Chapter 35 Autologous Chondrocyte Implantation
INTRODUCTION
Anterior cruciate ligament (ACL) and meniscus injuries are often combined with articular cartilage injuries, in which the cartilage injury is the most serious and most difficult to treat in the end. Noyes and coworkers18,19 and Shelbourne and associates24 found that acute and chronic ACL injuries were associated with articular cartilage injuries in 40% to 70% of knees. There are reports that in cases of meniscus injury, 40% to 50% of patients also have cartilage damage.25 Hjelle and colleagues11 reported on 1000 consecutive patients with symptoms requiring arthroscopy. Chondral or osteochondral lesions of any type were found in 61% of the patients. Curl and coworkers5 found that in 31,516 patients who underwent arthroscopy, 63% had a cartilage lesion and 19% had an Outerbridge grade IV cartilage lesion. Bone bruises have been reported in acute knee injuries12 and in combination with ACL injuries in 80%.17 The damage to the trabecular bone may heal, but the overlying cartilage may degrade over time.
INDICATIONS
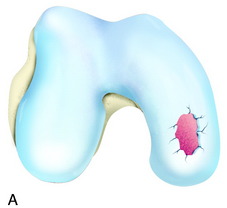
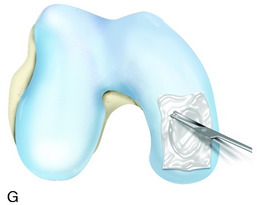
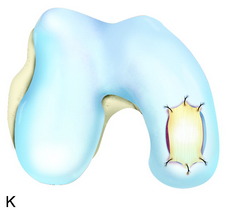
The recognition of serious articular cartilage injuries, including bipolar lesions in young and middle-aged individuals, has along with increased experience widened the indications for ACI (Fig. 35-1).
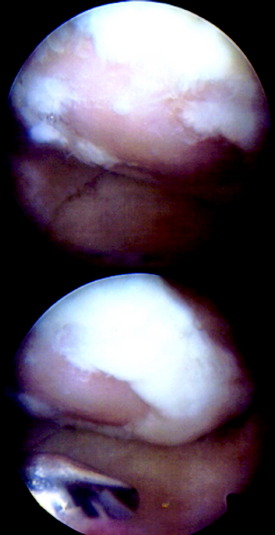
INTRAOPERATIVE EVALUATION
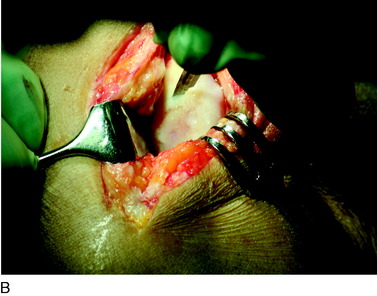
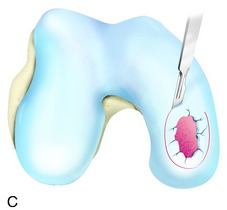
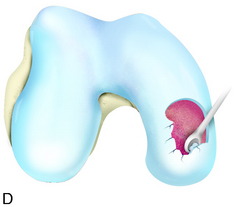
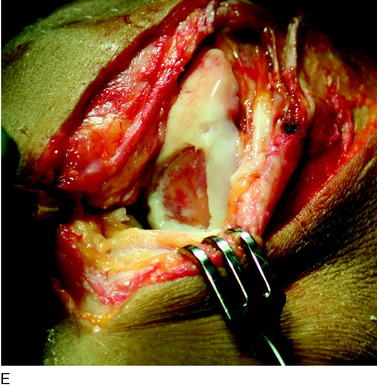
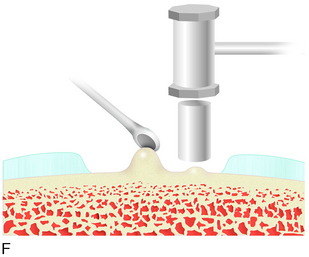
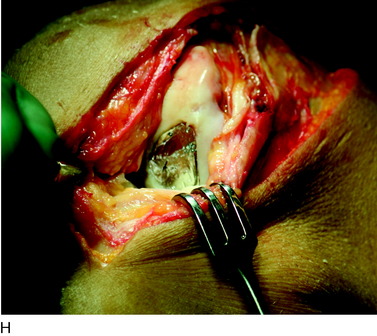
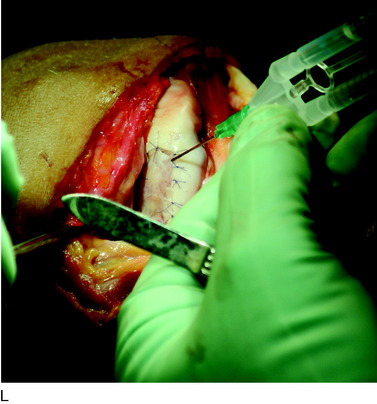
During the first-stage arthroscopy, knee stability is reassessed under anesthesia. The lesion is examined to decide whether it meets the indications for ACI. The lesion should be surrounded by healthy cartilage (but uncontainment is not a contraindication) and the opposing articular surface should be undamaged or have only minor superficial cartilage injury (Fig. 35-2). All intra-articular structures should be examined. The location, size, and depth should be noted using, for instance, the ICRS Knee Evaluation Package. Slices of cartilage with subchondral bone are harvested with a curet. The recommended harvest sites are the medial and lateral proximal rim of the femoral trochlea and the lateral aspect of the intercondylar notch in the knee joint. In more than 1400 patients who have had cartilage removed from the proximal medial trochlea for cell culturing, no complications or late symptoms have occurred from the donor site area. An optimal harvesting of cartilage is of great importance for the success of the cell culturing. Optimal cell quality is necessary for a successful result of this procedure and should be performed according to good laboratory practice or national regulations if present.
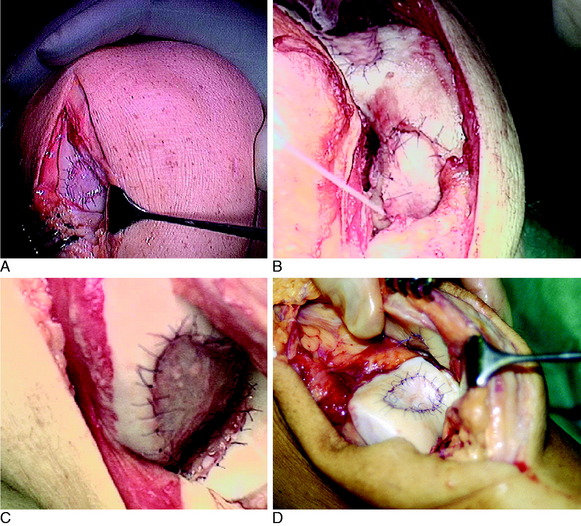
OPERATIVE TECHNIQUE
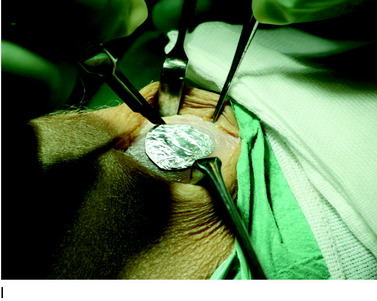
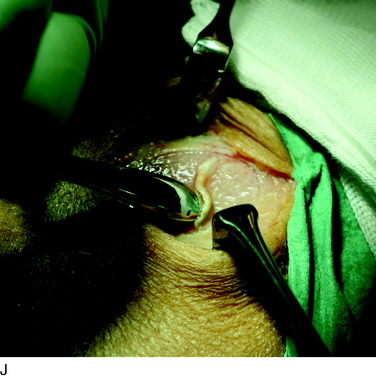
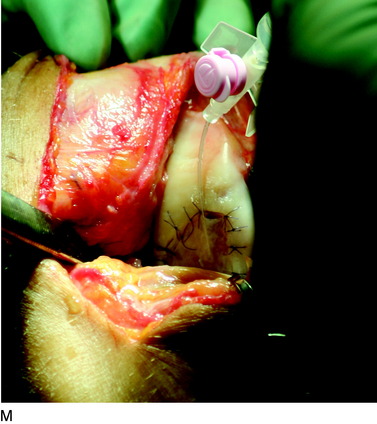
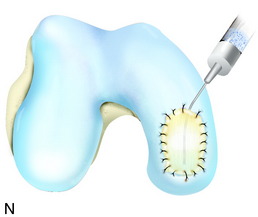
The flap is sutured to the cartilage rim of the defect at the level of the surrounding cartilage and the periosteum-cartilage border is sealed with fibrin glue. A gentle saline injection under the flap reveals any leakage along the cartilage rim, which must be sealed. After aspiration of the saline, the cultured chondrocytes are injected underneath the periosteal cover and the injection site is sutured and sealed (Fig. 35-3). The arthrotomy is closed in separate layers. Bandaging including the foot, lower leg, and knee is applied.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree