflammatory agents.12 The pain may be associated with tenderness and vasomotor disturbance. Classically, the osteoblastic nidus arises within the cortex or spongiosa of long bones. The nidus is less than 1 cm in diameter and induces dense surrounding change. If the nidus is in a subarticular location diagnosis can be difficult as the reactive changes may not occur. Intracapsular osteoid osteomas around the elbow have been mistaken for tuberculous synovitis13 and rheumatoid arthritis.14 It is also well recognized that longstanding disease can induce degenerative changes (Fig. 49.3).15
The radiological investigation consists initially of a plain X-ray. This may or may not reveal the characteristic nidus. When marked reactive bone is seen, the differential diagnosis is between bone abscess, sclerosing osteomyelitis of Garré, osteochondritis and stress fracture.16 Bone scintigraphy will usually help localize a nidus, but is relatively non-specific. Classically the area has two levels of activity. Computed tomography (CT) if performed in close 2 mm sections, will be successful in accurately locating the nidus prior to surgical resection or ablation.17
The treatment of osteoid osteoma requires excision or ablation of the nidus. As the nidus is surrounded by dense reactive bone, this can be difficult to achieve. The traditional approach requires wide exposure with radiological verification of the site. More recently the use of intra-operative scintigraphy has been described, although this is rarely required.18 Excisional biopsy of the nidus under CT control is now being successfully performed (Johnson and Cannon, manuscript in preparation). In addition, radiofrequency ablation is now being undertaken with encouraging results. The latter is performed by radiologists under CT control, the only real contraindication being where the nidus is adjacent to either vascular or neural tissue.19,20
Osteoblastoma
This is a similar lesion histologically to osteoid osteoma, although larger and not inducing so much reactive surrounding change. It is an extremely rare lesion affecting mainly the axial skeleton, although any bone may be affected. Patients are predominantly male in their second or third decades.21
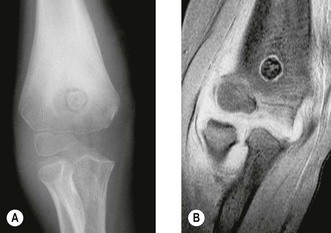
The clinical presentation is of vague pain, less marked than osteoid osteoma, and not particularly relieved by salicylates. Occasionally there may be both swelling and joint dysfunction. The radiological features classically show an expansile lesion, radiolucent with a thin shell of peripheral new bone (Fig. 49.4).
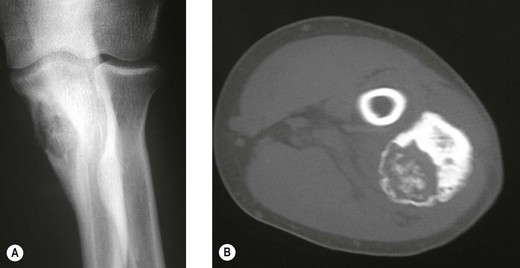
Figure 49.4 (A) Anteroposterior X-ray and (B) computed tomography scan, showing osteoblastoma of the proximal ulna.
Treatment consists of either curettage with or without bone grafting or local excision. Local excision gives good local control but reconstruction may be required. Recurrence rate may be as high as 20% following curettage, although the use of radiotherapy in extra-spinal cases is rarely required.22
Malignant
Osteosarcoma
Osteosarcoma is the most common primary bone tumour and can occur at any age, although most cases occur in the first two decades of life.23 Although commonly affecting the metaphysis around the knee, the humerus is the third most frequently affected bone, with tumours often arising at an earlier age than in the lower limb, and usually affecting the proximal metaphyseal area.24
Osteosarcomas are usually subtyped on the basis of their histological patterns into fibroblastic, chondroblastic, telangiectatic or mixed. Although the subtype was originally thought to have a bearing on prognosis, more recently carefully controlled studies have shown this is not to be the case.25 Histological grading as a prognostic indication is also unreliable.26
Patients usually present with a short history of pain followed by swelling and joint dysfunction. Occasionally they can present with a pathological fracture. The many radiological advances which have occurred in the last decade have allowed very accurate staging of the disease. Plain X-rays remain the primary diagnostic tool but can underestimate the extent of the tumour. CT scanning will give a more accurate visualization of cortical destruction and soft tissue spread but may miss skip lesions in the same bone. It is a useful technique in planning biopsy and local excisions.27 The accepted staging investigation to assess for the presence of pulmonary metastases is CT of the chest. Local staging of disease depends heavily on magnetic resonance imaging (MRI) of the whole bone. This, particularly in T2 mode, will delineate accurately the intramedullary extent and soft tissue extent of the lesion. It will outline the relationship of the tumour to the adjacent joints and to the blood vessels.
Preoperative bone scanning using technetium isotopes is useful in identifying skip lesions and metachronous lesions in other bones. A radioisotope scan may also on occasions, demonstrate pulmonary metastases.28 Having performed the radiological staging procedures, it is necessary to establish the pathological diagnosis. For this a biopsy is required. Many argue that this should be an open biopsy. However, Stoker et al29 have shown the accuracy of Jamshidi needle biopsy cores obtained under local anaesthesia and image intensification. This can also be supplemented by other imaging techniques.
Historically, the treatment of osteosarcoma has always been surgical ablation. In the upper limb this has led to either disarticulation at the shoulder for lesions in the distal humerus or forequarter amputation for more proximal growths. Survival, however, has been poor with 5-year survival rates varying between 11% and 25%. Cade30 reported an alternative method of treatment combining preoperative radiation followed by surgical ablation in cases without pulmonary metastases. Fewer amputations were performed but the survival rate was unaltered. This has subsequently been confirmed by other series.31
By the early 1970s it became evident that this appalling survival rate might be improved by the use of adjuvant chemotherapy. Many different protocols were produced, some claiming very high survival rates,32 although controlled trials of therapies were not performed. In the UK the Medical Research Council combined with the European Organisation for Research into Cancer Treatment (EORTC) set up controlled trials of adjuvant chemotherapy. These have advanced to neoadjuvant chemotherapy, which allows treatment of undetected micro-metastases and causes necrosis of the tumour which may result in some shrinkage of the primary tumour. This latter effect may allow easier local resection and reconstruction of the limb. The initial trials compared the effect of doxorubicin (Adriamycin) and cisplatin with or without high-dose methotrexate. The two-drug combination performed better, giving a survival of 65% at 5 years. This combination has also been compared with a Rosen T10 regimen. The trial assessed 400 cases and analysis revealed significant difference between the treatment modalities. A further trial into which all osteosarcoma cases without metastases were entered was an attempt at dose intensification using the rescue factor granulocyte cell stimulating factor. It aimed to allow three courses of chemotherapy to be given in 6 rather than 9 weeks. This trial unfortunately did not show any major difference in survival. Currently an international randomized trial is ongoing for the aim of optimizing treatment strategies for resectable osteosarcoma based on histological response. Effectively those with a good early response to induction chemotherapy, which consists of methotrexate, Adriamycin and cisplatin, are randomized to a standard arm of the same therapy in the postoperative period plus or minus interferon, whereas for those that are recognized to have a poor response, chemotherapy is enhanced with the addition of ifosfamide and etoposide.33 The outline of this regimen in seen in Figure 49.5.
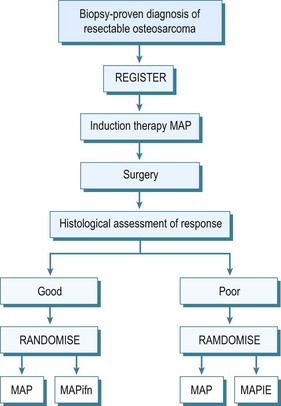
Figure 49.5 EURAMOS 1. A = adriamycin; P = cisplatin; ifn = interferon; i = ifosfamide; e = etoposide.
Parosteal osteosarcoma
This is a low-grade malignant tumour which develops on the external surface of large bones. The disease was first reported by Geschickter and Copeland in 1951.34 It has a long natural history and tends to affect patients in the second and third decades of life. Most patients present with a long-standing swelling associated with a dull ache. The elbow is only rarely affected.
Treatment consists of wide local resection of the tumour with appropriate reconstruction. It is generally accepted that in most cases chemotherapy is not indicted. Histological analysis of the tumour may show high-grade changes in the fibrous elements. These tumours have a poor prognosis,35 and chemotherapy may be indicated. Occasionally the low-grade component may transform or dedifferentiate to a high-grade osteosarcoma. Treatment is then as for an osteosarcoma.36
Periosteal osteosarcoma
This is a very rare tumour, with many still doubting its existence,37 although it probably is a variant of parosteal osteosarcoma with a prominent cartilaginous component.38 These are usually small unicortical lesions. Treatment is by wide resection and appropriate reconstruction. Whether chemotherapy is required in their management is still unclear.
Cartilage forming tumours
Benign
Osteochondroma
These cartilage-capped bony protrusions may develop in any bone derived from cartilage. They are usually discovered in childhood and many are found only as incidental findings on radiology. They are usually painless, although discomfort can be invoked by mechanical irritation or nerve compression. A pseudoaneurysm has also been reported specifically in the popliteal region.39
In diaphyseal aclasis the patient may also present with growth abnormality and subluxation at the radiohumeral joint. In this condition, removal of the lesions is rarely enough and the patients often require major reconstructions to correct the deformities.40 The patient should be warned specifically of the possibility of malignant change associated with growth after maturity. Known lesions which cannot be palpated, should be monitored by radiography, particularly the depth of the cartilage cap. Unfortunately, bone isotope scanning cannot reliably differentiate benign from malignant cartilage lesions.41 MRI and positron emission tomography (PET) may be a reasonable alternative.
Chondroblastoma
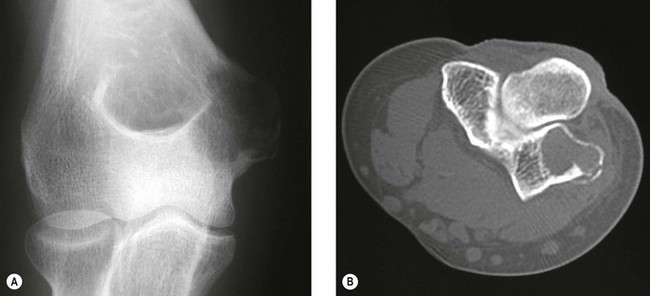
This is a rare bone tumour usually located in the epiphyseal plate. It is essentially benign. Jaffe and Lichtenstein42 consider that it is a tumour developed from cartilaginous germ cells, although Higaki et al43 consider the cell of origin to be histiocytic. Typically, the patient is in the second decade of life and is more likely to be male. A long prodromal history is typical and there may be muscle wastage and joint restriction.44 The most common site of affliction is the upper humeral epiphyseal plate, although they can be associated with any primary or secondary site of ossification.
Typically, the lesion is radiolucent crossing the growth plate – intralesional calcification can be seen particularly with tomography.45 CT may better illustrate the local invasive properties of the tumour. It is well recognized that chondroblastoma may exhibit metastatic implantation to the pulmonary tissues. These areas if resected have a ‘benign’ histological appearance, and, therefore, represent implantation of benign tumour transported via the haematogenous route, rather than true metastases (Fig. 49.6).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree