Chapter 12 Scientific Basis of Rehabilitation after Anterior Cruciate Ligament Autogenous Reconstruction
FACTORS THAT AFFECT POSTOPERATIVE REHABILITATION AND OUTCOMES
Considerable advances have been made in the treatment of complete anterior cruciate ligament (ACL) ruptures and reconstruction methods since the mid 1980s. These include the appropriate selection of patient candidates and criteria that should be achieved before surgery, such as resolving limitations of knee motion, muscle atrophy, gait abnormalities, pain, and joint effusion. Appropriate graft selection, harvest, implantation, tensioning, and fixation are all paramount to achieving a reliable and desirable outcome, and considerable attention has been devoted to these principles in the orthopaedic literature.18,19,62
Appropriate postoperative rehabilitation after ACL reconstruction is critical to achieve normal knee function and prevent complications such as arthrofibrosis and reinjury. The goals of postoperative therapy are to regain normal knee motion, gait mechanics, lower extremity muscle strength, coordination, proprioception, and neuromuscular indices using exercises and modalities that are not deleterious to the healing graft. Dye introduced the concept of restoring normal knee osseous and soft tissue homeostasis after ACL injury and reconstruction using an appropriate combination of medical and therapeutic measures.45,46 The exercise program should not produce undue forces on the patellofemoral or tibiofemoral compartments or result in chronic joint effusions. Unfortunately, few investigations have studied the effect of specific exercises and treatment modalities commonly used after ACL reconstruction. In fact, entire protocols have appeared and been used extensively based on clinical observations and retrospective analyses instead of prospective, randomized, controlled clinical trials.83,179
One of the difficulties in conducting rehabilitation investigations is the multitude of factors that may affect both the initial and the long-term recovery after ACL reconstruction. In addition, few ACL ruptures are truly isolated in nature, because approximately 80% of patients sustain concomitant bone bruises52,93 and 60% suffer meniscus tears.130,136 The factors that affect recovery include
Critical Points FACTORS THAT AFFECT POSTOPERATIVE REHABILITATION AND OUTCOMES
Goals of Postoperative Therapy
Authors have noted no benefit28,117 and, in some cases, deleterious outcomes120,176,182,205 when ACL reconstruction is performed early after injury before the resolution of limitations in knee motion, muscle atrophy, swelling, and pain. Meighan and coworkers117 conducted one of the few prospective, randomized investigations regarding this issue. These investigators followed 31 patients who received either an early ACL hamstring reconstruction (within 2 wk of the injury) or a delayed reconstruction (8–12 wk) to determine outcomes up to 1 year postoperatively. The early reconstruction group had significantly less knee extension and flexion at 2 weeks postoperatively, and greater deficits in quadriceps isokinetic work and power at 12 weeks postoperatively, than the delayed group. No other differences were noted in outcome throughout the study period. The authors concluded that early reconstruction did not provide any benefit to athletic individuals. Shelbourne and associates182 noted an increased rate of arthrofibrosis in patients who underwent acute ACL reconstruction (within 1 wk of the injury) compared with those in whom the reconstruction was delayed for 21 days or more. Shelbourne and colleagues166,177 have heavily emphasized the need to restore normal knee motion when possible before surgical intervention. The exception is the presence of a mechanical block to extension, such as a bucket-handle meniscus tear or ruptured ACL that is impinged in the intercondylar notch. In these cases, early surgical intervention is warranted to repair the meniscus tear or remove the ACL, regain full knee extension and flexion, and then proceed later with ACL reconstruction.
Modern studies of ACL bone–patellar tendon–bone (B-PT-B) autograft reconstruction typically report low failure rates of approximately 5% to 10%.10,67,129,143,181 Failure is defined as an increase in anteroposterior (AP) displacement of 6 mm or greater compared with the normal contralateral limb or a fully positive (grade II or III) pivot shift test. However, the percentage of grafts that undergo some amount of elongation, resulting in 3 to 5 mm of increased AP displacement or a mildly positive (grade I) pivot shift test, is quite variable and ranges from 5% to 50%.1,7,72,129,143,173,174,206 Therefore, although the overall rate of failure is low, some authors express concern that any amount of abnormal anterior tibial translation may be detrimental to the knee joint over the long term.20,148 Whether graft elongation occurs as a result of technical aspects of the operative procedure, inconsistencies in maturation of the collagenous and bony components of the construct, the rehabilitation program, or a combination of these controllable and uncontrollable factors is unknown. Only one study has been conducted, to our knowledge, that followed knees reconstructed with ACL B-PT-B autografts with serial KT-2000 testing throughout the postoperative period (2 yr), discussed in detail later in this chapter.10
The purpose of this chapter is to review the current knowledge surrounding rehabilitation after ACL autogenous reconstruction. The scientific basis for immediate knee motion, early weight-bearing, specific exercises, and evaluation criteria for return to activity are reviewed. The authors’ ACL postoperative rehabilitation programs formulated from the scientific literature and nearly 3 decades of empirical clinical experience are detailed in Chapter 13, Rehabilitation of Primary and Revision Anterior Cruciate Ligament Reconstruction.
AUTOGENOUS ACL GRAFT MATURATION IN HUMANS
More than 100,000 ACL reconstructions are performed in the United States each year.146 Few studies have been conducted on the maturation process of ligament grafts in humans; only a minuscule sampling (<1%) of B-PT-B and semitendinosus-gracilis (STG) grafts have undergone histologic analysis after implantation. The strong potential for sampling error prevents conclusions on when graft maturation is complete, indicated by when the transplanted tissue resembles a normal ACL’s histologic, structural, biomechanical, and material properties.
Rougraff and coworkers164 performed biopsies on 23 B-PT-B autografts 3 weeks to 6.5 years after implantation. The authors reported that all patients had “clinically stable” knees; however, Lachman, pivot shift, and knee arthrometer data were not provided. The patients were allowed full weight-bearing immediately postoperatively and returned to sports between 3 and 6 months after surgery. The authors described four stages of ligamentization. The first stage occurred during the first 2 postoperative months and was characterized by an increased number of fibroblasts compared with time zero, preservation of mature collagen, and early neovascular ingrowth. The second stage, composed of rapid remodeling, occurred from 2 to 10 months postoperatively. A rapid increase in the number of metabolically active fibroblasts and replacement of approximately two thirds of mature collagen with immature matrix was noted. The third stage, in which the fibroblast count slowly decreased, collagen matrix matured, and vascularity decreased, occurred from 1 to 3 years postoperatively. In the final stage, the grafts were noted to be less cellular and vascular and appeared similar to those of control ACLs. The study did not include electron microscopy to evaluate the pattern of collagen fibril diameters in the grafts.
Critical Points AUTOGENOUS ACL GRAFT MATURATION IN HUMANS
B-PT-B Autografts: Four Stages of Ligamentization
B-PT-B and STG Autografts
B-PT-B, bone–patellar tendon–bone; STG, semitendinosus-gracilis.
Rougraff and Shelbourne165 performed biopises on nine B-PT-B autografts between 3 and 8 weeks after implantation. All patients followed an accelerated rehabilitation protocol179 and agreed to the biopsy for investigational purposes. All specimens showed evidence of survival of portions of the original tendon. Vascular invasion was present at 3 weeks postoperative, with increased cell counts compared with controls found in all samples. All specimens had areas of acellularity and degeneration; however, these were small (no more than 30% of the biopsied area) in comparison with the areas of vascularity and normal-appearing tendon tissue.
Petersen and Laprell153 examined the insertion of B-PT-B and STG autografts to bone in 14 knees that required revision ACL reconstruction. The time from the primary ACL reconstruction to revision ranged from 6 to 37 months. The hamstring specimens revealed a fibrous insertion of the graft into the periphery of the tibial bone tunnel. The patellar tendon (PT) specimens taken from the femoral tunnel showed fibrocartilage at the bone-tendon interface and resembled the chondral insertion of a normal ACL.
Johnson94 performed biopsies on 20 STG ACL grafts between 20 days and 44 months postoperative. All patients required follow-up arthroscopy for various symptoms, at which time biopsy samples were taken from the tendon graft and surrounding fibrous tissue. The reconstructed ACL was a composite of these two distinctly different tissues, which had diverse histologic properties. The 3-month postoperative tendon samples appeared normal histologically, with no signs of inflammation and organized collagen bundles. Specimens taken beyond 3 months showed continued tissue maturation and crimping of collagen bundles similar to those of the native ACL. Specimens taken from the fibrous tissue had a disorganized cellular pattern and hypervascularity at 3 months postoperative and, although with time they developed increasing amounts of collagen, never resembled a normal tendon.
Beynnon and associates23 examined the knees of a B-PT-B autograft recipient 8 months after implantation after the patient had died of causes unrelated to the knee joint. The patient’s rehabilitation program included immediate continuous passive motion (CPM) from 0° to 90°; however, he underwent a prolonged period of partial weight-bearing for 12 weeks for unknown reasons. At 4 months postoperative, he was examined clinically and had a stable reconstruction. The postmortem examination revealed 6 mm of increased AP displacement, a 13% reduction in ultimate failure loads, and a 53% reduction in energy absorbed at failure compared with the contralateral native ACL. Histologic analyses were not conducted. Interestingly, the ultimate failure load of the patient’s contralateral native ACL of 1015.6 N was well below the expected value of 1725 ± 269 N.132
Delay and colleagues40 inspected a retrieved whole B-PT-B autograft 18 months after implantation in a patient who died from a traumatic injury. Whereas the graft was mainly cellular and resembled a normal ACL morphologically, there were areas of acellularity deep within the tendinosis portion and also in the femoral intra-articular region where the autograft emerged from the tibial tunnel. The graft had been placed in an over-the-top position that the authors speculated could have caused increased stresses, resulting in inconsistent remodeling.
Falconiero and coworkers53 obtained biopsies of 35 B-PT-B and 8 STG autografts in 48 patients from 3 to 120 months after implantation. The rehabilitation program the patients followed was not described. Significant differences were noted in vascularity, cellularity, and fiber pattern between grafts that were examined 3 to 6 months postoperative and those that were observed greater than 12 months after implantation. Grafts that were biopsied 7 to 12 months postoperatively were described as transitional in terms of vascular maturity. No difference was found between fiber patterns and vascularity in grafts that had been implanted 7 to 12 months before the biopsy and those that had been implanted more than 12 months before the biopsy. The authors concluded that whereas graft maturity generally occurs over a 12-month period, some autografts may reach full maturation even earlier.
Although it appears from the literature reviewed that ACL autografts undergo increasing maturation postoperatively, the small sample size of examined grafts and the host of both intrinsic and extrinsic variables affects this process and prevents definitive conclusions. Questions remain regarding whether the healing process produces a graft that has histologic, ultrastructural, and biomechanical characteristics equivalent to those of a native ACL. It is probable that the delicate nature and microgeometry of native ligament fibers is never regained, but that the ligament functions more as a checkrein to resist gross knee displacements rather than replicating normal kinematics.89 Recent studies have demonstrated that even though B-PT-B and STG autograft reconstructions frequently restore normal AP displacements as measured with a KT-2000 arthrometer (<3 mm side-to-side difference), normal knee kinematics may not be restored, causing abnormal joint motions when the knee is subjected to weight-bearing activities.148 The delay in maturation of ACL allografts and the potential effects on postoperative recovery are discussed in Chapter 5, Biology of ACL Graft Healing.
IMMEDIATE KNEE MOTION
The scientific basis supporting immediate knee motion after ACL reconstruction is well established.18,78,87,131,134,135,158,163 Early knee joint motion decreases pain and postoperative joint effusions, aids in the prevention of scar tissue formation and capsular contractions that can limit normal knee flexion and extension, decreases muscle disuse effects (Fig. 12-1), maintains articular cartilage nutrition, and benefits the healing ACL graft.* There is a consensus in the medical community that immobilization is detrimental to the knee joint structures and may result in a permanent limitation of knee motion, prolonged muscle atrophy, patella infera, and articular cartilage deterioration.33,70,95,139–141,150,170
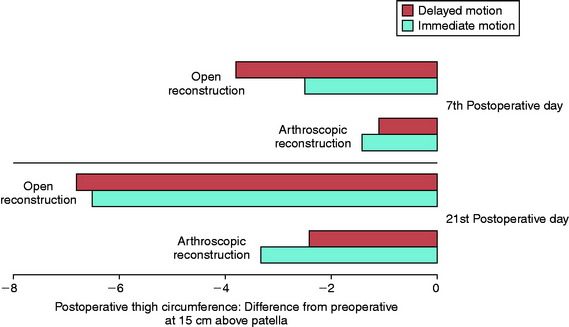
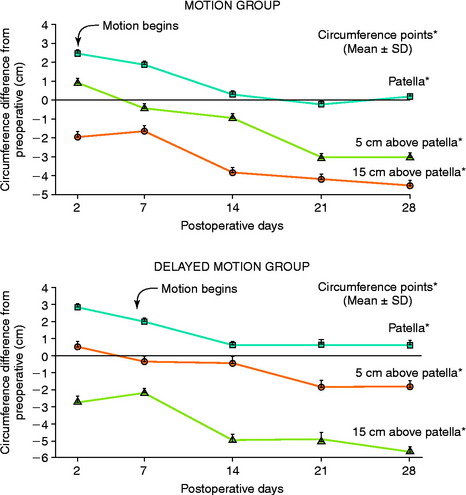
In 1983, Noyes and associates133 first published recommendations for immediate knee motion after ACL B-PT-B autograft reconstruction after a successful 4-year experience with this program. Advances in knee ligament reconstructive and fixation techniques allowed the controlled movement of the knee from 0° to 90° and did not disrupt the healing graft. In 1987, these authors134 published one of the first clinical studies that assessed joint effusion, joint motion, muscle atrophy, and the integrity of ACL grafts in a series of 18 patients who underwent ACL allograft reconstruction. Patients were randomly placed into either an immediate motion group that began CPM on the 2nd postoperative day or a delayed motion group that initiated passive and active-assisted flexion and extension exercises on the 7th postoperative day. All other parameters of the postoperative rehabilitation program were identical for the two groups, including exercises, bracing, and modalities. The results showed no differences between groups for knee joint effusion, the amount of knee flexion and extension achieved by 3 months postoperative, decrease in thigh circumference loss (Fig. 12-2), and frequency and type of pain medication used while in the hospital. There was no difference between groups in AP displacements at 12 months postoperative. Patients in whom the ACL graft had been implanted through an arthrotomy had significantly greater thigh circumference loss compared with those who had an arthroscopic-assisted technique, noted by the 7th postoperative day.
Richmond and colleagues in 1991158 compared the effectiveness of CPM when used for the first 4 postoperative days to utilization during the first 14 days in 19 patients who underwent ACL B-PT-B autograft reconstruction. CPM was used for at least 6 hours a day. All patients also performed passive motion exercises from 0° to 90° three times daily. There were no differences between the groups at 6 weeks postoperative for swelling, thigh girth measurements, knee flexion and extension, time to regain full motion, or AP displacements. Immediate motion was concluded to be safe and not deleterious to the healing grafts in the initial postoperative period. No long-term benefit of CPM was demonstrated.
Rosen and coworkers163 followed 75 patients who had ACL B-PT-B autogenous reconstruction and were sorted into three groups based on a knee motion and rehabilitation program. Patients who began active-assisted early knee motion exercises had similar results in regard to knee motion achieved at week 1 and at monthly intervals for the first 6 postoperative months as those who used CPM an average of 7 hours a day for 4 weeks postoperatively. There were no deleterious effects of early motion on AP displacements at the final follow-up evaluation, 6 months postoperative. The authors concluded that the CPM device offered no advantage and increased the cost of physical therapy by $1,800.
Beynnon and associates21 conducted in vivo measurements of the elongation of B-PT-B autografts in 20 patients immediately after implantation. In 11 knees, the length of the graft increased after 14 knee flexion-extension motion cycles, and in 9 patients, the graft length decreased. The predicted maximum increase in anterior tibial translation after 20 knee motion cycles was only 1 mm.
The current authors135 followed 207 knees that underwent arthroscopically assisted ACL allograft reconstruction and immediate knee motion. All patients began CPM for 8 to 10 hours a day immediately after surgery. In addition, passive and active-assisted motion exercises were initiated on the 2nd postoperative day. Partial weight-bearing was allowed immediately postoperative and was gradually progressed to full by 4 to 6 weeks based on resumption of normal gait mechanics. Any patient who demonstrated a limitation of knee flexion or extension from predetermined goals was placed into a specific treatment program as early as the 7th postoperative day. The program included overpressure exercises and modalities initially (Fig. 12-3), followed, if required, by a serial extension cast program (Fig. 12-4) and in rare instances, arthroscopic débridement of scar tissues (Fig. 12-5). Of the 207 knees, 189 (91%) regained at least 0° to 135° of motion without additional intervention. Eighteen knees were placed into the phased treatment program, of which 14 regained normal motion and 2 lacked 5° of full extension. Two other patients who had not complied with the rehabilitation program had a permanent significant limitation of motion. The incidence of postoperative motion problems was related to concomitant surgical procedures: medial collateral ligament (MCL), 23%; meniscus repair, 12%; iliotibial band extra-articular procedure, 10%; and isolated ACL, 4%.
In a second study from the authors’ center,131 443 knees that had an arthroscopic-assisted ACL B-PT-B autograft reconstruction were followed to determine the effectiveness of an immediate knee motion program and treatment protocol for limitations of flexion and extension. A CPM device was not used in this group of patients. A normal range of motion (ROM) was achieved in 436 knees (98%) and a mild limitation of extension of 5° was detected in 7. Treatment intervention was required in 23 knees, all of which achieved normal knee motion. The 7 patients whose knees had mild losses of extension refused additional treatment. Only 3 knees (<1%) required an arthroscopic lysis of adhesions that, combined with an in-patient epidural program, resulted in successful resolution of the motion limitations. The incidence of knee motion problems based on concomitant surgical procedures was 22% for MCL repairs, 18% for patellar realignment procedures, 8% for meniscus repairs, and 6% for isolated ACL reconstructions.
In 2002, Henriksson and coworkers78 compared the outcome of 5 weeks of immobilization with early knee ROM exercises in 45 patients after ACL B-PT-B autograft reconstruction. Patients in the early ROM group began motion exercises from 0° to 90° from the 7th postoperative day. The early ROM group demonstrated greater extension and flexion for the first 20 weeks. Patients in the immobilization group generally required more physical therapy visits to achieve full knee motion.
EARLY WEIGHT-BEARING
Whereas many authors have recommended early partial or full weight-bearing immediately after ACL reconstruction, few studies have examined this factor and its potential effect on both short- and long-term recovery. The effect of immediate full weight-bearing on the outcome in knees with noteworthy articular cartilage damage or those in which major concomitant operative procedures were required is unknown. In addition, gait abnormalities that may ensue from immediate full weight-bearing due to pain, knee joint effusion, and muscle weakness have not been examined. It does appear from the literature and the authors’ experience that immediate partial weight-bearing is safe and not deleterious to the healing graft.10
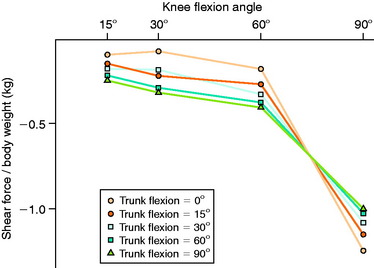
In 1991, Ohkoshi and associates144 recommended early weight-bearing exercises after ACL reconstruction based on results of a study in which shear forces on the tibia during standing were predicted from an analytical model at various trunk and knee flexion angles (Fig. 12-6). Although weight-bearing was normally prohibited in the initial postoperative period at the time of this publication, these investigators reported that the calculated shear forces were negative in all positions, with increasing posterior drawer forces found as trunk flexion angles increased (at knee flexion angles of 30° and 60°). Co- contraction of the quadriceps and hamstrings was observed at all knee and trunk flexion angle positions, with increasing hamstrings activity measured with increasing trunk flexion angles. Exercises performed in the standing position with the knees flexed and the trunk anteriorly flexed, such as half-squatting, were believed not only to be safe but also to have the potential advantages of increasing muscle strength, endurance, and proprioception and decreasing bone atrophy.
Critical Points EARLY WEIGHT-BEARING
Ohkoshi et al.144
Tyler and colleagues in 1998203 evaluated the effect of immediate full weight-bearing after ACL B-PT-B autograft reconstruction on ROM, patellofemoral pain, and AP displacement in a randomized, controlled trial. The results of 25 patients who were allowed immediate full weight-bearing were compared with those of 20 patients in whom no weight-bearing was allowed for 2 weeks postoperative. All patients performed immediate ROM exercises and followed an identical rehabilitation protocol (which was not detailed). There was no difference between groups for AP displacement, ROM, and vastus medialis obliquus (VMO) electromyographic activity at follow-up an average of 7 months postoperatively. Patients in the delayed weight-bearing group had a higher incidence of anterior knee pain than those in the immediate weight-bearing group (35% and 8%, respectively). Extension deficits of 5° or greater were reported in 14% of patients in the delayed weight-bearing group and in 20% in the immediate weight-bearing group. There was no correlation between loss of knee extension and anterior knee pain. Patients in the immediate weight-bearing group had significantly greater VMO electromyographic activity at 2 weeks postoperative compared with those in the delayed weight-bearing group, and the authors hypothesized that this aided in lowering the incidence of anterior knee pain by preventing the reflex inhibition of the VMO. Approximately 15% in each group had greater than 5 mm of increased AP displacement at follow-up.
POSTOPERATIVE BRACING
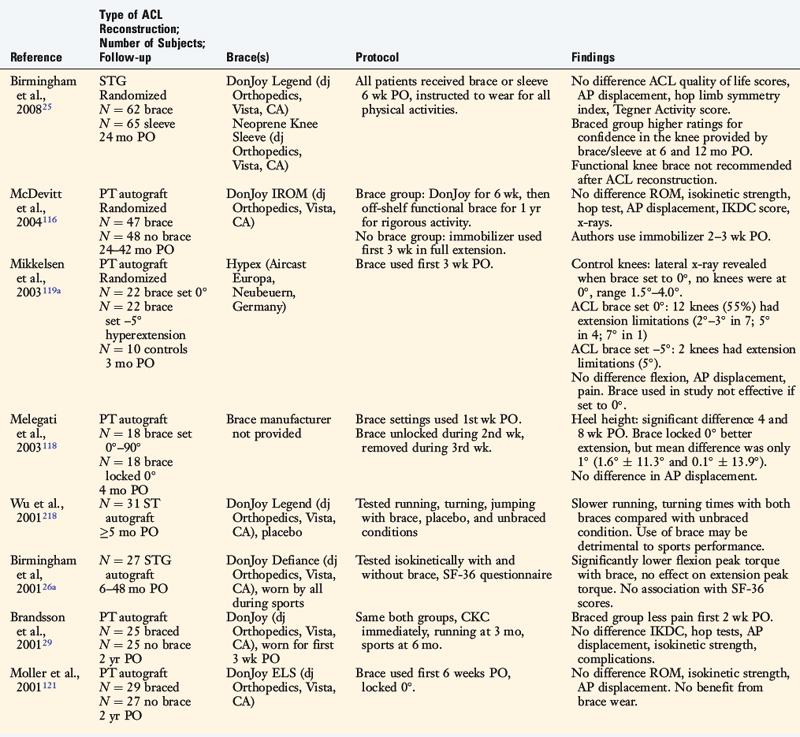
The effects of postoperative and functional braces on pain, return of normal knee motion, complications, sports activity performance, balance, and proprioception have been assessed by many investigators (Table 12-1). Most studies report that postoperative braces do not provide significant benefit in the amount of time postoperatively that normal range of knee motion is achieved, isokinetic lower extremity muscle strength, AP displacements, lower limb symmetry assessed by single-leg hop tests, and functional knee rating and activity level scores.* Negative effects of braces have been reported regarding quadriceps strength,160 running and turning times,218 and hamstrings isokinetic peak torque.26 One study reported that a functional brace improved standing balance, but not more difficult tasks such as balance after a forward hop.26 In regard to proprioception, two investigations reported significant, although small, improvements in static joint sense of 1° to 2° with brace utilization (Table 12-2).26,219 The clinical impact of these improvements remains questionable. Risberg and coworkers159 reported no improvement in the threshold to detect passive motion with application of a functional brace an average of 24 months after ACL B-PT-B autograft reconstruction. These investigators also found no impairment in proprioception in the ACL-reconstructed knees compared with those of controls.
Critical Points POSTOPERATIVE BRACING
Most studies: postoperative braces do not provide significant benefit:
Significant variation among surgeons surveyed on the use of braces postoperatively.
Authors recommend postoperative braces in complex multiligament reconstructions.
Marx and associates114 published a survey of data collected from 1998 to 1999 conducted by the American Academy of Orthopaedic Surgeons. Significant variation was reported among surgeons on the use of braces in the postoperative period, with half of the surgeons indicating they prescribed a brace for the first 6 postoperative weeks and the remaining half indicating no brace utilization. Surgeons who performed more ACL reconstructions reported less brace use. Approximately 60% of all surgeons responded that they recommended a brace for sports participation.
Beynnon and colleagues18 concluded from a review of this topic that there “appears to be a consensus among investigators that, during the early phase of recovery, the use of a rehabilitation brace results in fewer problems with swelling, lower prevalence of hemarthrosis and wound drainage, and less pain compared to rehabilitation without a brace; however, at longer-term follow-up, rehabilitation bracing does not appear to have an effect on clinical outcome.” The authors recommended postoperative bracing in complex multiligament reconstructions for protection of healing grafts and unloading braces in medial and posterolateral reconstructions to allow early partial weight-bearing.
LOWER EXTREMITY MUSCLE STRENGTH ATROPHY AND RECOVERY AFTER SURGERY
Lower extremity muscle weakness represents an unresolved problem after ACL reconstruction.* Studies have reported that the magnitude of quadriceps atrophy and strength loss exceeds 20% and 30%, respectively, in the first few months postoperatively.54,55,68,117,160 Deficits in semitendinosus and gracilis muscle volume of 10% and 30%, respectively, have been noted after STG reconstructions.86,211 Conflicting data have been published regarding the return of normal quadriceps and hamstrings muscle strength 6 months or longer postoperatively.
Critical Points LOWER EXTREMITY MUSCLE STRENGTH RECOVERY AFTER SURGERY
Lower extremity muscle weakness is an unresolved problem after ACL reconstruction.
Pathophysiology of the loss of muscle size and strength after major knee surgery is unknown.
The pathophysiology of the loss of muscle size and strength after major knee surgery remains speculative. Konishi and coworkers101 proposed abnormal gamma loop function in the quadriceps muscles due to the lack of normal sensory function in the reconstructed ACL as a potential explanation. Other authors advocated that the loss of native ACL mechanoreceptors, which have an important role in enhancing normal activity of gamma motoneurons, was a potential cause of this problem.90–92,187,192,193 Young and associates224 attributed quadriceps atrophy to a reduction in muscle fiber size from histologic findings in 14 patients. Indeed, some clinical investigators have suggested that fast-twitch muscle fibers (accounting for ~60% of the rectus femoris muscle187) become hypotrophic after ACL injury and reconstruction.13,48 Residual abnormal anterior tibial displacement after reconstruction may play a role in persistent quadriceps weakness.84 Finally, the magnitude of preoperative muscle weakness may inhibit the ability of a patient to regain normal strength and endurance after surgery.
The question of whether differential atrophy exists between type I and II muscle fibers in the quadriceps remains unclear. Haggmark and colleagues71 obtained muscle biopsies before surgery and after 5 weeks of postoperative immobilization in nine patients who underwent ACL reconstruction. A decrease in the cross-sectional area (CSA) of type 1 fibers and a reduction in the oxidative capacity of quadriceps muscle were noted, indicating a rapid fall in aerobic capacity. The authors concluded that selective atrophy of type 1 fibers occurred and that type 2 fibers were not significantly affected. In contrast, Gerber and coworkers63 studied the changes in the quadriceps muscles of 41 chronic ACL-deficient knees and reported that neither histologic nor electron microscopy showed any selective loss of muscle fiber type. These authors concluded that there was no scientific rationale for selective rehabilitation of type 1 or type 2 fibers and that vigorous training of quadriceps muscles was essential after ACL injuries. Lorentzon and associates109 studied the morphology of muscle types with biopsy and isokinetic testing of 18 male subjects with chronic ACL deficiency. These authors did not find any evidence of selective type 1 or type 2 fiber atrophy. In addition, there was no correlation between isokinetic test data and muscle size or morphologic changes. These authors concluded that decreases in muscle strength cannot be explained by muscle atrophy or structural change. They speculated that nonoptimal activation of muscles during voluntary contractions is the most causative mechanism of strength decrease found in patients with chronic ACL deficiency.
A comparison of high- and low-intensity electrical muscle stimulation (EMS), combined with an intensive rehabilitation program, on recovery of quadriceps strength and gait mechanics was conducted in two investigations.189,191 A variety of ACL grafts implanted among 110 patients were included in these studies, including Achilles tendon allografts, B-PT-B allografts, STG autografts, and B-PT-B autografts. Although high-intensity EMS significantly increased rectus femoris strength recovery (compared with the opposite limb) compared with the low-intensity EMS protocol, this effect was not as pronounced for patients who underwent ACL PT autogenous reconstruction. The strength of the quadriceps correlated with flexion and extension excursions of the knee during stance. Patients who were treated with high-intensity EMS walked with more normal excursions, which the authors attributed to the gains in quadriceps strength. Low-intensity EMS, delivered via portable units for home use, was ineffective in promoting return of normal quadriceps strength.
Fitzgerald and colleagues58 reported modest (effect size, 0.48) improvements in quadriceps isometric peak torque 16 weeks after ACL reconstruction when a “modified” EMS-training protocol was incorporated into the postoperative rehabilitation program of 21 patients. EMS was delivered with the patient positioned supine, lying passively with the knee in full extension. This protocol was modified from previously reported training methods, in which high-intensity EMS was delivered with the patient seated in an isokinetic dynamometer, the knee positioned in approximately 60° of flexion, and the patient actively contracting the quadriceps during application of the electrical current.41,189,190 The EMS protocol was modified for patients who experienced patellofemoral pain during the training sessions. The protocol involved 10 contractions to maximum patient tolerance, conducted twice weekly for 16 weeks. At 12 weeks postoperative, the experimental group demonstrated significantly greater quadriceps isometric peak torque compared with that of a matched group of patients who did not receive EMS training (75.9 ± 16.8 and 67.0 ± 19.9, respectively; P < .05). However, no significant differences were measured at 16 weeks postoperative between these groups. A greater proportion of the EMS-trained subjects initiated agility training at 16 weeks compared with the control group (62% and 32%, respectively; P < .05). Still, the authors concluded that the original high-intensity EMS-training protocol was preferred when possible based on patient tolerance and equipment availability.
Investigators and clinicians have recently questioned whether rehabilitation programs should focus more efforts on eccentric training to reduce the early postoperative loss of muscle CSA, volume, and strength following ACL reconstruction.64–66,105 In an exhaustive review of the literature on the effectiveness of concentric, eccentric, or combined concentric-eccentric training in normal, uninjured subjects, Wernbom and coworkers207 concluded that there was no evidence to support one mode of training over another with regard to achieving superior muscle hypertrophy. These authors assessed published training programs according to frequency, intensity, and duration of work on CSA and volume of the quadriceps and elbow flexor muscle groups. The observation was made that training protocols to induce muscle hypertrophy may differ from those designed to produce maximum strength.
Eccentric training is believed by some authors to be superior to concentric training owing to its potential to overload the muscle and produce greater increases in muscle size and strength.64,66 There are concerns with high-force eccentric training after ACL reconstruction, including the potential for inducing damage to the muscle and healing graft. A repeated, gradual, and progressive exposure to this type of training was successfully accomplished by Gerber and associates64 who conducted eccentric training in patients who had either ACL B-PT-B autograft (N = 20) or STG autograft (N = 20) reconstruction. The patients were randomly assigned to either a 12-week program of eccentric exercises or a standard rehabilitation program. All patients followed a similar protocol for the first 3 postoperative weeks that emphasized regaining full knee motion and basic quadriceps function. Then, patients in the eccentric exercise group initiated progressive exercises using a recumbent eccentric ergometer. The duration and intensity of the negative-work training gradually increased throughout the 12-week period. The patients underwent magnetic resonance imaging (MRI) 3 and 15 weeks postoperative.
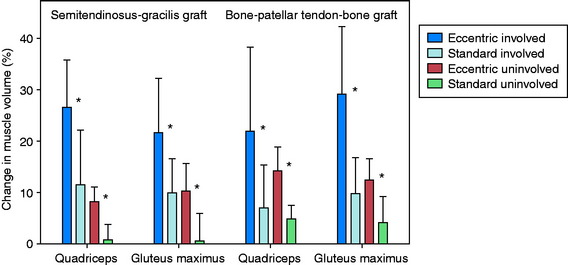
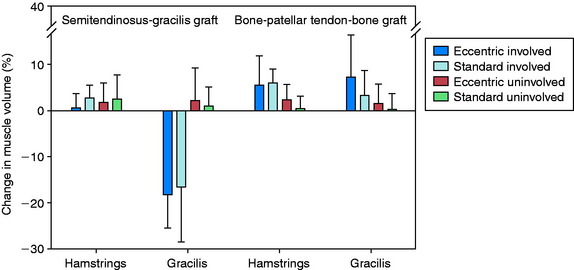
The volume and CSA measurements of the quadriceps in the reconstructed knees improved significantly (P < .001) in both groups (Fig. 12-7) between the pre- and the post-training time periods. The increases in these measurements were significantly greater (P < .001), by more than twofold, in the eccentrically trained patients than in the standard rehabilitation group. In the eccentric group, quadriceps volume increased 23.1 ± 12.9% and peak CSA increased 24.2 ± 12.6%. In comparison, in the standard rehabilitation group, quadriceps volume increased 8.8 ± 9.3% and peak CSA increased 9.3 ± 9.4%. The increase was not related to the type of graft used for the ACL reconstruction. Significant increases in these measurements were also found in the quadriceps in the noninvolved side.
There were no significant differences between the training groups in the improvement in volume and CSA of the hamstring muscles (Fig. 12-8). When analyzed according to graft type, significant improvements were noted in these measurements in the B-PT-B autograft patients (P ≤ .006), but not in the STG autograft patients. A reduction in gracilis muscle volume of nearly 20% was found 3 weeks postoperative in the STG group, which increased to a deficit of 35% by 15 weeks postoperative. The authors concluded that the gradual progressive eccentric exercise program safely and effectively improved quadriceps structure in comparison with a standard rehabilitation program. Whether the short-term results of this investigation will demonstrate longer-term benefits are unknown.
Shaw and colleagues175 conducted a prospective, blinded, randomized trial to determine the effectiveness of instituting quadriceps exercises immediately after ACL reconstruction on a number of parameters. A total of 91 patients who underwent either a B-PT-B or an STG autograft reconstruction were randomized into either the quadriceps exercise–training group or the no-quadriceps exercise–training group. The quadriceps training included straight leg raises and isometric quadriceps contractions performed three times daily for the first 2 postoperative weeks. After 2 weeks, all patients were entered into a similar rehabilitation program. At 6 months postoperative, no difference was found between groups for isokinetic quadriceps strength or lower limb symmetry on functional hop testing.
It is important to note that several authors have documented quadriceps muscle inhibition from an experimentally induced knee joint effusion.147,194,199,200 This problem has been documented in separate studies during walking, jogging, and landing from a jump. Therefore, it is imperative that knee joint effusion be avoided and, if noted, treated immediately to lessen its deleterious impact on quadriceps function.
OPEN VERSUS CLOSED KINETIC CHAIN EXERCISES: BIOMECHANICAL, IN VIVO, AND CLINICAL STUDIES
The process of graft maturation and healing is assumed to be influenced by strains and forces applied to the ACL during weight-bearing and exercises. Whereas a general consensus exists that some strain is necessary to promote the process of ligamentization,20 questions remain regarding the amount of load that is safe and the amount that may produce graft elongation. In addition, ligamentization (the return to normal native ACL characteristics) has never actually been shown to occur with graft remodeling and healing. Since the mid 1980s, investigations have attempted to measure, through either analytical or direct means, force and strain incurred on the ACL during common exercises used in rehabilitation. These exercises are typically referred to as either closed kinetic chain (CKC) or open kinetic chain (OKC). During CKC exercises, the foot is fixed to a platform or surface, the motion at the knee joint is accompanied by predictable motions at the hip and ankle joints, and the entire limb is loaded such as during a leg press or squat. In OKC exercises, the foot is mobile and not fixed to a surface, and motion at the knee joint occurs independent of motion at the hip and ankle joints. Common examples include the leg extension and hamstring curl exercises.
Cadaver Studies
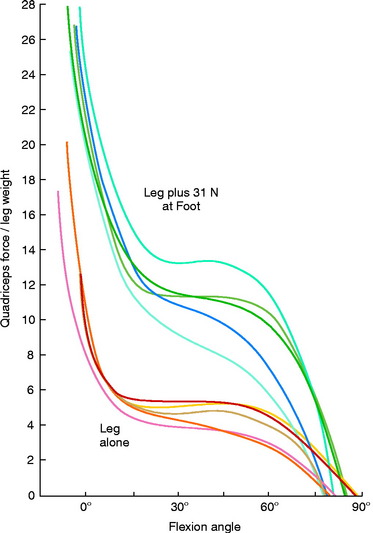
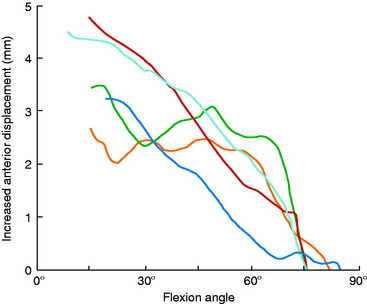
Grood and coworkers69 biomechanically examined the knee extension exercise in cadavers to determine the conditions in which the ACL was loaded, the quadriceps force that developed during knee flexion-extension, and the forces that were incurred on the extensor mechanism as the knee was extended. Both ACL-intact and ACL-sectioned conditions were created, and the OKC knee extension exercise was simulated both without resistance and with 31 N (7 lb) of resistance applied with an ankle weight at the foot. Although the quadriceps force required to extend the knee remained at a constant value of 177 N between 50° and 15°, it rose rapidly to 350 N at 0° (Fig. 12-9). The addition of resistance doubled the quadriceps force that was required to extend the knee, resulting in large muscle forces on the patellofemoral joint. There was no change in the quadriceps force required to extend the knee when the ACL was removed. However, loss of the ACL resulted in an increase in anterior tibial displacement from 0° to 30° of extension without resistance. The addition of 31 N of resistance resulted in further increases in anterior tibial displacement throughout the range of extension, with a mean increase of 3.8 mm at 15° (Fig. 12-10). The authors concluded that the range of 0° to 30° of flexion should be avoided during rehabilitation of ACL-deficient or ACL-reconstructed knees and in patients with patellofemoral symptoms.
Critical Points OPEN VERSUS CLOSED KINETIC CHAIN EXERCISES
Cadaver Studies
Effect of Open and Closed Kinetic Chain Exercises on Anterior Tibial Displacement in Intact and ACL-deficient Knees
Calculated ACL Forces, Tibiofemoral Compressive Forces, and Muscle Forces in Human Subjects
Measurement of In Vivo ACL Strain in ACL-deficient and Intact Knees during Common Open and Closed Kinetic Chain Exercises
Muscle Recruitment Patterns during Common Open and Closed Kinetic Chain Exercises
Comparative Clinical Studies
Bynum et al.31: Prospective, Randomized Study: OKC or CKC Program after ACL B-PT-B Autograft
Mikkelsen et al.119: Prospective, Randomized Study: CKC or Combined CKC-OKC Program (Began OKC Exercises Week 6) after ACL B-PT-B Autograft
Three investigations same laboratory: No benefit from OKC short-term postoperative period.
Beynnon et al.24: Prospective, Randomized Double-Blind Study of Two Rehabilitation Programs (“Accelerated” Weight-Bearing, OKC, and Traditional) after ACL B-PT-B Autograft
Renstrom and associates157 measured the strain of cadaver ACL specimens under simulated isometric contractions of various muscle groups. The authors reported that isolated hamstring contractions decreased ACL strain relative to normal passive strain at all knee flexion angles tested (0°–120°). Isolated quadriceps isometric and isotonic contractions increased ACL strain from 0° to 45° of flexion (Fig. 12-11); the greatest magnitude (5% above the normal passive strain) was measured at full knee extension. Isometric co-contractions of the quadriceps and hamstrings also increased ACL strain from 0° to 30°, with the maximum increase of 5% measured at 0° and 15° of flexion. The authors concluded that isolated quadriceps isometric and isotonic exercises, and isometric co-contraction exercises, could produce potentially harmful forces on healing ACL grafts.
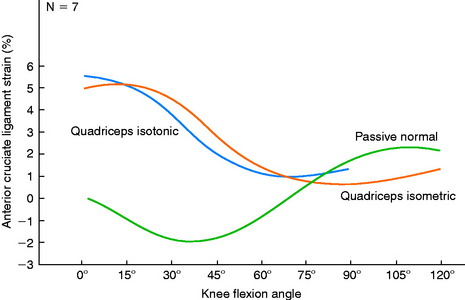
FIGURE 12-11 Mean passive normal, simulated quadriceps isometric, and simulated quadriceps isotonic strain patterns.
(From Renstrom, P.; Arms, S. W.; Stanwyck, T. S.; et al.: Strain within the anterior cruciate ligament during hamstring and quadriceps activity. Am J Sports Med 14:83–87, 1986.)
More and colleagues122 used a cadaver model that incorporated quadriceps and hamstrings muscle loads to examine knee kinematics and ACL loads during the squat exercise. In the intact knees, the addition of a hamstrings load resulted in a significant reduction of anterior tibial translation and internal tibial rotation during flexion. After the ACL was sectioned, the amount of anterior tibial translation increased significantly between 15° and 45° during the squat compared with the intact knee. After ACL reconstruction, maximal graft tension was measured at full extension. During the squat exercise, the addition of a hamstrings load caused a significant decrease in graft load that was most evident between 15° and 45°. The authors concluded that the squat exercise may be safe in the early postoperative period after ACL reconstruction.
Effect of OKC and CKC Exercises on Anterior Tibial Displacement in Intact and ACL-deficient Knees
Several investigators measured AP tibial displacements in ACL-intact and ACL-deficient knees during various OKC and CKC exercises, including active knee extension and squatting.97,104,220 It is important to note that the amount of AP displacement is influenced by many factors including muscle activation, joint compression forces, geometry, and ligament restraints, primarily the ACL. Overall, significantly greater anterior tibial displacements were found in the ACL-deficient knees during OKC activities than during CKC exercises.
Yack and coworkers220 measured the amount of anterior tibial displacement in 11 patients with unilateral ACL-deficient knees during an OKC exercise (resisted knee extension) and a CKC exercise (parallel squat). The average anterior tibial displacement during the squatting exercise (~7.5 mm) was significantly less in the ACL-deficient knees than that measured in the knee extension exercise from 66° to 10° (~14 mm). There was no difference in anterior tibial displacement between these exercises in the contralateral normal knees. The conclusion was reached that CKC exercises produced significantly less stress to the ruptured ACL.
In a second study, Yack and associates221 measured anterior tibial displacement in 14 patients with unilateral ACL-deficient knees during an OKC position and during progressive weight-bearing CKC positions. The progressive weight-bearing protocol included 25%, 50%, 75%, and 100% of each subject’s body weight (BW) placed on the foot during a squat held at 20° of flexion. Hamstring muscle activation was controlled to be less than 10% of its maximum activation level during all test conditions. The amount of anterior tibial translation induced during an 89-N Lachman test was measured and compared with that produced during the weight-bearing and non–weight-bearing conditions. The authors reported that anterior tibial translation was significantly less during all weight-bearing conditions than that measured during the Lachman test and the non–weight-bearing conditions. The clinical implication was that rehabilitation after ACL reconstruction could institute weight-bearing exercises without causing excessive strain to the passive restraints. Progressive loading during non–weight-bearing activities should be delayed until the later stages of rehabilitation.
Similar findings were reported by Kvist and Gillquist,104 who measured anterior tibial translation and lower limb muscle activation in patients with ACL-intact and ACL-ruptured knees during active knee extension (with and without resistance) and squatting exercises in which the center of gravity was placed either over, behind, or in front of the feet. The CKC squat exercises produced less anterior tibial translation than the knee extension in the ACL-deficient knees. In the normal knees, anterior tibial translation increased with increasing loads in all of the exercises except during the squats with the center of gravity behind the feet. This activity produced less translation than did all of the other exercises in both the normal and the ACL-deficient knees. Overall, hamstring muscle activity was low in all knees during both the OKC and the CKC exercises, but co-activation of the gastrocnemius and quadriceps muscles was noted. The authors concluded that the CKC exercises caused increased joint compression forces and co-activation of the gastrocnemius and quadriceps muscles, thereby resulting in decreased anterior tibial displacements in ACL-deficient knees.
Jurist and Otis97 measured anterior and posterior tibial displacement in five knees with intact ACLs to determine the effects of knee flexion angle (30°, 60°, and 90°) and the position of external resistance (proximal, middle, or distal on the long axis of the tibia) during isometric contractions. The results demonstrated that a proximal position of external load produced posterior displacement of the tibia. The authors concluded that quadriceps muscle strengthening could be safely initiated between 90° and 30° of flexion in ACL-reconstructed knees early postoperatively.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree