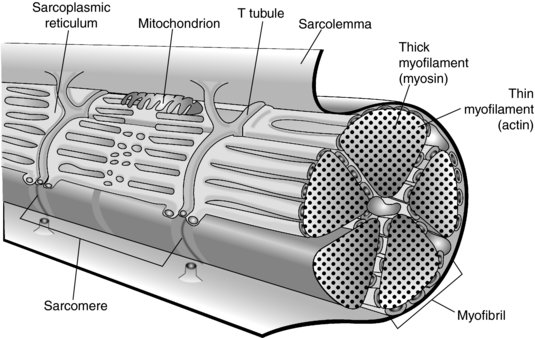
An individual muscle fibre is a long cylindrical cell with multiple nuclei bound by its sarcolemma (Lutz and Lieber 1999). The sarcolemma invaginates around the muscle fibre forming T-tubules. Each muscle fibre contains a large number of myofibrils, which are the contractile elements of the muscle and consist of protein myofilaments. These are actin (thin filament, isotropic, I bands) and myosin (dark filament, anisotropic, A bands) and it is these bands that give muscle its striated appearance. These myofilaments are organised into repeating functional units called sarcomeres. In a resting muscle the actin filaments overlap the myosin to a certain extent. Each myofibril is surrounded by the sarcoplasmic reticulum, which is a store for release of calcium (Kossmann and Huxley 1961; Huxley 1975; Lutz and Lieber 1999) (see Figure 4.2).
Physiology
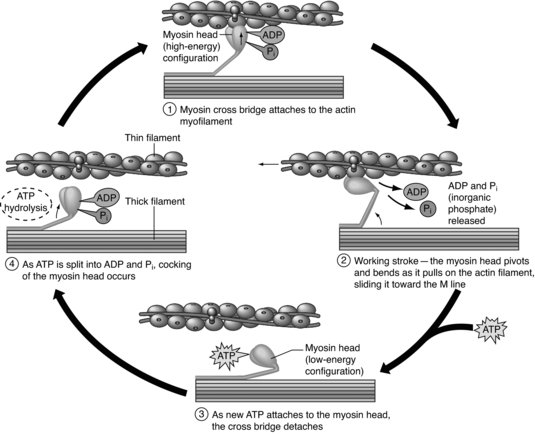
Skeletal muscles are designed to produce voluntary movement by applying forces to bones and joints via a muscle contraction. When a muscle contracts the sarcomeres shorten. This is due to the ‘Sliding Filament Theory’ proposed by Huxley in 1954. The myosin filaments remain static and the actin filaments slide in and out producing force (the A band remains a constant length; the I band becomes shorter). The force is generated by crossbridges forming between the myosin (via the globular myosin heads) and actin binding sites (Kossmann and Huxley 1961; Huxley 1975) (see Figure 4.3).
For this to occur, the muscle fibre is stimulated by a nerve impulse creating an action potential across the sarcolemma. The action potential is propagated down the T-tubules triggering the release of calcium from the sarcoplasmic reticulum. The calcium binds to troponin, a regulatory protein on the surface of actin, exposing the actin binding site. A crossbridge is then formed between actin and myosin, resulting in a contraction cycle powered by ATP. Each muscle fibre is part of a motor unit so a number of muscle fibres are stimulated at the same time (Kossmann and Huxley 1961; Huxley 1975).
Pathophysiology
There are a number of types of muscle injury that can occur: laceration, contusion and strain (Garrett 1996; Huard et al. 2002; Jarvinen et al. 2005). A laceration occurs when the muscle is cut by an external object, this usually occurs during traumatic accidents such as road traffic or industrial accidents. A contusion occurs when there is a compressive force to the muscle and usually occurs in contact sports (Jarvinen et al. 2005), for example in football when two players collide, knee to thigh in a tackle. Strain injuries occur when muscle fibres cannot withstand excessive tensile forces placed on them and are therefore generally associated with eccentric muscle action (Mair et al. 1996; Pull and Ranson, 2007). Strains most commonly occur in muscles working across two joints e.g. hamstrings, gastrocnemius (Jarvinen et al. 2005) during periods of rapid acceleration and deceleration, by placing the muscle in a lengthened state over two joints and contracting forcefully (Stanton 1989; Farber and Buckwalter 2002; Brockett et al. 2004; Hoskins and Pollard 2005; Askling et al. 2006, 2007a, 2007b).
When the muscle is strained the initial injury is usually associated with disruption of the distal myotendinous junction and fibres distal to this but still near the myotendinous junction. Injuries to the muscle belly only occur with the application of very high forces. The contractile elements are the first tissues to be disrupted; with the surrounding connective tissue not being damage until high forces are applied (Hasselman, et al. 1995). The contractile elements are relative stiff in comparison to the surrounding connective tissue and hence become disrupted at lower forces than the surrounding connective tissue.
Muscles heal by a repair process that can be divided into two phases: (1) the destruction/injury phase resulting in rupture and necrosis of the muscle fibres; and (2) repair and regeneration.
Destruction/injury phase
This phase results in damage to the vascular supply and as oxygen can no longer reach the cells, they die and release lysosomes (Schiaffino and Partridge 2008). Excessive force to a muscle fibre results in tearing of the sarcoplasm and the cells respond by forming a contraction band (condensation of cytoskeletal material) creating a protective barrier. The lysosomes are pivotal in this vital process keeping the necrosis to a local area and preventing it from spreading along the whole length of the cell (Jarvinen et al. 2005).
Within 15 minutes of injury the damaged tissue consists of disrupted extracellular tissue and dead cells, platelets and plasma, which themselves release powerful enzymes such as thrombin thereby setting off an inflammatory cascade (Schiaffino and Partridge 2008). A haematoma is formed to fill the gap between ruptured muscle fibres.
Clinically tissue inflammation presents as redness, heat, swelling and pain of the tissues. The redness, heat and swelling are due to an increased blood flow and so blood within vascular beds in the area, with the swelling developing as a result of the increased local tissue pressure due to inflammatory exudates (local capillaries become more permeable) leaking into the interstitial space (Schiaffino and Partridge 2008). The pain is due to the initial damage to local nerves and irritation of nerves in the area from the inflammatory chemicals release by the damaged tissue (Evans 1980).
Repair and regeneration
After the initial injury, the inflammatory chemicals released from the injured tissues attract lymphocytes and macrophages to the area (Jarvinen et al. 2005). Macrophage phagocytosis of the necrotic material then occurs removing the debris (Tidball and Wehling-Henricks 2007). The regeneration process starts within 3–6 days following injury, reaching a peak between day 7 and 14. The regenerative capacity of skeletal muscle is provided by satellite cells, specialised cells underneath the basal lamina of each muscle fibre. In response to injury they proliferate and differentiate into myoblasts and then become multinucleated myotubes (Jarvinen et al. 2005). These then fuse with parts of the muscle fibre that have survived the initial injury and attempt to breech the gap in the muscle (Lieber and Friden 2002). Recent findings have shown that bone marrow stem cells may play a role in contributing to regenerating muscle fibres and replenishing the satellite cells, however it is debatable as to how big a role this is (Gates and Huard 2005; Jarvinen et al. 2005).
At the same time as myotubes are attempting to cross the injury site, fibrin is being laid down creating an irregular meshwork of short fibres by the fibroblasts, which have differentiated from the macrophages drawn to the area earlier in the process, this acts as a connective tissue scaffold (Schiaffino and Partridge 2008). This irregular fibrin network obviously reduces the ability of the myotubes to cross the rupture and make good an effective contractile unit. It is possible to minimise the irregular nature of this connective tissue mesh and also the amount present by applying suitable tensile loads (i.e. not high enough to cause re-rupture) (Lehto et al. 1985).
This regeneration process can occur rapidly, within 10 days post injury the injured muscle can regain much of its contractile ability with progressively applied loading, set at levels below the pain threshold (Nikolaou et al. 1987).
Treatment of muscle injuries
It is frequently cited within the sports medicine literature that the initial treatment of musculoskeletal injuries should be rest, ice, compression and elevation (RICE). However, this acrimony is likely to be more valid in the less metabolically active tissues such as ligament and bone (Gates and Huard 2005). Muscle with its capacity for rapid regeneration due to the nature of its constituent tissues requires a modified approach (Peterson and Holmich 2005; Thorsson et al. 2007). This modification is based around the balance between absolute rest and the absolute level of loading to stimulate appropriately the rapidly developing tissues.
Rest and mobilisation
Early mobilisation, rather than immobilisation or complete rest, has been advocated (Thorsson et al. 2007). Studies have shown early mobilisation aids with regeneration of muscle fibres (stimulation of satellite cells and myotube formation improves capillary growth into the area and aids with more parallel orientation of collagen and muscle fibres (Nikolaou et al. 1987, Taylor et al. 1993; Goldspink 1999). The exact level of loading is difficult to judge, it must be sufficient to stimulate and challenge the developing tissues, but not so great that it causes tissue breakdown (Kannus et al. 2002). This includes early weight bearing to help promote scar tissue re-alignment (Croisier 2004; Jarvinen et al. 2005) and controlled running to reduce muscle inhibition (Herrington, 2000)
Immobilisation and poorly controlled early loading (low or high), have been shown to lead to the development of contracted scar tissue, which blocks the linking of myotubes across the injury, thereby stopping the formation of a functional contractile element, and the surrounding areas then become more susceptible to further injury (Beiner and Jokl 2001; Lehto et al. 1985).
Ice
Studies have shown that ice results in a significantly smaller haematoma and less inflammation in the initial stages of the injury (Jarvinen et al. 2005). Reducing tissue temperature results in vasoconstriction, thereby limiting the amount of bleeding in the area (Beiner and Jokl 20012). It also reduces the metabolic rate of the tissue and therefore reduces the demand for oxygen (Hubbard et al. 2004) decreasing the hypoxic damage.
Clinical studies have shown the optimal duration to apply ice is for 5–10 minutes in the initial stages and repeat every 60 minutes (Croisier 2004; Hubbard et al. 2004) within the first 24–48hours to reduce the inflammatory effects.
Compression and elevation
Compression is an area where research is lacking, it has been stated that it results in a reduction in the severity of bleeding and swelling following an injury (Kannus et al. 2002; Jarvinen et al. 2005), though the only evidence often present in support of this theory is from studies involving ligament injuries. A recent clinical study by Thorsson et al. (2007) utilising 40 athletes with calf injuries found compression resulted in no significant difference in reducing muscle haematoma, or speed of recovery of the injury using compression
Elevation is still one of the preferred and easiest methods of immediate management used in sports medicine for muscular injuries (Hergenroeder 1998). It simply relies on the use of gravity to promote venous return and lymphatic flow to drive swelling/oedema from the area (Hergenroeder 1998). Again research is lacking in this area, with most authors when citing research, using studies on ankle ligament injuries and even here the actual positive benefits when the limb is dependent again are very limited (Tsang et al. 2003). It would appear that the greatest benefit that comes from compression and elevation is that it ensures that the athlete rests during the acute inflammatory phase of the first 72 hours following injury.
Strengthening exercise
Isometric exercise can begin after 2–5 days and should be performed within the limits of pain (Jarvinen et al. 2005). Frequency, duration and intensity are limited by the patients’ pain. Some therapists advocate three sets of 10 repetitions using 5–10 second holds to begin with at intensity within pain tolerance (Pull and Ranson 2007). These then are undertaken at multiple angles, beginning in mid range then progressing to inner range (shortened position) then outer range (lengthened position). Once these can be undertaken in a pain-free manner throughout the available range, then isotonic exercises can commence. Dynamic movement and isotonic contraction is then incorporated again starting in the strongest position (mid range, close to a 90° joint angle) progressing to and finishing in the functionally most relevant (outer range with eccentric and concentric contractions most often).
There is preferential atrophy of type 1 muscle fibres with disuse (Stockmar et al. 2006), and high loads and rates of force development are most likely to over-stress the healing tissue. Therefore, initially an endurance based programme should be used, (three sets of 15 repetitions at 40–60% of one repetition maximum) this would be progressed to strength (4–6 sets of 3–6 repetitions at 85–95% of one repetition maximum and then power training (3–5 sets of 3-5 repetitions at 75–85% of one repetition maximum) (Kraemer et al. 2002), or plyometrics depending on the specific requirements of the muscle. The exact nature of the progressions, directions and velocity of movement will depend on which muscle has been injured and the requirements of the sport, as will whether or not the progressions have a major stability/proprioception element.
Within the body certain muscles can be regarded as having a role that is predominately about the generation of force/power and they tend to work mostly in a single plane (usually the sagittal) these are often called mobiliser muscles. Other muscles can be regarded as having a stability role, they control motion of the body and often have the ability to contract in multiple planes; they are often called stabiliser muscles (see Table 4.1).
Table 4.1 Examples of different muscle’s roles
| Mobiliser muscles | Stabiliser muscles |
| Hamstrings | Gluteus medius/minimus |
| Quadriceps | Adductor longus |
| Gastrocnemius | Tibialis posterior |
| Pectorialis major | Infraspinatus |
| Latissimus dorsi | Subscapularis |
The nature of the role of the muscles affects the choice of rehabilitation exercise we will choose towards the end stage of rehabilitation. Power/mobiliser muscles do not have a stability role and so will not require exercise progressions that involve a proprioceptive challenge, whereas, the stabiliser muscles will. Similarly, those muscles that predominately work in an open kinetic chain manner will not require exercise progressions involving closed kinetic chain exercises.
Stretching
Passive stretching (at the end of available range) should be avoided for the first 72 hours as a minimal period, possibly the athlete should not stretch for the first 7–10 days following injury (Neidlinger-Wilke et al. 2002). The reasons for this are twofold. Firstly, the healing tissue is weak and intolerant of tensile loading and so is likely to be damaged by uncontrolled stretching. The second reason is there is no physiological need in the early stages to stretch, as the scar does not beginning to shrink until around the tenth day post injury when fibroblasts begin to be converted to myofibroblasts and contract and draw the wound ends together. Prior to the tenth day post injury it would be more appropriate to take the muscle through its full available pain-free range without any attempt to force the muscle beyond this point.
Once it is appropriate to begin stretching the muscle; that is, elongating the tissue beyond its available range, then careful passive stretching can be performed. Each stretch should be held at the end of available range within the limits of pain. Time, frequency, duration and intensity of stretch remain debateable in the literature. Some research suggests passive stretching should be held for a minimum of 15 seconds with 6–8 sets per day (Roberts and Wilson 1999), however, Bandy et al. (1997) reviewed stretches at 15 seconds, 30 seconds and 60 seconds. They determined that 30 seconds was the optimum duration, also stating that longer periods after 30 seconds was ineffective in promoting additional stretch.
As with strengthening exercises stretching exercises need to be progressed in order that the tissue adapts to the different types of load once the athlete is comfortable (pain free) with passive stretching (Bandy et al. 1997).
Electrotherapy
Pulsed shortwave diathermy is particularly helpful for enabling re-absorption of the muscular haematoma as it is particularly effective in more vascularised tissues such as skeletal muscle (Robertson and Baker 2001). It is thought to work at the cell membrane level, resulting in an ‘up regulation’ of cellular behaviour. This results in an improved rate of oedema dispersion, resolution of the inflammatory process and promotes a more rapid rate of fibrin fibre orientation and deposition of collagen (Robertson and Baker 2001).
Another modality that can be used to help speed up a recovery muscle injury is therapeutic ultrasound. This is the use of high intensity sounds waves, which research has been shown can help recovery of tissues at a cellular level by increasing ion transport across cells and increasing metabolism within the cell (Wilkin et al. 2003) and increasing fibroblastic and angiogenic activity (ter-Haar et al. 1978). However, evidence-based research remains limited in the effectiveness and reliability of therapeutic ultrasound treatment with the majority of supporting evidence coming from in-vitro cell culture studies (Robertson 2002) even though many clinicians still use it as their main electrotherapy treatment.
Muscle stimulation may prove a further useful electrotherapeutic adjunct for the treatment of muscle injuries. It has been shown to decrease oedema, muscle inhibition and the rate of strength loss with inactivity (Thornton et al. 1998). However, muscle stimulation has not been shown to be useful in the regaining of strength in the injured athlete (Snyder-Mackler et al. 1995).
Other factors
As discussed earlier, management of muscle injuries includes prevention of re-injury. For example, with hamstring injuries, many factors have been cited in literature as potential causes of re-injury. These include: previous injury (Verall et al. 2001; al. Crossier et al. 2002, 2003; Arnason et al. 2004; Foreman et al. 2006); lack of flexibility (Knapik et al. 1991; Hennessey and Watson 1993; Jonhagen et al. 1994; Bennell et al. 1998; Cross and Worrell 1999; Witvrouw et al. 2000; Funk et al. 2001; Brockett et al. 2004; Foreman et al. 2006); inadequate warm up (Worrell 1994; Worrell et al. 1994); fatigue (Worrell 1994; Worrell et al. 1994); muscle strength imbalance (Knapik et al. 1991; Yamamoto, 1993; Cameron et al. 2003; Crossier et al. 2003; Foreman et al. 2006); and poor coordination (Cameron et al. 2003; Brockett et al. 2004; Foreman et al. 2006). Hamstring strains have also been associated with eccentric loading (Kujala et al. 1997; Cameron et al. 2003; Brockett et al. 2004), such as during rapid deceleration.
An understanding of the mechanics of the sport can be helpful in analysing the cause of the injury in the first place. This information can normally be revealed during the initial subjective assessment of the athlete, using simple questioning such as ‘how did the injury happen, can you demonstrate (without re-injuring of course)’.
Example of muscle injury (hamstring)
Hamstring strains are one of the most common injuries in sport and can result in a lengthy period out of the game if not treated effectively (Clark et al. 2005).
The hamstring is a two-joint muscle and is most susceptible to injury in sports involving sprinting and kicking (Stanton 1989; Brockett et al. 2004; Hoskins and Pollard 2005; Askling et al. 2006). The majority of injuries occur in the biceps femoris and at the musculotendinous junction, although they can also occur to the semimembranosus during stretching (Askling et al. 2007b).
Mechanism of injury
As stated earlier, injury often occurs during sprinting and the point of failure has been shown to occur in the terminal swing phase just prior to foot strike. This is when the hamstrings have to work eccentrically to decelerate the tibia and control knee extension (Clark et al. 2005). Hamstring strains are commonly reported in sprinters when speed is maximal or close to maximal (Askling et al. 2006) and during powerful eccentric muscle actions (Brockett et al. 2004). The strain is most likely to occur during two phases of the running cycle; late forward swing and toe off (Stanton 1989) as during this phase the hamstrings decelerate hip flexion and knee extension (Hoskins and Pollard 2005) resulting in large eccentric loads. It has also been found that whilst sprinters sustain their injuries during high-speed running, dancers sustain injuries whilst performing slow stretching type exercises (Askling et al. 2006). In activities such as dancing most hamstring injuries occur during stretching (hip flexion with knee extension) (Askling et al. 2006, 2007a, 2007b), resulting in an eccentric load, with the proximal end of the semimembranosus as the site of injury (Askling et al. 2007b).
Predisposing factors
The predisposing factors of hamstring strain injury are multifactorial, including poor lumbar posture (Hennessey and Watson 1993), previous injury (Crossier et al. 2003; Arnason et al. 2004; Foreman et al. 2006), lack of flexibility (Knapik et al. 1991; Hennessey and Watson 1993; Bennell et al. 1998; Kaminski et al. 1998; Cross and Worrell 1999; Funk et al. 2001; Brockett et al. 2004; Foreman et al. 2006), inadequate warm up (Worrell 1994; Worrell and Smith 1994), fatigue (Worrell 1994; Worrell and Smith 1994), strength imbalance and inadequate quadriceps to hamstring ratio (Knapik et al. 1991; Cameron et al. 2003; Crossier et al. 2003; Foreman et al. 2006), and poor coordination (Cameron et al. 2003; Brockett et al. 2004; Foreman et al. 2006).
The length of the muscle when peak torque is produced has also been postulated as a predisposing factor, with the hypothesis that the greater the knee extension angle at which peak torque is produced the lower the risk of injury (Clark et al. 2005).
The literature suggests that there are two types of hamstring strain; one resulting from high-speed running, as in football (Woods et al. 2004) and athletics (Yamamoto 1993), and the other during stretching movements carried out at extreme range of motion (ROM) (Askling et al. 2002). Reported causes of hamstring strains include poor lumbar posture (Hennessey and Watson 1993), previous injury (Verall et al. 2001; Crossier et al. 2002; Arnason et al. 2004; Foreman et al. 2006), lack of flexibility (Hennessey and Watson 1993; Funk et al. 2001; Brockett et al. 2004; Jonhagen et al. 1994; Cross et al. 1999; Witvrouw et al. 2000; Bennell et al. 1998; Foreman et al. 2006; Knapik et al. 1991), inadequate warm up (Worrell, 1994; Worrell et al. 1994), fatigue (Worrell, 1994; Worrell et al. 1994), strength imbalance and inadequate Quadriceps to Hamstring ratio (Knapik et al. 1991; Crossier et al. 2003; Yamamoto, 1993; Cameron et al. 2003; Foreman et al. 2006), and poor coordination (Cameron et al. 2003; Brockett et al. 2004; Foreman et al. 2006). Hamstring strains have also been associated with eccentric loading (Kujala et al. 1997; Cameron et al. 2003; Brockett et al. 2004), such as during rapid deceleration.
Symptoms
Hamstring strains can often be diagnosed by the mechanism of injury resulting in sudden onset of pain. The patient presents with reduced hamstrings contraction against resistance and reduced stretch. Local bruising/haematoma is often present with pain on palpation. They may also find it difficult to walk and are unable to run or sprint (Croisier et al. 2002).
Treatment
Acute management is as stated previously in that relative rest, ice, compression and elevation are indicated to reduce inflammation and provide optimal environment for repair (Thorsson et al. 2007).
Gentle stretches and early weight bearing can also be commenced within the pain-free range to assist with correct fibre orientation (Bandy et al. 1994, 1997, 1998; Goldspink 1999; Sherry and Best 2004; Peterson and Holmich 2005). Localised soft tissue techniques, including cryotherapy in the acute phase (Herrington 2000; Hubbard et al. 2004) and electrotherapy once the acute phase has settled, aid the reduction of any muscle spasm and helps soft tissue repair (Herrington 2000; Robertson and Baker 2001). In particular, ultrasound may be used to assist in the breakdown of scar tissue and promotion of tissue healing, and is interferential in the reduction of swelling and inflammation (Wilkin et al. 2004).
Strengthening exercises are vital to try to prevent further injury. Initially starting with isometric and progressing onto isotonic as pain allows (Jarvinen et al. 2005). As injury is most likely to occur during eccentric activity, it is vital that eccentric exercise is incorporated into the programme in the later stages. The Nordic eccentric exercise has been shown to improve the torque angle at the knee, and rugby union teams that incorporated it into their training programmes found a reduced number of hamstring strains (Kujala et al. 1997; Clark et al. 2005; Brooks et al. 2006; Gabbe et al. 2006; Arnason et al. 2008). Eccentric training programmes should be closely monitored as they can lead to delayed onset muscle soreness; a low volume (3–5 sets of three repetitions) high frequency (3–4 times a week) may be most appropriate.
Exercises are then progressed to functional activities (e.g. running/sprint training), a return to sport once there is full strength and pain-free movement, and to completion of progressive running programmes and functional tests (Herrington 2000). As discussed earlier, sports specific rehabilitation is vital to return the athlete back to their functional sport. Not only can this return the athlete back to sport quicker, but help prevent further injury later on.
Functional fitness must also be maintained throughout the rehabilitation process without aggravating the injury. Examples could include cycling, walking, upper body weights and swimming, so long as these are pain free (Croisier et al. 2002). This would not be functional for most sports, but may be useful in reducing the detraining effect regularly associated with a reduction in training volume and intensity following injury.
Prevention
As stated in the example given there are a number of factors that can predispose to skeletal muscle injury and if these can be controlled/prevented then the risk of injury will be reduced.
Examples may include adequate warm up and stretches prior to sports participation; a conditioning programme consisting of eccentric, plyometric, sports specific and cardiovascular exercise and optimum treatment following previous injury (Kujala et al. 1997; Herrington, 2000; Clark et al. 2005; Brooks et al. 2006; Gabbe et al. 2006; Arnason et al. 2008). As an example, a hamstring in a football can be caused by a running/sprint deceleration activity which incorporates eccentric control of the hamstring muscle at higher speeds. This eccentric control of muscle must be re-implemented within the rehabilitation programme once the initial injury is managed in order to rehabilitate the fibre re-orientation and to help reorganise the neuromuscular pathways which control the activity at such speed (Marqueste et al. 2004). Therefore, as a simple example sports specific rehabilitation may involve sprinting over short distances with sudden stopping and re-sprinting in another direction over many repetitions in order to mimic the control required for football. To begin with, this may require slower timed sessions to begin with, and as time progresses, this process is speeded up to match speed. This not only helps recruit the right muscle fibre type, but also helps restore proprioception and regain functional fitness.
Fatigue has also been shown to be a precursor to injury as a fatigued muscle is not able to absorb as much energy prior to failure due to a reduced ability to generate force (Mair et al. 1996). Therefore, rehabilitation should also be geared towards increasing the duration of rehabilitation sessions and trying to maintain fitness even with injury. Using the example above, the footballer with the hamstring strain can still cycle, swim, aqua-jog to help maintain a certain level of fitness without re-injuring the hamstring during treatment. Similarly, once the injury is repaired, emphasis should be placed on increasing duration of sessions, and hence helping to ensure endurance is not compromised.
It can be seen that rehabilitation therapists have a vital role to play in preventing and treating injuries to skeletal muscle.
Summary key points of muscle healing and rehabilitation
- RICE should be implement as soon as possible following acute injury
- Early mobilisation and weight bearing should also be encouraged
- Stretching and strength exercises can start within pain-free range as soon as possible
- Fitness and conditioning of the athlete should be incorporated within the early rehabilitation programme without compromising the injury
- Specificity, and functional fitness are imperative to help return the athlete back to sport without recurrence.
References
Arnason, A., Sigurdsson, S.B., Gudmundsson, A., Holme, I., Engebretsen, L. and Bahr, R. (2004) Risk factors for injuries in football. American Journal Sports Medicine, 32 (1), S4–16.
Arnason, A., Anderson, T.E., Holme, I., Engebretsen, L. and Bahr, R. (2008) Prevention of hamstring strains in elite soccer: an intervention study. Scandinavian Journal of Medincine and Science in Sports, 18 (1), 40–48.
Askling, C. M., Lund, H., and Saartok, T. and Thorstensson, A. (2002) Self reported hamstring injuries in student dancers. Scandinavian Journal of Medincine and Science in Sports, 12 (4), 230–235.
Askling, C. M., Saartok, T. and Thorstensson, A. (2006) Type of acute hamstring strain affects flexibility, strength, and time to return to pre-injury level. British Journal of Sports Medicine, 40 (1), 40–44.
Askling, C.M., Tengvar, M., Saartok, T. and Thorstensson, A. (2007a) Acute first-time hamstring strains during high-speed running: a longitudinal study including clinical and magnetic resonance imaging findings. American Journal of Sports Medicine, 35 (2), 197–206.
Askling, C.M, Tengvar, M., Saartok, T. and Thorstensson, A. (2007b) Acute first-time hamstring strainsduring slow-speed stretching: clinical, magnetic resonance imaging, and recovery characteristics. American Journal of Sports Medicine, 35 (10), 1716–24.
Bandy, W.D. and Irion, J.M. (1994) The effect of time on static stretch on the flexibility of the hamstring muscles. Physical Therapy, 74 (9), 845–852.
Bandy, W.D., Irion, J.M. and Briggler, M. (1997) The effect of time and frequency of static stretching on the flexibility of the hamstring muscles. Physical Therapy, 77 (10), 1090–1096.
Bandy, W.D., Irion, J.M. and Briggler, M. (1998) The effect of static stretch and dynamic range of motion training on the flexibility of the hamstring muscles. Journalof Orthopaedic Sports and Physical Therapy, 27 (4), 295–300.
Beiner, J. M. and Jokl, P. (2001) Muscle contusion injuries: Current treatment options. Journal of American Academy of Orthopaedic Surgery, 9 (4), 227–237.
Bennell, K., Wajswelner, H., Lew, P., Schall-Riaucour, A., Leslie, S., Plant, D. and Cirone, J. (1998) Isokintic strength does not predict hamstring injury in Australian Rules footballers. British Journal of Sports Medicine, 32 (4), 309–314.
Brockett, C.L., Morgan, D.L. and Proske, U. (2004) Predicting hamstring strain injury in elite athletes. Medicine and Science in Sports and Exercise, 36 (3), 379–387.
Brooks, J.H.M., Fuller C.W., Kemp S.P.T. and Reddin, D.B. (2006) Incidence, risk and prevention of hamstring muscle injuries in professional Rugby Union. American Journal of Sports Medicine, 34, 1297–1306.
Cameron, M., Adams, R. and Maher, C. (2003) Motor control and strength as predictors of hamstring injury in elite players of Australian football. Physical Therapy in Sport, 4, 159–166.
Clark, R., Bryant, A., Culgan, J.P., and Hartley, B. (2005) The effects of eccentric hamstring strength training on dynamic jumping performance and isokinetic strength parameters: a pilot study on the implications for the prevention of hamstring injuries. Physical Therapy in Sport, 6, 67–73.
Croisier, J.L. (2004) Factors associated with recurrent hamstring injuries. Sports Medicine, 34 (10), 681–695.
Croisier J.L., Forthomme, B., Namurois, M-H., Vanderthommen, M. and Crielaard J.M. (2002) Hamstring muscle strain recurrence and strength performance disorders. American Journal of Sports Medicine, 30, 199–203.
Croisier, J.L., Forthomme, B., Namurois, M.H., Vanderthommen, M., and Crielaard, J.M. (2003) Hamstring muscle strain recurrence and strength performance disorders. American Journal of Sports Medicine, 30 (2), 199–203.
Cross, K.M. and Worrell, T.W. (1999) Effects of a static stretching program on the incidence of lower extremity musculotendinous strains. Journal of Athletic Training, 34 (1), 11–14.
Dadebo, B., White, J. and George, K.P. (2004) A survey of flexibility training protocols and hamstring strains in professional football clubs in England. British Journal of Sports Medicine, 38 (4), 388–394.
Evans, P. (1980) The Healing Process at Cellular Level: A review. Physiotherapy, 66 (8), 256–259.
Farber, J., and Buckwalter, K. (2002) Magnetic resonance imaging in non-neoplastic muscle disorders of the lower extremity. Radiologic Clinics of North America, 40 (5), 1013–1031.
Foreman, T.K., Addy, T., Baker, S., Burns, J., Hill, N. and Madden, T. (2006) Prospective studies into the causation of hamstring injuries in sport: A systematic review. Physical Therapy in Sport, 7, 101–109.
Funk, D., Swank, A.M., Adams, K.J. and Treolo, D. (2001) Efficacy of moist heat pack application over static stretching on hamstring flexibility. Journal of Strength and Conditioning Research, 15 (1), 123–126.
Gabbe, B.J., Branson, R. and Bennell, K.L. (2006) A pilot randomised controlled trial of eccentric exercise to prevent hamstring injuries in community-level Australian Football. Journal of Science and Medicine in Sport, 9 (1–2), 103–109.
Gabbett, T.J. and Domrow, N. (2005) Risk factors for injury in subelite rugby league players. American Journal of Sports Medicine, 33 (3), 428–434.
Garrett, W.E. (1996) Muscle strain injuries. American Journal of Sports Medicine, 24 (6), S2–8.
Gates, C. and Huard, D. (2005) Management of skeletal muscle injuries in military personnel. Operative Techniques in Sports Medicine, 13, 247–256.
Gibbs, N. (1994) Common rugby league injuries. Recommendations for treatment and preventative measures. Sports Medicine, 18 (6), 438–450.
Goldspink, G. (1999) Changes in muscle mass and phenotype and the expression of autocrine and systemic growth factors by muscle response to stretch and overload. Journal of Anatomy, 194 (3), 323–334.
Hasselman, C.T., Best, T.M., Seaber, A.V., Garrett, W.E. (1995) A threshold and continuum of injury during active stretch of rabbit skeletal muscle. American Journal of Sports Medicine, 23, 65–73.
Hawkins, R.D., Hulse, M.A., Wilkinson, C., Hodson, A. and Gibson, M. (2001) The association football medical research programme: an audit of injuries in professional football. British Journal of Sports Medicine, 35 (1), 43–47.
Hennessey, L. and Watson, A.W.S. (1993) Flexibility and posture assessment in relation to hamstring injury. British Journal of Sports Medicine, 27 (4), 243–246.
Hergenroeder, A. (1998) Prevention of sports Injuries. American Journal of Paediatrics, 101 (6), 1057–1063.
Herrington, L. (2000) Patients with hamstring muscle strains returning to sport in less than fourteen days, a report of the treatment used. Physical Therapy in Sport, 1 (4), 137–138.
Hoskins, W. and Pollard, H. (2005) The management of hamstring injury – Part 1: Issues in diagnosis. Manual Therapy, 10 (2), 96–107.
Hoskins, W., Pollard, H., Hough, K., and Tully, C. (2006) Injury in rugby league. Journal of Science and Medicine in Sport, 9, 46–56.
Huard, J., Li, Y., Fu, F.H. (2002) Muscle injuries and repair: Current trends in research. Journal of Bone and Joint Surgery, 84, 822–832.
Hubbard. T.J., Aronson, S.L. and Denegar, C.R. (2004) Does cryotherapy hasten the return to participation? A systematic review. Journal of Athletic Training 39 (1), 88–94.
Huxley, A.F. (1975) The origin of force in skeletal muscle. Ciba Foundation Symposium, 31, 271–90.
Jarvinen, T.A., Jarvinen, T.L., Kaariainen, M., Kalimo, H., and Jarvinen, M. Muscle injuries: Biology and treatment. American Journal of Sports Medicine, 33( 5), 745–764.
Jonhagen, S., Nemeth, G. and Eriksson, E. (1994) Hamstring injuries in sprinters. The role of concentric and eccentric hamstring muscle strength and flexibility. American Journal of Sports Medicine, 22 (2), 262–266.
Kaminski, T.W., Webberson, C.V. and Murphy, R.M. (1998) Concentric versus enhanced eccentric hamstring strength training: Clinical implications. Journal of Athletic Training, 33 (3), 216–221.
Kannus, P., Parkkari, J., Järvinen, T.A. and Järvinen, A.H. (2002) Basic science and clinical studies coincide: active treatment approach is needed after a sports injury. Scandinavian Journal of Medicine and Science in Sports. 13 (3), 150–154.
Kossmann, C.E. and Huxley, H.E. (1961) The contactile structure of cardiac and skeletal muscle. Circulation, 24, 328–335.
Knapik, J.J., Bauman, C.L., Jones, B.H., Harris, J.M. and Vaughan, L. (1991) Preseason strength and flexibility imbalances associated with athletic injuries in female collegiate athletes. American Journal of Sports Medicine, 19 (1), 76–81.
Kraemer, W.J., Adams, K., Cafarelli, E., Dudley, G.A., Dooly, C., Feigenbaum, M.S., Fleck, S.J., Franklin, B., Fry, A.C., Hoffman, J.R., Newton, R.U., Potteiger, J., Stone, M.H., Ratamess, N.A. and Triplett-McBride, T. (2002) American College of Sports Medicine, Position Stand. Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 34 (2), 364–380.
Kujala, U.M., Orava, S. and Jarvinen, M. (1997) Hamstring injuries: current trends in treatment and prevention. Sports Medicine, 23 (6), 397–404.
Lehto, M., Duance, V.C., and Restall, D. (1985) Collagen and fibronectin in a healing skeletal muscle injury: an immunohistological study of the effects of physical activity on the repair of injured gastrocnemius muscles in rats. Journal of Bone and Joint Surgery, 67B (5), 820–828.
Lieber, R.L. and Friden, J. (2002) Mechanisms of muscle injury gleaned from animal models. American Journal of Physical Medical Rehabilitation, 81, S70–79.
Lutz, G.J. and Lieber, R.L. (1999) Skeletal muscle myosin II structure and function. Exercise and Sports Science Reviews, 27, 63–77.
Mair, S.D., Seaber, A.V., Glisson, R.R. and Garrett, W.E. (1996) The role of fatigue in susceptibility to acute muscle strain injury. American Journal of Sports Medicine, 24 (2), 137–143.
Marqueste, T., Alliez, J., Alluin, O., Jammes, Y. and Decherchi, P. (2004) Neuoromuscular rehabilitation by treadmill running on electrical stimulation after peripheral nerve injury and repair. Journal of applied Physiology, 96, 1988–1995.
Neidlinger-Wilke, C., Grood, E., Claes, L. and Brand, R. (2002) Fibroblast orientation to stretch begins within three hours. Journal Orthopaedic Research, 20, 953–956.
Nikolaou, P.K., Macdonald, B.L., Glisson, R.R., Seaber, A.V. and Garrett, W.E. (1987) Biomechanical and histological evaluation of muscle after controlled strain injury. American Journal of Sports Medicine, 15, 9–14.
Orchard, J. (2004) Missed time through injury and injury management at an NRL club. Sport Health, 22 (1) 11–19.
Peterson, J. and Holmich, P. (2005) Evidence based research of hamstring injuries in sport. British Journal of Sports Medicine, 29 (6), 319–323.
Pull, M. R., and Ranson, C. (2007) Eccentric muscle actions: Implications for injury prevention and rehabilitation. Physical Therapy in Sport, 8, 88–97.
Roberts, J. and Wilson, K. (1999). Effect of stretching duration on active and passive range of motion in the lower extremity. British Journal of Sports Medicine. 33 (4), 259–263.
Robertson, V. (2002) Dosage and treatment response in a randomised control trial of therapeutic ultrasound. Physical Therapy in Sport, 3 (3), 124–133.
Robertson, V.J. and Baker, K.G. (2001) A review of therapeutic ultrasound: effectiveness studies. J Physical Therapy, 81 (7), 1339–50.
Schiaffino, S. and Partridge, T. (2008) Skeletal Muscle Repair and Regeneration. London: Springer.
Seward, H., Orchard, J., Hazard, H., et al. (1993) Football injuries in Australia at the elite level. Medical Journal of Australia, 159(5), 298–301.
Sherry, M.A. and Best, T.M. (2004) A comparison of 2 rehabilitation programs in the treatment of acute hamstring strains. Journal of Orthopaedic and Sports Physical Therapy, 34 (3), 116–125.
Snyder-Mackler, L., Delitto, A. and Bailey S. (1995) Strength of the quadriceps femoris muscle and functional recovery after reconstruction of the anterior cruciate ligament. Journal of Bone and Joint Surgery, 77A, 1166–1173.
Stanton, P.E. (1989) Hamstring injuries in sprinting – the role of eccentric exercise. Journal of Orthopaedic and Sports Physical Therapy, 10, 343–9.
Stockmar, C., Lill, H., Trapp, A., Josten, C. and Punkt, K. (2006) Fibre type related changes in the metabolic profile and fibre diameter of human vastus medialis muscle after anterior cruciate ligament rupture. Acta Histochemica, 108 (5), 335–42.
Taylor, D.C., Dalton, J.D., Seaber, A.V., Garrett, W.E. (1993) Experimental muscle strain injury. Early functional and structural deficits and the increased risk for reinjury. American Journal of Sports Medicine, 21, 190–194.
Ter-Haar, G., Dyson, M. and Tlabert, D. (1978) Ultrasonically induced contraction of mouse uterine smooth muscle in-vivo. Ultrasonics, 16, 275–276.
Thornton, R., Mendel, F. and Fish, D. (1998) Effects of electrical stimulation on edema formation in different strains of rats. Physical Therapy, 78, 386–394.
Thorsson, O., Lilja, B., Nilsson, P. and Westlin, N. (2007) Immediate external compression in the management of acute muscle injury. Scandinavian Journal of Medicine and Science in Sports, 7 (3), 182–190.
Tidball, J. G., and Wehling-Henricks, M. (2007) Macrophages promote muscle membrane repair and muscle fibre growth and regeneration during modified muscle loading in mice in vivo. Journal of Physiology, 578 (1), 327–336.
Tsang, K.K., Hertel, J., Denegar, C. (2003) Volume decreases after elevation and intermittent compression of postacute ankle sprains are negated by gravity-dependent positioning. Journal of Athletic Training, 38, 320–324.
Verrall, G.M., Slavotinek, J.P., Barnes, P.G., Fon, G.T. and Spriggins, A.J. (2001) Clinical risk factors for hamstring muscle strain injury: a prospective study with correlation of injury by magnetic resonance imaging. British Journal of Sports Medicine, 35, 435–439.
Wilkin, L.D., Merrick, M.A., Kirby, T.E. and Devor, S.T. (2004) Influence of therapeutic ultrasound on skeletal muscle regeneration following blunt contusion. International Journal of Sports Medicine, 25 (1), 73–77.
Witvrouw, E., Bellemans, J., Lysens, R., Danneels, L., and Cambier, D. (2001) Intrinsic risk factors for the development of patellar tendonitis in the athletic population. A two year prospective study. American Journal of Sports Medicine, 29 (2), 190–195.
Woods, C., Hawkins, R., Hulse, M. and Hodson, A. (2002) The Football Association medical research programme: an audit of injuries in professional football – analysis of preseason injuries. British Journal of Sports Medicine, 36 (6), 436–441.
Woods, C., Hawkins, R. D., Maltby, S., Hulse, M., Thomas, A. and Hodson, A. (2004) Football Association Medical Research Programme. The Football Association Medical Research Programme: an audit of injuries in professional football – analysis of hamstring injuries. British Journal of Sports Medicine, 38 (1), 36–41.
Worrell, T.W. (1994) Factors associated with hamstring injuries: an approach to treatment and preventative measures. Journal of Sports Medicine, 17 (5), 338–345.
Worrell, T.W., Smith, T.L. and Winegardner, J. (1994) Effect of hamstring stretching on hamstring muscle performance. Journal of Orthopaedic and Sports Physical Therapy, 20 (3), 154–159.
Yamamoto, T. (1993) Relationship between hamstring strains and leg muscle strength. A follow up study of collegiate track and field athletes. Journal of Sports Medicine and Physical Fitness, 33 (2), 194–199.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree