CHAPTER 23 Revision Anterior Cruciate Ligament Reconstruction
Reconstruction of the torn anterior cruciate ligament (ACL) has seen a significant increase in volume over the past 3 decades. It is currently one of the most common procedures performed by orthopaedic surgeons in the United States and across the globe. Current estimates cite approximately 250,000 procedures performed annually in the United States. Although the most of these patients have a fairly uneventful recovery and return to sport and/or work, there are patients who present as clinical failures, with three categories represented by recurrent instability, loss of motion, and pain.1 With the ever-increasing numbers of primary surgeries performed, it follows that the numbers of failed primary procedures will subsequently increase. Orthopedic surgeons will be required to tackle these challenging situations.
Although the causes of failure may be multifactorial, these failures may be broadly classified into three categories: loss of motion, recurrent instability, and pain.2 Another potential category may be progressive joint deterioration without the above findings after ACL reconstruction. Any of these factors may play a role in the failure of a reconstruction, but the final goal of revision reconstruction remains constant—provide a stable and functional ACL that most accurately reproduces the normal knee kinematics, allow the individual to return to an active lifestyle, and potentially decrease the development of early osteoarthritis.3,4
ANATOMY
Although commonly overlooked throughout the past 1 to 2 decades, recent anatomic studies have further defined our precise understanding of the anatomy of the native ACL, grossly and arthroscopically.5,6 Recognition of the ACL as a functional two-bundle ligament comprised of distinct anteromedial and posterolateral bundles, as well as their relationship to bony insertion site anatomy, has led to a revolution in discussing ACL reconstruction techniques.7 It is now understood that re-creation of the native anatomy of the ACL may lead to improved kinematic and functional results. Prior nonadherence to these principals may be a major root cause of many primary reconstruction failures.8
Causes of Reconstruction Failure
Loss of Motion
Loss of motion after reconstruction of the ACL may lead to profound functional disability. Depending on the source, the patient may experience loss of extension, flexion, patellar entrapment, and/or global loss of motion as measured in all three planes. The loss of extension often has a more deleterious functional effect on the patient than loss of flexion, because it leads to difficulty with simple ambulation and restoration of the normal gait cycle by feedback loop inhibition of the extensor mechanism.9–11 This may also lead to patellofemoral symptoms, referred or secondary joint arthralgias caused by antalgic gait, and loss of athletic performance. Increased forces are experienced in the patellofemoral compartment, leading to early osteoarthritic changes in these young patients. Loss of minor flexion is often asymptomatic and rarely causes significant disability unless total flexion is limited to less than 120 degrees.10
The causative factors for motion loss are often complex and may be intimately interrelated. Possible factors include poor technique causing graft impingement, infection, immobilization, inadequate or excessive rehabilitation, capsulitis-arthrofibrosis, and complex regional pain syndrome.9,10,12 Nonanatomic technique often creates a situation in which the ACL graft is not anatomically positioned in respect to the femoral or tibial insertion sites, or both. This nonanatomic placement may result in a situation in which the graft impinges on the wall of the lateral femoral condyle, roof of the intercondylar notch, or posterior cruciate ligament (PCL), resulting in mechanical loss of motion in flexion or extension and graft attrition, as well as eventual stretching of the graft because of nonanatomic placement of the graft. Loss of flexion may be related to relative anterior placement of the graft on the femoral and tibial sides; conversely, loss of extension may occur with excessive posterior placement of the graft in relation to the tibial and femoral anatomic positions.1,2,13
Another cause of mechanical motion loss includes the phenomenon known as the cyclops lesion, which is rather uncommon. This represents fibroproliferative nodules that may occur on the anterior surface of the ACL graft that limit extension by a mechanical block in the final few degrees.14 These may develop as a result of failure to remove excess debris from the intercondylar notch produced during reaming, or may be a scarring response to repeated impingement of the graft on the roof of the intercondylar notch. Preventive measures against the cyclops lesions include immediate passive and active extension exercises prior to formation of the nodules.
Some patients may experience a condition known as regional capsulitis, or arthrofibrosis, which may also result in loss of motion.1,15 This may be broken down into primary and secondary conditions, with primary being an idiopathic exaggerated healing response to surgical trauma and secondary representing the results of inappropriate surgical timing and/or rehabilitation. In primary or idiopathic arthrofibrosis, the patient presents with excessive postoperative pain, swelling, warmth, resistance to motion in flexion and extension, and severely limited patellar motion in all planes. Treatment generally focuses on restoration of full motion through gentle rehabilitation modalities while, most importantly, reducing the associated pain and inflammation. Early operative intervention should be avoided, because this often accentuates the process. Eventually, this process may result in permanent or fixed global loss of motion caused by caused by intra-articular adhesions.16 If initial conservative measures are inadequate for restoration of motion, surgical manipulation and lysis of these adhesions may be considered once the period of acute inflammation has defervesced and pain controlled. Although this may result in improved motion, these patients rarely achieve objective full motion and only occasionally experience clinical recurrent instability.
Technical Errors
Nonanatomic surgical technique in the primary ACL reconstruction represents the most common cause of failure of these procedures.4 Technical errors include inadequate clearance for the graft in the intercondylar notch, malpositioned nonanatomic tunnels, poor tensioning of the grafts, and inadequate fixation of the graft. Although the concepts of roofplasty and wallplasty are controversial, the surgeon must confirm that the graft is free of bony impingement throughout the full knee range of motion, which may require some degree of bone and soft tissue removal, particularly in the chronic revision ACL-deficient knee.
Placement of tibial and femoral tunnels in nonanatomic positions may result in graft attrition and mechanical entrapment or capture of the knee. Additionally, these errors in tunnel management may result in a graft that allows for persistent or recurrent instability, even in the face of an intact graft. Estimates show that approximately 70% to 80% of failures may be attributed to improper tunnel placement, with nonanatomic femoral locations representing the most commonly encountered error in tunnel placement. Cadaveric and arthroscopic studies have confirmed the topographic bony insertion and origin sites of the ACL and their relationship to the reconstructed ligament. Related studies cast doubt that the common endoscopic transtibial approach to femoral tunnel placement may result in an anatomic location of the femoral insertion of the reconstructed ACL.17,18 This transtibial endoscopic technique most commonly leads to common mistakes in graft placement, including excessive posterior placement on the tibial side combined with a femoral site too high and/or anterior on the medial wall of the lateral femoral condyle. In the best case scenario, there is a mismatch in graft placement within the native insertion site, more closely recreating the posterolateral (PL) site on the tibia to the anteromedial (AM) site on the femur. This combination results in poor tensioning in relation to overall knee kinematics, possible impingement of the graft on the PCL, and a vertical graft (Figs. 23-1 and 23-2).19 This situation leaves the graft in a position that may provide some anteroposterior stability, but sacrifices rotational stability and possibly limits range of motion of the knee. In this case, the knee may actually have been made worse by the reconstruction. Revision surgery is almost inevitably required, depending on the patient’s level of activity.
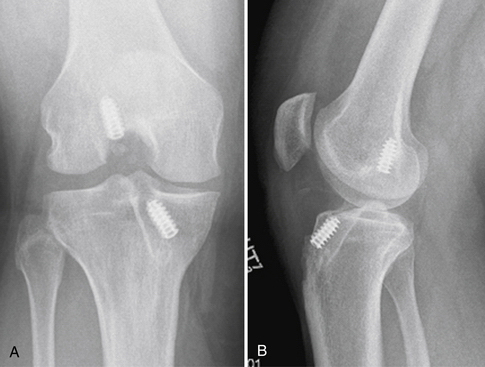
FIGURE 23-1 Failed vertical ACL reconstruction. A, AP view. B, Lateral view.
(From Shaver J, Johnson D. Revision anatomic double-bundle anterior cruciate ligament surgery. Oper Tech Sports Med. 2008;16:157-164.)
FIGURE 23-2 Failed ACL reconstruction with a vertical intact graft but a grossly positive pivot shift on EUA.
The tibial insertion site may be somewhat more forgiving to mistakes in tunnel placement, but significant deviations from anatomic locations may result in less ideal outcomes. Excessive anterior placement results in superior root impingement in full extension and excessive graft forces in flexion, whereas posterior placement results in flexion laxity and possible impingement on the PCL. Additionally, medial or lateral deviations may result in PCL and/or wall impingement and eventual graft attrition and failure.20–23
Failure of Biologic Incorporation
Failure of the graft to incorporate into the host bone may also represent a source of failure of primary ACL reconstruction. This often presents with recurrent instability in the face of an anatomically well-positioned graft. This may be a result of poor fixation on the femoral or, more commonly, the tibial side, excessive forces experienced by the graft during rehabilitation and biologic incorporation, immune responses of the host versus the graft in the case of allografts, or lack of recognition of certain graft-host mismatches. The tibial metaphyseal bone has been identified as the weak link in graft fixation, and therefore additional backup fixation is often recommended, particularly with all soft tissue ACL reconstruction.24 Excessively aggressive rehabilitation protocols may not allow the proper ligamentization of the graft and may eliminate or delay the reparative process that incorporates the graft with the host bone. Each graft substitute has a biologic timetable of incorporation that must be recognized and rehabilitation tailored to the individual situation.25,26 Immune responses have been known to occur between host bone and allografts and/or certain fixation devices, which may result in tunnel lysis and failure of incorporation of the graft. Recognized graft-host mismatches include soft tissue allografts and autografts in young hyperlax females and high-demand athletes, which may stretch out over time and result in recurrent instability.27–29
Concurrent Laxity
Another cause of failure of primary ACL reconstructions is lack of recognition of concurrent injuries to the important secondary stabilizers of the ACL. The native ACL is the primary restraint to anterior displacement of the tibia, but must work in concert with multiple other structures to function optimally.30 This remains true for the reconstructed ACL, and may be even more important because prior to realization of full strength, the graft must undergo incorporation and ligamentization over an extended period of time. If the secondary stabilizers are absent or nonfunctional, nonphysiologic forces are placed on the graft during the ligamentization process, resulting in a nonfunctional lax graft that does not recreate the stability of the native ACL. The secondary stabilizers are numerous, and include the menisci (medial > lateral), medial collateral ligament (MCL), posteromedial corner (including the posterior oblique ligament [POL]), posterior cruciate ligament (PCL), and the posterolateral corner (PLC).31 These structures may be injured in conjunction with the original ACL injury, or may become lax because of repeated episodes of instability in a chronically ACL-deficient knee.32 A reconstruction of the ACL in the face of concurrent laxity places nonphysiologic stresses on the graft, and often results in premature failure of these reconstructions.33,34 Recognition by thorough examination and proper management strategies for these additional sources of laxity must be taken into account in the primary and revision ACL reconstruction situations.
Traumatic Causes
Finally, the well-placed primary graft may simply fail because of traumatic events inherent to the stresses experienced in grafts of many of this active subset of patients. These failures can be caused by excessive strain placed on the immature graft by excessive rehabilitation, premature return to high-level sports or activities, or shear bad luck during high-level sporting activities. These are generally characterized as early failures (less than 1 year), which occur during graft remodeling and incorporation prior to realization of the final graft strength, or late failures (more than 1 year, with no subjective evidence of prior instability).13 Late failures often present in a similar fashion to their initial traumatic event, often with an acute hemarthrosis and recurrent instability. It is important to evaluate the patient critically who has failed primary surgery and has never returned to high-level sports. These patients often have some component of technical errors that precluded their full return after the index procedure.
PATIENT EVALUATION
The most important aspect of preoperative planning for the revision ACL patient is elucidation of the primary cause of the index surgery’s failure, as noted. Another vital component is the patient’s expectations and desires following the revision reconstruction. It is imperative that the surgeon and patient have realistic goals in the revision situation, as studies have shown a significantly lower rate of return to high-level sports.35–37 Some patients may have a degree of injury that makes this goal unrealistic, and the revision may be considered a salvage procedure to limit any further damage.38 The patient and surgeon must also understand and agree that in the revision situation, a significant investment of time and effort is often required for successful outcomes.
Physical Examination
The physical examination in the revision ACL situation must include a global assessment of the limb. This includes an evaluation of overall limb alignment and gait, because significant variations such as a varus thrust may compromise the longevity of an ACL reconstruction.39 Limitations of motion of not only the knee but also the hip and ankle must be taken into account and possibly addressed prior to revision. The condition and any dysfunction of the supporting thigh musculature need to be addressed, because this may be key to rehabilitation after any revision reconstruction. Residual infectious processes must be ruled out in all cases, because this will doom any revision. The locations of previous incisions are taken into account, and future incisions planned to ensure cutaneous tissue viability.
Diagnostic Imaging
Good-quality radiographic evaluation of the knee aids significantly in preoperative planning and should always be performed. These include standing full extension anteroposterior and lateral views of the knee, as well as 45-degree flexion weight-bearing posteroanterior views. Additional examinations that may be of aid include axial views of the patella, full-length mechanical axis views, varus and valgus stress views, and views of the contralateral knee for comparison. These radiographic studies are evaluated for prior tunnel placement, tunnel expansion, location of prior hardware, presence of degenerative changes, and notch geometry.40 These patients often present with MRI scans, which this may be reviewed for concomitant injury to the secondary stabilizers, condition of the menisci and articular cartilage, and placement and condition of the prior graft.









