Chapter 135 Vitamin A
 Introduction
Introduction
Nomenclature
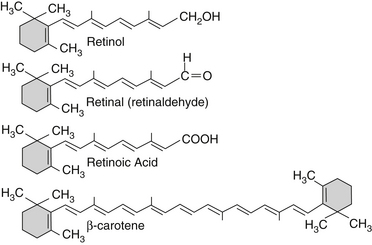
When isolated in its pure form, vitamin A is a pure, lipid-soluble yellow crystal with a condensed formula C20H29OH. Vitamin A is termed retinol, signifying that it is an alcohol that is intricately involved in the function of the retina of the eye. All-trans retinol is found in nature primarily as long-chain retinyl esters. The aldehyde form of all-trans retinol is commonly designated retinaldehyde or retinal, whereas the acidic form is termed retinoic acid. It has been suggested that retinol serves only as a precursor to these two active forms of vitamin A—retinal being primarily involved with vision and reproduction and retinoic acid being important in other somatic functions, such as growth and differentiation (Figure 135-1).1
Recommended Dietary Allowance
Vitamin A was originally measured in international units (IUs), with 1 IU being defined as 0.3 mg of crystalline all-trans retinol or 0.6 mg β-carotene. In 1967, an Expert Committee of the United Nations Food and Agriculture Organization and World Health Organization recommended that vitamin A activity be referred to in terms of retinol equivalents rather than in IU, with 1 mg of retinol being equivalent to 1 retinol equivalent (RE). The amount of beta-carotene required for 1 RE is 6 mg, whereas the amount required for other provitamin A carotenoids is 12 mg. In 1980, The Food and Nutrition Board of the National Research Council/National Academy of Sciences adopted this recommendation, and the 1980 recommended dietary allowance (RDA) for vitamin A is stated in milligrams and retinol equivalents; in 2001, the RDAs were revised to today’s values (Table 135-1).
TABLE 135-1 Recommended Dietary Allowances
| LIFE STAGE GROUP | RETINOL MCG/DAY (RETINOL EQUIVALENTS) | INTERNATIONAL UNITS (IU) |
|---|---|---|
| Infants (adequate intakes) | ||
| 0 to 6 months | 400 | 1320 |
| 7 to 12 months | 500 | 1500 |
| Children | ||
| 1 to 3 years | 300 | 1000 |
| 4 to 8 years | 400 | 1320 |
| Males | ||
| 9 to 13 years | 600 | 2000 |
| 14 to 18 years | 900 | 3000 |
| 19 to >70 years | 900 | 3000 |
| Females | ||
| 9 to 13 years | 600 | 2000 |
| 14 to 18 years | 700 | 2310 |
| 19 to >70 years | 700 | 2310 |
| Pregnancy | ||
| <19 years | 750 | 3750 |
| 19 to >50 years | 770 | 3950 |
| Lactation | ||
| <19 years | 1200 | 4000 |
| 19 to >50 years | 1300 | 4300 |
Dietary Sources
The most concentrated sources of preformed vitamin A are liver, kidney, butter, whole milk, and fortified skim milk, whereas the leading sources of provitamin A are dark-green leafy vegetables (collards and spinach) and yellow-orange vegetables (carrots, sweet potatoes, yams, and squash) (Table 135-2). Ingestion of excessive amounts of liver—2.7 to 11 kg/week—has been reported to cause hypervitaminosis A.2
TABLE 135-2 Food Sources of Vitamin A
| FOOD | PORTION SIZE | IU/PORTION |
|---|---|---|
| Meats | ||
| Beef liver, fried | 100 g | 50,375 |
| Calf liver, cooked | 100 g | 26,872 |
| Chicken liver, cooked | 2 livers | 25,760 |
| Vegetables | ||
| Sweet potatoes, baked | 1 medium | 14,600 |
| Carrots, raw | 1 large | 11,000 |
| Spinach, raw | 100 g | 8100 |
| Carrots, cooked | ½ cup | 8000 |
| Pumpkin, cooked | ½ cup | 8000 |
| Spinach, cooked | ½ cup | 7300 |
| Collard greens | ½ cup | 5400 |
| Broccoli, cooked | ½ cup | 1900 |
| Fruits | ||
| Watermelon |  melon melon | 5310 |
| Cantaloupe | ¼ melon | 3400 |
| Apricots, dried | 4 halves | 2275 |
| Apricots, raw | 2-3 medium | 2700 |
| Nectarines, raw | 1 medium | 1650 |
Data from Selhorst JB, Waybright EA, Jennings S, et al. Liver lover’s headache: pseudotumor cerebri and vitamin A intoxication. JAMA 1984;252:3365.
 Deficiency
Deficiency
Other Effects
Prolonged vitamin A deficiency results in the characteristic signs of follicular hyperkeratosis (buildup of cellular debris in the hair follicles, giving the skin a goose-bump appearance, which occurs most often on the back of the upper arm), night blindness, and a higher rate of infection. As the condition worsens, the mucous membranes of the respiratory tract, gastrointestinal tract, and genitourinary tract also become affected, and the classic eye disease known as xerophthalmia due to vitamin A deficiency ensues. Even a mild vitamin A deficiency is associated with a significant rise in mortality. This association is extremely significant, because vitamin A deficiency is particularly widespread in developing countries, especially in Asia, where as many as 10 million children are found to have xerophthalmia each year.3
Xerophthalmia
The term xerophthalmia is generally used to cover all the ocular manifestations of vitamin A deficiency. Blindness is one of the most serious consequences of vitamin A deficiency. Although it rarely occurs in the United States, it is the major preventable cause of blindness in Asia. The xerophthalmia of vitamin A deficiency is staged as shown in Table 135-3.
TABLE 135-3 Staging of the Xerophthalmias
| STAGE | DIAGNOSIS | SIGNS AND SYMPTOMS |
|---|---|---|
| XO | Night blindness | Poor dark adaptation |
| X1A | Xerosis of conjunctiva | Dryness with “lackluster” appearance, thickening, wrinkling, and diffuse pigmentation of conjunctiva |
| X1B | Bitot spots | Usually triangular collections of desquamated, keratinized epithelial cells and mucus |
| X2 | Xerosis of cornea | Dryness of cornea, leading to keratinization and a hazy, milky appearance |
| X3 | Keratomalacia | Ulceration, distortion, and softening of the cornea with eventual perforation and prolapse and infection of the iris |
 Metabolism
Metabolism
Absorption
• The presence of fat, protein, and antioxidants in food
• The presence of bile and a normal complement of pancreatic enzymes in the intestinal lumen
The absorptive efficiency of dietary vitamin A is usually quite high (80% to 90%), with only a slight reduction in efficiency at high doses. In contrast, beta-carotene’s absorptive efficiency is much lower (40% to 60%), and it diminishes rapidly with increasing dosage. Carotene supplements are better absorbed than the carotenes from foods.4
Transport, Storage, and Excretion
Upon reaching the liver, vitamin A is stored primarily in special perisinusoidal lipocytes (Ito cells); the hepatocytes contain only a minor fraction of the total vitamin A stored in the liver. Although small amounts of vitamin A are found in most tissues (Table 135-4), more than 90% of the total body vitamin A content is stored in the liver. It is stored as a lipoglycoprotein complex consisting of 96% retinyl esters and 4% unesterified retinol. The retinyl esters are hydrolyzed by a tightly bound retinyl ester hydrolase, which transfers the released all-trans retinol to intracellular retinol-binding protein (RBP). The bound retinol is then processed through the Golgi apparatus and secreted into the plasma, where it forms a reversible 1:1 molar complex with prealbumin.1,5
TABLE 135-4 Distribution of Vitamin A in Some Human Tissues (mcg/kg)
| TISSUE | VITAMIN A |
|---|---|
| Adrenal gland | 10.4 ± 7.1 |
| Liver | 149 ± 132 |
| Testis | 1.14 ± 1.23 |
| Fat | 1.46 ± 1.55 |
| Pancreas | 0.52 ± 0.28 |
| Spleen | 0.89 ± 0.88 |
| Lung | 0.91 ± 1.89 |
| Thyroid | 0.43 ± 0.33 |
Adequate dietary protein, iron, and zinc are necessary for proper retinol mobilization. The half-lives of RBP and prealbumin are less than 12 hours, making them particularly likely to be deficient during protein-calorie malnutrition or other situations in which protein metabolism is abnormal. A zinc, iron, or vitamin E deficiency also severely impairs vitamin A metabolism, because these nutrients function synergistically in many physiologic processes of vitamin A metabolism (absorption, transport, and mobilization in particular).6,7
Vitamin A metabolites are excreted mainly through the feces (via the bile) and the urine. During periods of deficiency there appears to be an adaptation in utilization, as evidenced by a reduction in the rate of vitamin A catabolism.1,5
Conversion of Beta-Carotene to Vitamin A
Emerging research is revealing that genomic variations that result in impaired conversion of beta-carotene to vitamin A are surprisingly common. Single nucleotide polymorphisms have been identified that result in decreased activity of 15,15’-monoxygenase, the key enzyme responsible for the conversion of beta-carotene to retinol. The R267S and A379V variants are common, with frequencies in the population of 42% and 24%, respectively. Those with both 267S and 379V have a 57% reduction in conversion rate. Those with these variant have increased blood levels of beta-carotene and decreased blood levels of vitamin A.8
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree