CHAPTER 92 Injection Procedures
BACKGROUND
The natural history of low back pain has been reported to be quite favorable in some studies and is often quoted to patients as such. It has been reported that 40–50% of patients are symptom free within 1 week and up to 90% have resolution of symptoms without medical attention in 6–12 weeks.1–4 However, recent studies suggest that lumbar axial pain is a more significant condition characterized by acute exacerbations and variable periods of remission. Deyo and Tsui-Wu5 reported 33.2% of patients with low back pain reported symptoms with a duration of less than 1 month, with 33% reporting pain for 1–5 months and 32.7% reporting pain for longer than 6 months. Recurrence rates from 60–85% have been reported in the first 2 years following an acute episode of low back pain.6,7 More recently, Von Korff et al.8 observed that over a 2-year follow-up period, 44% report chronic symptoms (defined as more than 90 days of back pain in the previous 6 months). In this study, most patients were experiencing low levels of back pain with 20% rating their pain as 4 or greater on a 0–10 scale; 13% rated their current pain as 5 or greater and only 8% reported pain of 6 or greater. Furthermore, Von Korff et al.8 reported that 15–20% of primary care patients with low back pain show moderate to severe activity limitations during a 1-year follow-up period after the initial episode has resolved.
PATHOPHYSIOLOGY
Descriptions of the treatment for low back pain dates to Hippocrates (460–370 BC), with the reporting of joint manipulation and the use of traction.9 It often is theorized that the onset of low back pain is associated with bipedal ambulation and that this transformation in the mechanics of locomotion is the inciting evolutionary event that has made the lumbar spine susceptible to a relatively accelerated rate of degenerative disease. Degeneration is universal to the structures that comprise the basic functional spinal unit, which is composed of two adjacent vertebral bodies, the intervertebral disc, and the two zygapophyseal (Z-) joints at the same segment functioning as a tri-joint complex. As humans age, endure various micro- and macrotraumas, and undergo changes in body habitus that alter and unevenly distribute biomechanical forces on the lumbar spine, there is a natural progression of degeneration of the lumbar motion segment with corresponding anatomic, biomechanical, radiologic, and clinical findings.10 It is this evolution that provides the insight into the root cause of lumbar spinal pain syndromes.
The posterior elements of the lumbar spinal functional unit bear less weight than the anterior elements in all positions. During sitting, the anterior elements bear over 90% of the forces transmitted through the lumbar spine, whereas during standing, this fraction decreases to approximately 80%.10 As the degenerative process progresses, the relative anterior to posterior force transmission approaches 50%.10 The spine functions best within a realm of static and dynamic stability. The bony architecture and associated specialized soft tissue structures, especially the intervertebral disc, provide static stability. Dynamic stability, however, is accomplished via a system of muscular and ligamentous supports which act in concert during various functional, occupational, and avocational activities. The overall mechanical effect of these structures maintains the histologic integrity of the tri-joint complex. The net shear and compressive forces must be maintained below respective critical minimums in order to maintain the integrity of the tri-joint articulation. Persistent, recurrent, nonmechanical and/or excessive forces to the motion segment beyond minimal thresholds leads to microtrauma to the disc and Z-joints, which triggers and continues the degenerative process.10 This degenerative cascade, as described by Kirkaldy-Willis,10 is the widely accepted pathophysiologic model describing the degenerative process and ensuing spinal segment dysfunction as it affects the lumbar spine and its individual motion segments. This process is described as occurring in three phases in a progressive longitudinal manner over time. These phases are a continuum with a gradual transition rather than three distinct, clearly definable stages. Although the etiology of spinally mediated lumbar axial pain is variable and not always clearly definable, there are a finite number of potentially painful structural components of the lumbar spine, and the aforementioned degenerative cascade provides insight on the process by which these components develop pathology and can become symptomatic.
BASIC SCIENCE EVIDENCE OF INFLAMMATORY MECHANISMS IN DISCOGENIC-MEDIATED PAIN SYNDROMES
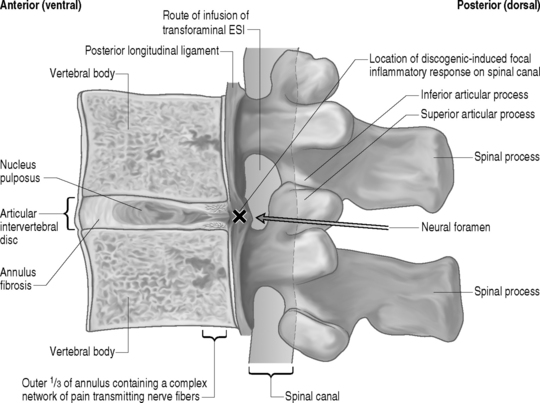
The anterior elements of the lumbar spine, most notably the intervertebral disc, are currently thought to be the primary source of the vast majority of spinally mediated lumbar axial pain syndromes. Several theories cite a traumatically induced acute annular tear as the inciting pathologic event. Others theorize the natural result of aging is the sole etiology of pathology of the lumbar disc. However, these models do not explain spontaneously occurring annular tears and disc degeneration in the young. Therefore, the cause of lumbar disc disease is most likely multifactorial. Various genetic, environmental, autoimmune, inflammatory, traumatic, infectious, toxin-induced, and other factors may alone, or in various combinations, result in the initiation and progression of degeneration of the intervertebral disc in a way that has not yet been elucidated. However, to some degree, the basic science literature has been able to elucidate the inflammatory mechanisms of discogenic mediated lumbar axial pain syndromes. In 1987, McCarron et al.11 published a study in which they reported on the incitation of an inflammatory response upon injection of autologous nucleus pulposus into the epidural space of dogs. In addition, they reported that a delay in the treatment of this inflammatory process could result in tissue fibrosis and cellular damage. In 1990, Saal et al.12 demonstrated significantly increased level of PLA2, the rate-limiting enzyme in the arachidonic acid cascade, activity in lumbar HNP further demonstrating the inflammagenic properties of nuclear material. In 1996, further study helped elucidate the biochemical factors by which the nucleus pulposus elicits an inflammatory process. Takahashi et al.13 demonstrated the presence of inflammatory cytokines, including IL-1a+b, IL-6, TNF, GMCSF, and L-B4, in herniated nucleus pulposus (HNP). Kang et al.14 reported increased levels of MMPs, nitric oxide, IL-6, and PGF2 in the culture media of HNP. These properties may in part be explained by the fact that the nucleus pulposus, a remnant of the embryonic notochord, is sheltered from the bloodstream during embryologic development and may therefore be construed as an antigen upon presentation to the immune system when entering the ventral aspect of the epidural space.15 Subsequently, epidural steroid injections (ESI) have become commonly employed interventions in treating spinally mediated lumbar axial pain syndromes thought to be potentially due to an inflammatory mechanism based on the aforementioned basic science literature as it pertains to specific discogenic induced spinal syndromes.
EVOLUTION OF INTERVENTIONAL SPINE PROCEDURES
The initial reports of epidural injections almost a century ago reported the instillation of cocaine into the epidural space to treat lumbago and sciatica. In the early 1900s, epidural injection of local anesthetic was used to treat intractable sciatica.16 In 1952, Robecchi and Capra17 reported success with the first ESI in treating lumbar and associated sciatic pain. Presently, ESIs are a commonly utilized intervention in the management of spinally mediated lumbar axial syndromes both with and without associated radicular symptomatology. Several studies have shown that up to 40% of blind (nonfluoroscopically guided) ESIs may erroneously deliver the injectate superficially into the soft tissues or inadvertently deep into the subarachnoid space, which may result in serious complications.18–21 Therefore, we strongly advise that any type of ESI be performed under real-time fluoroscopic guidance with contrast-enhanced visualization of flow within the epidural space to the target site prior to the injection of medication unless otherwise contraindicated. In 1972, Winnie et al. emphasized the importance of placing the medication as close to the site of pathology as possible for the clinician to optimize outcome. They demonstrated improvement in 80% of patients in whom corticosteroids were infused at the site of pathology.22 This logical concept must be considered when determining which of the three commonly utilized epidural access routes, transforaminal, interlaminar, or caudal, is utilized when performing an ESI.
EFFICACY OF TRANSFORAMINAL ESI FOR LUMBAR AXIAL PAIN SYNDROMES WITH RADICULAR INVOLVEMENT
The optimal route for injection of corticosteroids into the epidural space at the site of pathology in patients with discogenic-mediated lumbar axial pain syndromes with corroborative radicular involvement is via the transforaminal route. This approach allows the clinician to deliver the injectate, composed of a combination of 6–12 mg of betamethasone and 0.5–1 cc of 1% lidocaine, precisely to eradicate the known inflammatory response emanating from the potentially inflammagenic HNP focally on the specific inflamed nerve root sleeve (Fig. 92.1). The efficacy of this approach has been demonstrated in four different randomized, prospective, double-blind, controlled clinical trials. Riew et al.23 reported results of a prospective, randomized, controlled, double-blinded study using fluoroscopically guided lumbar transforaminal injections in 55 patients with imaging evidence of nerve root compression and corroborative radicular symptoms. Twenty-eight patients received bupivacaine and betamethasone, and 27 received only bupivacaine. At follow-up (13–26 months) 33.3% of the bupivacaine group decided not to have surgery, versus 71.4% of the bupivacaine and betamethasone group. This difference in surgical rates was statistically significant (p<0.004). This controlled study demonstrates the beneficial effect of precisely delivered corticosteroids in obviating the need for operative treatment in patients with HNP and/or spinal stenosis. Kraemer et al.24 published a study in which they reported long-term pain relief with transforaminal ESI in a study in which 49 patients with lumbar radicular pain were randomized into a corticosteroid group and control group. Karppinen et al.25,26 reported on 160 consecutive patients with symptomatic herniated discs with no prior history of lumbar spine surgery randomized into either a corticosteroid group or normal saline group. Outcome measures obtained at 2 weeks, 3 months, and 6 months included pain relief, sick leave, medical costs, the Nottingham Health Profile, and the future requirement for surgical intervention. They reported that transforaminal ESI provided significant short- and long-term improvement in all of the aforementioned outcome measures. Thomas et al.27 reported results of a prospective, randomized, controlled, double-blinded study comparing the relative effectiveness of fluoroscopically guided lumbar transforaminal ESIs versus blind interlaminar ESIs in patients with radicular pain. They demonstrated the superiority of transforaminal ESIs in all of a variety of outcome measures including finger-to-floor lumbar flexion, daily activity including work and avocational function, and Dallas pain scores. This direct comparison study underscores the importance of fluoroscopic guidance as well as delivering medication accurately and precisely to the site of a potential ongoing inflammatory response. In a non-blinded, randomized, prospective study by Butterman,28 transforaminal ESIs provided efficacy measured by reducing symptomatology and disability and obviating surgery at a follow-up of up to 3 years in patients with large (>25% of the cross-sectional area of the spinal canal) symptomatic lumbar herniated discs. Butterman28 also reported that in those patients who had short-term improvement or ineffectiveness of transforaminal ESIs and required surgical discectomy, there was no adverse affect in the outcome of that surgery from the temporal delay caused by the trial of transforaminal ESIs.
In addition to the aforementioned randomized clinical outcome studies, several prospective nonrandomized clinical trials on the efficacy of transforaminal ESI also strongly suggest the beneficial effects of transforaminal ESIs for HNP causing lumbar axial pain with corroborative radicular pain. For example, Weiner and Fraser29 showed 21 out of 28 patients with a computed tomography (CT) documented HNP and corroborative lower extremity pain receiving a single transforaminal infusion of betamethasone and 1% Xylocaine injectate had moderate or complete pain relief not requiring surgery at an average of 3.4 years follow-up. Lutz et al.30 reported on 69 patients, with an average of 22 weeks of symptoms, with magnetic resonance imaging (MRI) evidence of an HNP and radicular pain. Patients underwent an average of 1.8 transforaminal injections of betamethasone and 1% Xylocaine followed by a 6–12-week course of lumbar spine stabilization therapy. They reported a successful outcome, as defined by a 50% or greater reduction in pain and return to near-previous level of functioning, in 75% of patients at an average of 80 weeks follow-up. In a retrospective evaluation, Wang et al.31 demonstrated significant symptom improvement in both the short and long term and the avoidance of discectomy in 77% of patients with lumbar disc herniations treated with one to six transforaminal ESIs.
With respect to the interventional treatment of lumbar axial pain syndromes with corroborative radicular pain due to spinal stenosis, the previously stated Riew et al.23 study reported on the effectiveness of transforaminal ESI as it incorporated spinal stenosis in addition to HNP as the underlying etiology to their subjects symptomatology. In addition, Delport et al.32 conducted a retrospective review of treating symptomatic spinal stenosis a subgroup of which underwent transforaminal ESI. They reported that of 140 subjects, over 70% had symptom improvement and were at least somewhat satisfied with their response, over 50% reported sustained improvement in their function, and only 29% of the 140 subjects ultimately underwent surgical decompression. Botwin et al.33 conducted a prospective cohort outcome study assessing the efficacy of transforaminal ESIs in the treatment of symptomatic degenerative spinal stenosis. Thirty-four subjects were followed for 1 year. They reported that 75% of the patients had a successful long-term outcome as defined as >50% symptom reduction. They also reported more than 50% of the subjects improved standing and/or walking tolerance.
The aforementioned literature strongly suggests that transforaminal ESI should be the standard of care index interventional spine procedure for patients with spinally mediated lumbar axial pain syndromes associated with radicular involvement due to HNP and/or spinal stenosis when more conservative measures have failed. Furthermore, in the case of the majority of HNPs, the known phagocytic immunologic response and consequent benign anatomic natural history assists in the relatively high rate and long-term success of transforaminal ESIs.34,35
CONCEPTUAL PATHOPHYSIOLOGIC MECHANISMS OF LUMBAR AXIAL PAIN SYNDROMES WITHOUT RADICULAR INVOLVEMENT DUE TO A DISCOGENIC-MEDIATED INFLAMMATORY RESPONSE
Some important inferences from the aforementioned literature on the successful long-term treatment of HNP-associated lumbar axial pain syndromes with corroborative radicular involvement may be extracted that, when combined with known previously stated basic science literature and lumbar spinal anatomy, suggest a possible utilization for transforaminal ESIs in the interventional treatment of discogenic-mediated lumbar axial pain without radicular involvement. The similarities in the basic ideologies by which the initial incorrect thought processes of compression as the sole mechanism of radicular pain due to HNP requiring discectomy as stated nearly a century ago by Mixter and Barr36 mirror recent theory that motion segment instability and/or structural disc pathology is the sole mechanism of discogenic-mediated lumbar axial pain without radicular involvement. In the decades subsequent to Mixter and Barr’s36 landmark article identifying HNP pathology as a key source of lumbar axial and radicular pain, there have been many attempts to identify the precise pathophysiologic mechanism of discogenic-mediated lumbar axial pain without radicular involvement. As stated earlier in this chapter, over the years, there have been key pieces of literature to suggest that a tear in the outer one-third of the anulus of the intervertebral disc may be the most common etiology of acute spinally mediated lumbar axial pain.10,37 As with the painful sequelae of most degenerative spinal conditions, improvement and eventual remission within 6 weeks is the natural history. However, subacute symptoms can persist and the mechanism for this may be inflammatory in nature.38 The basic science literature discussed earlier in this chapter has repeatedly and clearly demonstrated the inflammagenic properties of the intervertebral disc, particularly the nucleus pulposus when it is exposed to the ventral epidural space.13–17,38 This literature lends credence to the mechanisms by which transforaminal ESI effectuate symptom relief on a molecular basis. Therefore, the possibility of a focally contained inflammatory response to a centrally or paracentrally contained herniated disc is entirely possible and may be instrumental in pain generation as well as inhibition of the natural healing processes known to exist. In fact, a contained HNP may chemically sensitize the posterior longitudinal ligament, resulting in axial pain in a fashion similar to that of inflammatory radicular pain due to an HNP sensitizing a nerve root. Furthermore, patients with internal disc disruption syndrome in which an annular fissure extends from the nucleus pulposus to the outer one-third of the anulus (which may or may not be associated with a HNP) and may pierce through the outer anulus and intermittently or consistently communicate microscopic amounts of nuclear material to the ventral epidural space ensuing in an intermittent and/or ongoing focal discogenic-induced inflammatory response (Fig. 92.2). This pathophysiologic model may explain why a randomized, prospective, double-blind clinical trial performed by Simmons et al.39 failed to demonstrate a statistically significant benefit to intradiscal corticosteroid infusions when compared to placebo. The aforementioned hypothesis suggests the inflammatory response occurs in the ventral portion of epidural space on the floor of the spinal canal juxtapositioned to the afflicted intervertebral disc(s) rather than within nuclear material encased by the anulus fibrosus.
EFFICACY OF TRANSFORAMINAL ESI FOR LUMBAR AXIAL PAIN SYNDROMES WITHOUT RADICULAR INVOLVEMENT
To date, there have been only two nonrandomized, retrospective studies reporting on the outcome of transforaminal ESI on spinally mediated lumbar axial pain due to discogenic pathology without imaging evidence of nerve root involvement. One is a subgroup reported by Rosenberg et al.40 stating greater than 50% pain reduction after one year in 59% of patients. The other study reported by Manchikanti et al.41 on patients with spinally mediated lumbar axial pain treated by one of three interventions: (1) blind interlaminar ESI, (2) fluoroscopically guided caudal ESI, and (3) fluoroscopically guided transforaminal ESI, states that superior short- and long-term pain relief is achieved via the transforaminal route. This conclusion makes anatomical sense as transforaminal ESIs likely distribute injectate more focally to the ventral epidural space when compared to interlaminar and caudal route and therefore are more target specific when attempting the deliver medication to a possible focal posterior discogenic-induced inflammatory response.
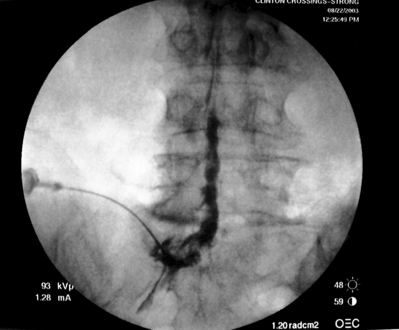
As previously implied, the best route for infusion of corticosteroids into the ventral epidural space in patients with a potential component of focal posterior discogenic inflammatory response is the transforaminal route. In many cases, the specific level of discogenic pathology has not yet been elucidated at the algorithmic point of consideration of ESIs for the treatment of lumbar axial pain thought to be potentially due to a discogenic inflammatory etiology. In fact, more than one segmental level may be contributing to inflammation in the ventral aspect of the lumbar epidural space. Therefore, unless imaging strongly suggests one segment, the S1 transforaminal ESI is recommended (Figs 92.3, 92.4). This route allows the clinician to mechanically drive the corticosteroid injectate, typically 12–18 mg of betamethasone, diffusely along the floor of the lumbar spinal canal via a pressure effect utilizing 3–5 cc of 1% lidocaine in an attempt to quell potential inflammation along the ventral epidural space incited by leakage of nuclear material. In those patients where one-level disease is strongly suspected, a smaller volume of local anesthetic may be infused after the corticosteroid dose at the foraminal level below the suspected level to deliver the corticosteroids in the cephalad direction more focally on the ventral aspect of the spinal canal at the posterior aspect of the suspicious intervertebral segment. In cases where a nonhealing annular tear(s) exists communicating nuclear material to the ventral epidural space, mediation of the inflammatory response may create a more optimal environment for spontaneous healing of the annular tear.
Technique of performing transforaminal ESI
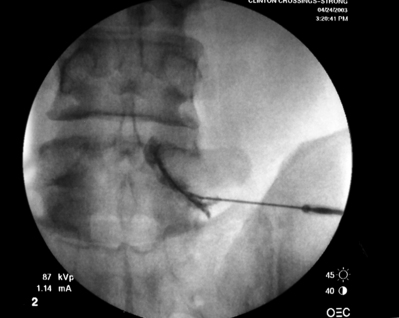
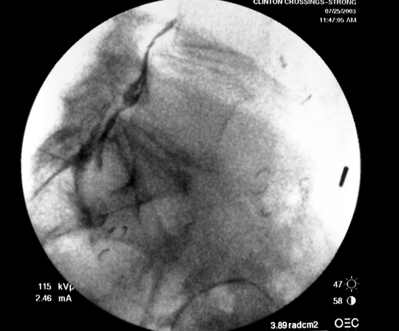
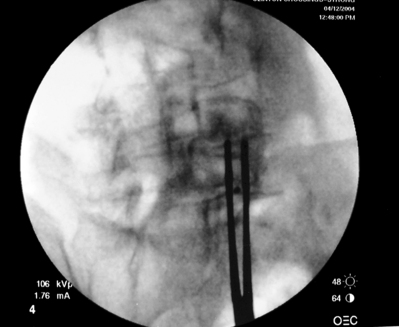
Transforaminal ESI absolutely requires fluoroscopic guidance in order for the site of pathology to be precisely targeted. The transforaminal route is the optimal access for delivering an injectate on the lumbar nerve sleeve and/or the ventral portion of the epidural space. Transforaminal ESIs are performed with the patient prone on the fluoroscopy table. The approximated area of skin overlying the level of the lumbar spine to be injected is cleaned thoroughly with povidone-iodine and the region is draped in a sterile fashion. Real-time fluoroscopy is utilized to identify the pedicle corresponding to the foramina through which injectate is to be delivered. In patients with a transitional segment, either a lumbarized S1 vertebral body or sacralized L5 vertebral body, the interventional spine specialist will need to count the vertebral bodies beginning with L1 after the most caudal rib (T12) to establish that the appropriate pedicle is identified. Once the targeted pedicle is correctly identified, the c-arm should be rotated ipsilaterally until the superior articular process of the juxtapositioned caudal vertebral body comes into view and advances to the medial portion of the targeted pedicle on the created oblique view. The targeted pedicle should now take on an ovoid shape once the correct degree of rotation is achieved as opposed to the circular shape on a true anteroposterior (AP) image with the lumbar spinous processes in the midline. Then, a skin wheal utilizing a combination of 1–3 cc of 1% lidocaine and 0.1–0.3 cc of sodium bicarbonate42 is raised just inferior to the midline portion of the targeted pedicle on the oblique image. A long-handled sponge clamp is utilized to guide a 20-gauge, 3.5 inch spinal needle through the raised skin wheal in a bull’s-eye manner (the needle hub concentrically outlines the needle tip (Fig. 92.5) just inferior to the midline portion of the targeted pedicle until a change in tissue resistance is appreciated or until the spinal needle hub abuts the skin. If the needle abuts skin prior to a change in tissue resistance, the 3.5 inch spinal needle is then utilized as an introducer and is positioned to align its tip to the midline portion of the targeted pedicle and a 25-gauge, 6 inch needle is guided through the index needle until a change in tissue resistance is appreciated. In either instance, the needle tip should then feel stable or locked into position if it has entered the targeted foramen. Verification of needle tip placement is achieved by rotating the c-arm in a manner that provides a true midline AP image. The needle tip is then slowly advanced to the six-o’clock position, proximated just inferior to the targeted pedicle. The lumbar spinal nerves exit the spinal canal just inferior and medial to the midposition of the corroborative pedicle on the AP image. Needle placement just superior to the spinal nerve and just inferior to the pedicle is confirmed by visualization of contrast medium centrally hugging the pedicle both inferiorly and medially as well as spreading cephalad along the ipsilateral ventral portion of the spinal canal, bathing the posterior aspect of the intervertebral discs as well as peripherally along the lumbar spinal nerve sleeve. Care must be taken and technique must be precise throughout needle advancement to maintain the bull’s-eye approach at all times via real-time fluoroscopy to prevent straying of the needle away from the targeted foramen. Inadvertent curvature of the needle outside the confines of the bull’s-eye approach may result in the needle tip hitting periosteum, such as the targeted pedicle if the needle tip strays superiorly, punctured viscus if the needle tip strays anteriorly towards the retroperitoneal and/or abdominal cavity, or punctured spinal nerve extraforaminally if the needle tip strays inferiorly. In some instances when performing a transforaminal ESI through the L5–S1 foramen, the targeted inferior aspect of the midline of the L5 pedicle may be accessible only by ipsilateral rotation due to one or a combination of a high-riding pelvis, significant loss of disc space height, large L5 transverse process, L5–S1 fusion, or a L5 on S1 spondylolisthesis. These obstacles can be overcome with a variable degree, depending on the severity of the aforementioned anatomical variations, of cranial to caudal tilt which will in effect move the pelvis inferiorly, creating direct access to the inferior portion of the targeted L5 pedicle. In cases of performing transforaminal ESI through the L5–S1 foramen in patients with a partially or completely sacralized L5 vertebral body, the approach is similar to that utilized when performing sacral transforaminal ESIs. When performing S1 transforaminal ESI, the skin wheal is raised superior and lateral to the targeted S1 pedicle. The needle is advanced through the skin wheal via a long-handled sponge clamp in the direction of the X-ray beam at a 45° angle to gently abut sacral periosteum just lateral to the S1 pedicle. Then, the needle is slightly withdrawn, reoriented inferiorly and medially, and slowly advanced, sliding to just inferior to the midline of the S1 pedicle into the targeted foramen. A slight suction or vacuum phenomena may be felt as the needle tip enters the targeted sacral foramen. A similar approach is utilized for L5–S1 transforaminal ESI in patients with partial or complete sacralization of the L5 vertebral body; however, the skin wheal is raised lateral and inferior to the targeted L5 pedicle and the needle too is advanced superiorly and medially after depth is achieved by contacting periosteum.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree