Chapter 176 Hypoglycemia
 Introduction
Introduction
Hypoglycemia is divided into two main categories: reactive hypoglycemia and fasting hypoglycemia. Reactive hypoglycemia, the most common, is characterized by the development of symptoms of hypoglycemia 3 to 5 hours after a meal and may herald the onset of early type II diabetes. Gastric surgeries may induce this condition,1 and anorexia nervosa has been a reported to be cause in the literature.2 Reactive hypoglycemia may also result from the use of oral hypoglycemic drugs. These sulfa drugs (sulfonylureas) appear to stimulate the secretion of additional insulin by the pancreas as well to as enhance the sensitivity of body tissues to insulin. Common examples of this class of drugs are listed in Box 176-1.
BOX 176-1 Oral Hypoglycemic Drugs
Some researchers, however, recommend that for many patients, this syndrome instead be designated idiopathic postprandial syndrome because, although symptoms do exist and are related to rapid drops in blood glucose, the absolute glucose levels are not reliable indicators of the syndrome. Many asymptomatic controls have glucose levels below 50, while many symptomatic patients have normal postprandial glucose levels.3–5
Fasting hypoglycemia is rare, because it usually appears only in severe disease states such as pancreatic tumors, extensive liver damage, prolonged starvation, autoantibodies against insulin or its receptor, various cancers, or as a result of excessive exogenous insulin in diabetic patients. Pregnant diabetic women using insulin or oral glycemic medications also have a high incidence of asymptomatic hypoglycemic events.6
Hypoglycemia can promote untoward physiologic changes in the body. Insulin-induced hypoglycemia is known to increase the levels of C-reactive protein, a known cardiac risk factor.7 Because glucose is the primary fuel for the brain, low levels affect the brain first. Symptoms of hypoglycemia can range from mild to severe, including the following:
 Diagnostic Considerations
Diagnostic Considerations
Clinical hypoglycemia is identified by modified Whipple’s criteria consisting of (1) central nervous system symptoms including confusion, aberrant behavior, or coma; (2) a simultaneous blood glucose level equal to or less than 40 mg/dL; and (3) relief of these symptoms by the administration of glucose.1 The normal fasting blood glucose level is between 65 and 100 mg/dL. A fasting plasma blood glucose measurement greater than 126 mg/dL on two separate occasions is diagnostic of diabetes.8 Although the most specific criterion for the presence of hypoglycemia is a blood glucose level of 40 mg/dL or less, a blood glucose level below 50 mg/dL should arouse clinical suspicion.1
A more functional test of blood sugar control is the oral GTT. It is used in the diagnosis of both reactive hypoglycemia and diabetes, although it is rarely required for the latter. After fasting for at least 12 hours, a baseline blood glucose measurement is made. Then the subject is given a liquid containing glucose to drink. The amount consumed is based on body weight: 1.75 g/kg. Blood sugar levels are measured at 30 minutes, 1 hour, and then hourly for up to 6 hours. Basically blood sugar levels greater than 200 mg/dL indicate diabetes. Levels below 50 mg/dL indicate reactive hypoglycemia. Table 176-1 explains in detail how to interpret the results of a GTT.
TABLE 176-1 Criteria of Response to the Glucose Tolerance Test
| Diagnosis | Response |
|---|---|
| Normal | No elevation >200 mg <200 mg at the end of the first hour <140 mg at the end of the second hour Never <20 mg below fasting |
| Flat | No variation more than ± 20 mg from fasting value |
| Prediabetic | >140 mg at the end of the second hour |
| Diabetic | ≥200 mg at the end of the second hour |
| Reactive hypoglycemia | A normal 2- or 3-hour response curve, followed by a decrease of ≥20 mg from the fasting level during the final hours |
| Probable reactive hypoglycemia | A normal 2- or 3-hour response curve, followed by a decrease of 10-20 mg from the fasting level during the final hours |
| Flat hypoglycemia | An elevation of >20 mg, followed by a decrease of ≥20 mg below the fasting level |
| Prediabetic hypoglycemia | A 2-hour response identical to the hypoglycemic prediabetic response but showing a hypoglycemic response during the final 3 hours |
| Hyperinsulinism | A marked hypoglycemic response with a value of <50 mg during the third, fourth, or fifth hour |
The Glucose-Insulin Tolerance Test
Relying on blood sugar levels alone is often not enough to diagnose hypoglycemia, because it is now widely recognized that the signs and symptoms of hypoglycemia can occur in individuals having blood glucose levels well above 50 mg/dL and there is a wide overlap between symptomatic patients and asymptomatic controls.3 Many of the symptoms linked to hypoglycemia appear to be the result of increases in insulin or epinephrine. Therefore, it has been recommended that insulin or epinephrine (adrenaline) be measured during a GTT because symptoms often correlate better with elevations in these hormones than with glucose levels.9,10 Several studies have shown that the glucose-insulin tolerance test (G-ITT) leads to a greater sensitivity in the diagnosis of both hypoglycemia and diabetes than the standard GTT.10,11
The G-ITT uses a standard 6-hour GTT coupled with measurements of insulin levels. The G-ITT appears to be one of the best diagnostic indicators for faulty sugar metabolism.11 As many as two thirds of subjects with suspected diabetes or hypoglycemia who have normal GTTs will demonstrate abnormal insulin tolerance tests. Table 176-2 lists the various patterns seen with the G-ITT.
TABLE 176-2 Criteria for the Glucose-Insulin Tolerance Test
| PATTERN | RESPONSE |
|---|---|
| Pattern 1 | Normal fasting insulin 0-30 units. Peak insulin at 0.5-1 hour. The combined insulin values for the second and third hours is <60 units. This pattern is considered normal. |
| Pattern 2 | Normal fasting insulin. Peak at 0.5-1 hour with a “delayed return to normal.” Second- and third-hour levels between 60 and 100 units are usually associated with hypoglycemia and are considered borderline for diabetes; values >100 units considered definite diabetes. |
| Pattern 3 | Normal fasting insulin. Definite diabetes. |
| Pattern 4 | High fasting insulin. Definite diabetes. |
| Pattern 5 | Low insulin response. All tested values for insulin < 30. If this response is associated with elevated blood sugar levels, it probably indicates insulin-dependent diabetes (“juvenile pattern”). |
24-Hour Continuous Glucose Monitoring
Continuous glucose monitors (CGMs) are electronic diagnostic systems that require the insertion of a sensing catheter under the skin of the patient’s abdomen. The catheter contains a miniaturized electronic device that measures blood sugar and then sends this information every few seconds to a pager-sized computer module worn on the patient’s belt for up to a week. The portable computer module translates and records blood sugar data, which can then be downloaded to a larger computer. A graph showing the average blood sugar reading every 5 minutes (288 blood sugar readings per day) can then be generated and studied in relation to food intake, appetite, food cravings, hypoglycemic symptoms, medication, and exercise. CGMs have been shown to be very useful tools in the diagnosis and monitoring of blood sugar control.12,13
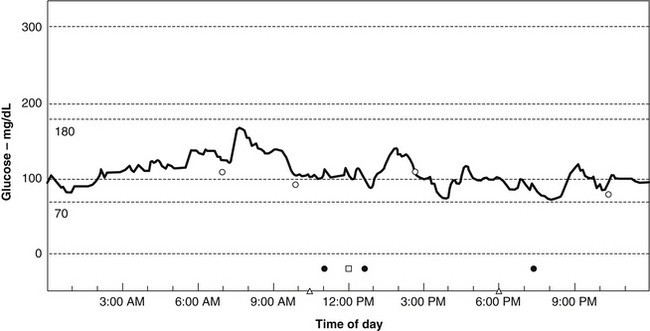
Using a CGM, Michael R. Lyon, MD, has discovered that most people with weight problems and insulin resistance go through their days with remarkably fluctuating blood sugar, or increased glycemic volatility. Rather than the absolute glucose level being the prime determinant of hypoglycemic symptoms, Lyon has found that feelings of hypoglycemia occur when the blood sugar drops rapidly, even when it was above the normal range (i.e., between 70 and 100 mg/dL) (Figure 176-1). In such cases, symptoms of hypoglycemia can range from mild to severe and include such things as food cravings, headache, depression, anxiety, irritability, blurred vision, excessive sweating, and mental confusion.
Rather than referring to these patients as suffering from hypoglycemia, Lyon and Michael T. Murray, ND, propose that they suffer from a new clinical entity they refer to as “elevated glycemic volatility.”63 These authors also believe that such volatility is at the heart of most weight problems. Their data indicate that rapidly fluctuating blood sugar levels are generally related to some degree of insulin resistance and made worse by more than moderate consumption of foods with a high glycemic impact.
The Hypoglycemia Questionnaire
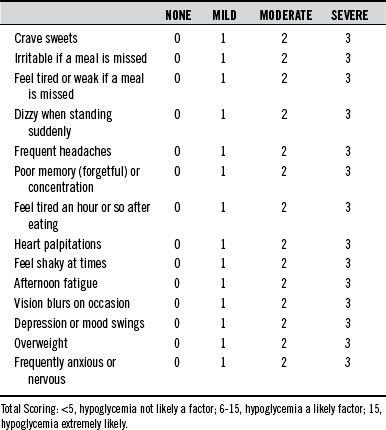
In many cases, when all is considered (especially cost to the patient), the most useful measure in the diagnosis of hypoglycemia remains an assessment of symptoms. In general, when symptoms appear 3 to 4 hours after eating and disappear with the ingestion of food, hypoglycemia should be considered. The questionnaire shown in Table 176-3 is an excellent screening method for hypoglycemia.
 General Considerations
General Considerations
In the 1970s, hypoglycemia was a popular “self-diagnosis” for a long list of symptoms; every symptom on the questionnaire in Table 176-3 was linked to hypoglycemia. Although all of these symptoms may be due to hypoglycemia, there are obviously other causes in many cases. The tremendous public interest in hypoglycemia and sugar intake was fueled by a number of popular books like Sugar Blues, by William Duffy; Hope for Hypoglycemia, by Broda Barnes; and Sweet and Dangerous, by John Yudkin. The popularity of these books and the diagnosis of hypoglycemia were met with much skepticism from the medical community. Editorials in the Journal of the American Medical Association and the New England Journal of Medicine during the 1970s denounced this public interest in hypoglycemia and tried to invalidate the concept.14,15
Research in the past 15 years has provided an ever-increasing amount of information concerning the roles that refined carbohydrates and the faulty control of blood sugar play in many disease processes. New terminology and descriptions are now used to describe the complex hormonal fluxes that are largely a result of ingesting too many refined carbohydrates. For example, the term metabolic syndrome or syndrome X has been introduced to describe a cluster of abnormalities that owe their existence largely to a high intake of refined carbohydrates, leading to the development of hypoglycemia, excessive insulin secretion, and glucose intolerance followed by diminished insulin sensitivity and leading to high blood pressure, elevated cholesterol levels, obesity, and, ultimately, type 2 diabetes. Metabolic syndrome is discussed in greater detail later.
Hypoglycemia, or idiopathic postprandial syndrome, is without question a valid clinical entity. A substantial amount of information indicates that hypoglycemia is caused by an excessive intake of refined carbohydrates.16,17 Although most medical and health organizations as well as the U.S. government have recommended that no more than 10% of the total caloric intake be derived from refined sugars added to foods, added sugar accounts for roughly 30% of the total calories consumed by most Americans.18 The average American consumes more than 100 lb of sucrose and 40 lb of high-fructose corn syrup each year. This sugar addiction plays a major role in the high prevalence of ill health and chronic disease in the United States.
Health Impacts of Hypoglycemia
Hypoglycemia and the Brain
The association between hypoglycemia and impaired mental function is well known. What is not as well known is the role that hypoglycemia plays in various psychological disorders. For example, despite numerous studies of depressed individuals showing a high percentage of abnormal glucose or insulin tolerance tests, rarely is hypoglycemia considered and rarely are depressed individuals offered dietary therapy.19,20 There is no explanation for this oversight among so many physicians, especially because dietary therapy (usually simply eliminating refined carbohydrates from the diet) is occasionally all that is necessary for effective therapy in patients who suffer from depression due to reactive hypoglycemia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree