Chapter 204 Psoriasis
 Diagnostic Summary
Diagnostic Summary
• Circumscribed red, thickened plaques overlying silvery-white scale.
• Characteristically involves the scalp; the extensor surfaces of the wrists, elbows, knees, buttocks, and ankles; and sites of repeated trauma.
• Positive family history in 30% of cases.
• Nail involvement results in characteristic pitting. Other nail changes include onycholysis, discoloration, thickening, and dystrophy.
 General Considerations
General Considerations
Psoriasis is a common skin disorder. Its rate of occurrence in the United States is between 2% and 4% of the population. Psoriasis affects few black people in tropical zones but is more common among those in temperate zones, though still lower than for whites. It appears commonly among Japanese but is rare in American Indians. Psoriasis occurs in men and women equally, and the mean onset is at 33 years of age,1 although 2% of patients show onset by 2 years of age.
Psoriasis is a classic example of a hyperproliferative skin disorder. The rate of keratinocyte proliferation, assessed by proliferating cell nuclear antigen staining, is highest in verrucae (72.1%) and psoriasis (45%), followed by squamous cell carcinoma (28%), actinic keratosis (23%), and basal cell carcinoma (21.4%). The apoptosis inhibitor survivin is a recently described member of the “inhibitor of apoptosis” family expressed in most human cancers and also known to be a regulator of mitosis. Survivin has been detected in 88% of patients with psoriasis. It is likely that other factors are also involved in the development of a hyperplastic epidermis.2
The pathogenesis of psoriasis is incompletely understood; it is a complex, multifactorial disease with a genetic predisposition. The etiopathogenicity of psoriasis can be examined from several different viewpoints. It is a polygenic disorder. Oxidative stress has been linked to the inflammation in psoriasis.3 It is an immune dysfunction. Angiogenesis plays a critical role. Neuropeptides are involved.4 Research into each of these factors reveals increasing levels of sophisticated studies. At this time, it does not seem possible to combine them all under one unifying theory.
HLA-Cw6, which lies in the major histocompatibility region on chromosome 6, is currently considered the major genetic determinant of psoriasis. Variants in or near genes that encode subunits of cytokines (IL12B, IL23A) or cytokine receptors (IL23R) are of interest. The gene product of IL12B, p40, is the target of a recently approved monoclonal antibody therapy for psoriasis (ustekinumab). Association with psoriasis has also been found in genes coding for tumor necrosis factor-alpha (TNF-α) and other cytokines. Many of these genetic variations are also associated with immune disorders considered psoriatic comorbidities, including Crohn’s disease and diabetes.5
Although in the past psoriasis was considered a disorder of keratinocytes, it is now generally accepted that it is primarily an immune-mediated disorder. The cells of the skin consist of cytokine-synthesizing keratinocytes, antigen presenting cells, epidermotrophic and circulating T lymphocytes, dermal capillary endothelial cells, mast cells, tissue macrophages, granulocytes, fibroblasts, and non-Langerhan cells. These cells all communicate by means of cytokine secretion and respond accordingly via stimulation by bacteria, chemical, ultraviolet light, and other irritating factors. The primary cytokine released in response to antigen presentation is TNF-α. Ordinarily this is a controlled process unless the insult to the skin is prolonged, in which case an imbalanced cytokine production in a susceptible host leads to a pathologic state such as psoriasis. Studies have shown that activated keratinocytes in psoriatic lesions not only undergo accelerated turnover but also are a major source of proangiogenic cytokines, such as vascular endothelial growth factor (VEGF), endothelial-cell stimulating angiogenesis factor, TNF-α, and platelet-derived growth factor (PDGF).6 Other angiogenic mediators—such as hypoxia-inducible factors, angiopoietins, interleukin-8 (IL-8), and IL-17—are upregulated in psoriasis.7 Patients with psoriasis receiving anti-VEGF treatment for cancer showed complete remission of their cutaneous symptoms.8
Initially, immature dendritic cells in the epidermis stimulate T cells from lymph nodes in response to antigen stimulation. The lymphocytic infiltrate in psoriasis comprises predominately CD4 and CD8 T cells. Adhesion molecules that promote leukocyte adherence are highly expressed in psoriatic lesions.9 After T cells receive primary stimulation and activation, a resulting synthesis of mRNA for IL-2 occurs, resulting in a subsequent increase in IL-2 receptors. The increased IL-2 from activated T cells and IL-12 from Langerhans cells ultimately regulate genes that code for the transcription of cytokines, such as IFN-gamma, TNF- α, and IL-2, which are responsible for the differentiation, maturation, and proliferation of T cells into memory effector cells. Ultimately, T cells migrate to the skin, where they accumulate around dermal blood vessels, producing proangiogenic cytokines and other angiogenic factors as described above. These are the first in a series of immunologic changes that result in the formation of acute psoriatic lesions.10 The above-described immune response is a somewhat normal response to antigen stimulation. It remains unclear why the T-cell activation that occurs, followed by subsequent migration of leukocytes into the epidermis and dermis, creates accelerated cellular proliferation. One factor that contributes to this is fibronectin (FN), a cell adhesion molecule that anchors cells to collagen or proteoglycan substrates and is involved in many cellular processes, including tissue repair, embryogenesis, blood clotting, and cell migration/adhesion. Transforming growth factor beta (TGF-β) stimulates extra domain A-FN (EDA-FN), a form of FN with an additional domain domain. EDA-FN in turn stimulates keratinocyte hyperproliferation, and through stimulation of Toll-like receptor (TLR) 4, produces proinflammatory cytokines such as TNF-α, IL-1, IL-6, and IL-12. These FN loops contribute to the maintenance and progression of psoriatic lesions. Although the association between psoriasis and cardiovascular disease remains somewhat unclear, one plausible link may be the promotion of atherosclerosis and thrombotic heart disease by EDA-FN.11
It is known that psoriasis develops in bone marrow transplant recipients from donors with psoriasis, clears in recipients from donors without psoriasis, and that immunosuppressive drugs are effective in reducing psoriasis.12,13 Given the genetic predisposition to this disease, what can be done to reduce the phenotypic expression besides resorting to immunosuppressive and biological therapies with their attendant side-effect profiles?
It is interesting that researchers have recognized “unidentified antigens” as ostensibly the source of the psoriatic cascade. A clear relationship of psoriasis with conditions like celiac disease14 and Crohn’s disease15 has been reported. Furthermore, the bowel mucosa of psoriatic patients without bowel symptoms have shown microscopic lesions and greater intestinal permeability, even when the mucosa appeared macroscopically normal.16,17 Given information regarding bowel toxemias, food allergies, low proteolytic and bile enzymes, as well as suboptimal liver function and food allergies (see the following sections), a plausible naturopathic suggestion here would be to consider that factors leading to poor intestinal function most likely encourage greater intestinal permeability and inflammation, which ultimately allow these antigenic and endotoxic compounds to leave the intestinal confines, travel through the bloodstream, and initiate activated immune cascades in susceptible tissues.
 Therapeutic Considerations
Therapeutic Considerations
Gastrointestinal Function
Incomplete Protein Digestion
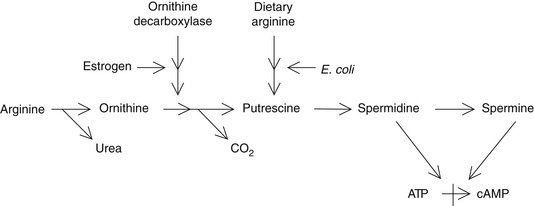
Incomplete protein digestion or poor intestinal absorption of protein breakdown products can result in elevations of amino acids and polypeptides in the bowel. These are metabolized by bowel bacteria into several toxic compounds. The toxic metabolites of the amino acids arginine and ornithine are known as polyamines (e.g., putrescine, spermidine, and cadaverine) and have been shown to be higher in individuals with psoriasis. Polyamines inhibit the formation of cyclic adenosine monophosphate and therefore contribute to the excessive rate of cell proliferation (Figure 204-1).18–20 Lowered skin and urinary levels of polyamines are associated with clinical improvement in psoriasis.18
A number of natural compounds can inhibit the formation of polyamines and may be of benefit in the treatment of psoriasis. For example, vitamin A and the alkaloids of Hydrastis canadensis (goldenseal) such as berberine inhibit bacterial decarboxylase, the enzyme that converts amino acids into polyamines.21,22 However, the best way to prevent the excessive formation of polyamines is to evaluate digestive function with the aid of Heidelberg Gastric Analysis and/or a comprehensive digestive stool analysis and then take the action necessary (e.g., hydrochloric acid supplementation) to ensure complete protein digestion and absorption (these assessment methods can be found in Chapters 18 and 27).
Bowel Toxemia
A number of gut-derived toxins are implicated in the development of psoriasis, including endotoxins (cell-wall components of gram-negative bacteria), streptococcal products, Candida albicans, yeast compounds, and immunoglobulin (Ig) E and IgA immune complexes.23–25 Endotoxins have been found in high levels in the blood of psoriatic patients,26 and these compounds lead to increases in cyclic guanosine monophosphate (GMP) levels within skin cells, thereby increasing the rate of proliferation dramatically. Overgrowth of C. albicans in the intestines (chronic candidiasis) may play a role in some patients with psoriasis.
A diet low in dietary fiber is associated with increased levels of gut-derived toxins.23 Dietary fiber is critical to maintaining a healthy colon. Many fiber components bind bowel toxins and promote their excretion in the feces. It is therefore essential that the diet of an individual with psoriasis be rich in beans, fruits, and vegetables. Natural compounds that bind endotoxins and promote their excretion may also be used. For example, an aqueous extract of the herb Smilax sarsaparilla was found in a 1942 study to be effective in psoriasis, particularly the more chronic, large plaque–forming variety.27 In this controlled study of 92 patients, S. sarsaparilla greatly improved the psoriasis in 62% of the patients and resulted in complete clearance in another 18% (i.e., 80% of the subjects experienced significant benefits). This benefit is apparently due to S. sarsaparilla’s components’ binding to and promoting the excretion of bacterial endotoxins.
Liver Function
The correction of abnormal liver function may be of benefit in the treatment of psoriasis.28 The connection between the liver and psoriasis relates to one of the liver’s basic tasks—filtering and detoxifying the blood returning through the portal circulation from the bowels. Structurally, the hepatic architecture may already be altered in psoriatic patients.29 As mentioned previously, psoriasis has been linked to the presence of several microbial by-products in the blood. If hepatic function is compromised by excessive levels of these toxins from the bowel or if there is a decrease in the liver’s detoxification ability, the systemic toxin level rises and the psoriasis worsens.
Alcohol consumption is known to significantly worsen psoriasis.30 Alcohol has this effect because it both increases the absorption of toxins from the gut (by damaging the gut mucosa) and impairs liver function. Alcohol intake must be restricted in individuals with psoriasis.
Silymarin, the flavonoid component of Silybum marianum, has been reported to be of value in the treatment of psoriasis.28 Presumably this is a result of its ability to improve liver function, inhibit inflammation, and reduce excessive cellular proliferation.31,32
Bile Deficiencies
In the psoriatic patient, endotoxins are able to translocate from the intestine into the bloodstream.26 Bile acids normally present in the intestines act to detoxify bacterial endotoxins. In the absence of sufficient amounts of bile acids, endotoxins translocating into the bloodstream can produce pathologic conditions that vary in severity depending on their amount, including the release of inflammatory cytokines known to play a role in psoriasis.
A fascinating Hungarian study of 800 psoriatic patients was conducted in which 551 were treated with oral bile acid (dehydrocholic acid) supplementation for 1 to 6 weeks or 3 to 8 weeks for acute or chronic cases, respectively. Conventional therapies were administered to 249 patients as a comparison group. Both groups were advised to eat a diet high in vegetables and fruits and were instructed to avoid hot spices, alcohol, raw onion, garlic, and carbonated soft drinks. Of the 551 patients receiving bile acid, 434 (78.8%) became asymptomatic, whereas only 62 (24.9%) of the 249 patients receiving conventional therapies demonstrated clinical recovery during this treatment period. Additionally, the curative effect of bile acid supplementation was more pronounced in the acute form of psoriasis; 95.1% of the patients in this group became asymptomatic. In follow-up assessments 2 years later, 319 of the 551 patients with acute and chronic psoriasis who had been treated with bile acid (57.9%) were asymptomatic, compared with only 15 of the 249 patients (6%) who had received the conventional treatment.26 The bile acid supplements used in the preceding study were either 2 or 3 dehydrocholate sodium (Suprachol) sugar-coated pills once a day or dehydrocholic acid powder (acidum dehydrocholicum pulvis) at two to three doses of 0.25 g/day.
Because there is a theoretical risk of malignant tumors in patients with sluggish intestinal function given long-term bile acid therapy, the investigators in this study recommended that their subjects not continue ingesting bile acids on a regular basis but to supplement with them only after a fatty meal once the initial treatment period had been completed. Two separate evaluations of bile acid effects on the proliferation of colonic mucosa report conflicting results.33,34 Two other studies actually found a cancer-protective effect of ursodeoxycholic acid. One was an observational study of 114 patients with primary biliary cirrhosis in whom a reduction in the risk for colon adenoma was discovered. The prevalence of colorectal adenomas was 13% in the treated group versus 24% in the untreated group. Additionally, the colon epithelial cell proliferation index was significantly lower in treated patients than in untreated patients.35 A second randomized controlled clinical trial of 52 subjects found significant declines in the risk for developing colorectal dysplasia or cancer in patients with ulcerative colitis and primary sclerosing cholangitis.36 More clinical research is needed to examine the different varieties of bile acids and their efficacy as well as their safety profile. Nevertheless, given the preceding information, it seems reasonable to consider the use of bile acids for the short-term treatment of psoriasis and to monitor the colon before and after treatment in patients at high risk for colon cancer.
Nutrition
Omega-3 Fatty Acids
The manipulation of dietary oils is extremely important in the management of psoriasis because serum levels of free fatty acids are typically abnormal in affected patients.37 Of particular benefit are the omega-3 fatty acids. Most of the clinical research has utilized fish oils rich in eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Several double-blind clinical studies have demonstrated that supplementing the diet with 10 to 12 g/day of fish oils (providing 1.8 g EPA and 1.2 g DHA) results in significant improvement.38–40 This amount of EPA and DHA would be equivalent to the amount of EPA in about 150 g of salmon, mackerel, or herring.
The improvement in psoriasis from EPA is partly due to the competition of EPA for arachidonic acid binding sites, producing leukotriene B5 (LTB5), which is only one tenth as potent as the inflammatory mediator LTB4. Levels of LTB4 have been shown to be elevated in psoriatic plaques and to demonstrate chemotactic properties necessary for the infiltration of leukocytes and the proliferation of keratinocytes.41 In the skin of individuals with psoriasis, the production of inflammatory leukotrienes from arachidonic acid is many times greater than normal.42 Leukotrienes are potent inflammatory agents and promoters of guanylate cyclase activity.
In the psoriatic epidermis, the cellular contents of free arachidonic acid and 12-hydroxyeicosatetraenoic acid (12-HETE; a product of lipoxygenase metabolism of arachidonic acid) are 250 and 810 times greater, respectively, than in uninvolved epidermal tissue.42
Immunomodulatory effects of omega-3 fatty acids include the suppression of lymphocyte proliferation, CD4 cells, antigen presentation, adhesion molecule presentation, Th1 and Th2 responses, and proinflammatory cytokine production (such as IL-1, TNF-α, and PDGF), which prevents vascularization within the psoriatic plaque.43
As might be expected, cyclooxygenase inhibitors (e.g., aspirin and most other nonsteroidal antiinflammatory agents) may exacerbate psoriasis, whereas lipoxygenase inhibitors (e.g., benoxaprofen) may bring improvement.44 Naturally occurring substances such as quercetin (the ubiquitous plant flavonoid), vitamin E, onion, and garlic are known to inhibit lipoxygenase and therefore may be of benefit. However, it is improbable that selective inhibition of one component or enzyme (e.g., a 5-lipoxygenase inhibitor) would do more than create an imbalance in this closely integrated network of mediators, which may not necessarily be beneficial.45
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree