Osteochondral Plug Transplantation
László Hangody
Zsófia Duska
Zoltán Kárpáti
INDICATIONS/CONTRAINDICATIONS
Efficacious treatment of full-thickness cartilage defects of the weight-bearing surfaces represents a multifaceted challenge for the orthopaedic surgeon. During the recent decade many efforts have been made to improve the quality of different cartilage repair techniques aiming to provide a hyaline-like surface in the resurfaced area. Autologous osteochondral transplantation represents one solution, enabled by promotion of a hyaline or hyaline-like repair of the affected area. Several series of dog and horse experiments and 14 years of clinical experience proved that autologous osteochondral mosaicplasty is a useful alternative in the treatment of full-thickness cartilage defects. Open and arthroscopic use of the mosaicplasty technique has already extended clinical experience, and recent data suggest both indications and contraindications. Besides femoral and tibial condylar and patellotrochlear applications, talar, femoral head, humeral head, and capitulum humeri implantations were carried out. Preclinical animal studies and subsequent clinical practice have confirmed the survival of the transplanted hyaline cartilage as well as fibrocartilage filling of the donor sites (located on the non-weight-bearing and less-weightbearing surfaces of the knee joint). Clinical scores, several types of different imaging techniques, control arthroscopies, histologic examination of biopsy samples, and cartilage stiffness measurements have been used to evaluate the clinical outcome and quality of the transplanted cartilage, and have confirmed the clinical efficacy of the mosaicplasty technique. This chapter discusses the indications, contraindications, technical details, rehabilitation, and pitfalls and complications of the procedure.
If the preoperative differential diagnosis includes a small or medium-sized focal defect of the weight-bearing surfaces, the patient should be advised of the possibility of an autologous osteochondral mosaicplasty. In such cases, the patient should also be prepared for an open procedure. The site or the size of the lesion can require a miniarthrotomy or an open procedure; however, the main goal should be an arthroscopic approach for this type of cartilage transplantation. Open procedures can lead to an overnight stay and altered weight-bearing status for several weeks.
Transplantation of osteochondral autografts is not new, but its clinical application was restricted because of certain technical difficulties, such as congruency and donor site problems. Osteochondral autograft donor sites must be taken from relatively non-weight-bearing surfaces, which limits the procurement field. On the other hand, use of large grafts can cause certain incongruities at the recipient site, permanently altering the biomechanics of the joint. Instead of single-block transfer, the use of small multiple cylindrical grafts may permit more tissue to be transplanted while preserving donor site integrity, and their use in a mosaic-like implanting fashion would allow progressive contouring of the new surface. Autologous osteochondral mosaicplasty has been introduced in clinical practice in 1992 in Hungary. This type of resurfacing can be indicated for 1- to 4-cm2 focal chondral or osteochondral defects of the weight-bearing area. The most common recipient sites are the femoral condyles, but clinical experience with the patellotrochlear, tibial, and talar applications is fairly extensive. As far as exceptional applications are concerned, a few implantations for certain osteochondral defects of the humeral and femoral head as well as the capitulum humeri were carried out.
As with other types of resurfacing, mosaicplasty requires the elimination of the underlying causes (if this is possible) in the same step. According to clinical experience, anterior cruciate ligament (ACL) reconstruction, meniscus surgery, and femorotibial realignment procedures are the most common concomitant procedures, but patellofemoral realignment techniques, lateral release, and so on, may also be required.
Any disadvantageous condition hindering the survival of the transplanted hyaline cartilage on the recipient site can be a contraindication, such as the following:
Generalized arthritis, rheumatoid and/or degenerative type
Lack of appropriate donor area
Infectious or tumor defects
Age greater than 50 years
Osteochondral defects deeper than 10 mm
PREOPERATIVE PLANNING
Localized full-thickness cartilaginous lesions are usually defined at arthroscopy, but preoperative clinical investigation may also give some useful information. Anamnestic data and actual clinical findings (e.g., tenderness in the medial joint space, swelling, clicking) may support the presence of a cartilage defect, but there are no specific signs for determining the exact type and location of existing chondral damage. Osteochondral lesions usually show more expressed complaints. Despite that small- or medium-sized chondral and osteochondral damages do not give a characteristic picture, the clinical examination is necessary to eliminate the actual biomechanical problems. Stability of the affected knee, femorotibial alignment, and patellofemoral traction conditions should be cleared at the clinical examination.
Standard and standing radiographic examinations are basic elements of the preoperative diagnostics. Computed tomography (CT) may inform about the subchondral bony condition. Ultrasound investigation or special sequences of magnetic resonance imaging (MRI) can give useful information about the location and extension of the chondral defects, but severity cannot always be determined exactly. The last step is usually the arthroscopic examination, which should determine the exact location and stage of the damage, evaluate the quality of the donor area, and check all the other intra-articular conditions.
Preoperative preparations should include antibiotic prophylaxis. General or local anesthesia with tourniquet control is recommended.
SURGERY
The patient is positioned supine with the knee capable of 120 degrees of flexion. The contralateral extremity is placed in a stirrup. Standard arthroscopic instrumentation and MosaicPlasty Complete System (Smith & Nephew, Inc., Endoscopy Division, Andover, MA) are required. Beside these reusable instruments, disposable chisels, drillbits, and tamps are also available to provide ideal conditions for precise graft harvest and tunnel preparation (Dispoposplasty System, Smith & Nephew, Inc., Endoscopy Division, Andover, MA). A fluid management system may support the procedure.
Choosing a procedure (arthroscopic or miniarthrotomy) depends on the type, size, and exact location of the defect determined during arthroscopy. As placing the grafts perpendicular to the surface is paramount to the success of the operation, the first task is to determine whether an arthroscopic or open procedure is required. Although certain trochlear defects can be resurfaced arthroscopically, patellotrochlear and tibial lesions always require an open procedure. Most of the femoral condylar defects can be managed arthroscopically. As for most of these lesions, central anterior medial and central anterior lateral portals will allow correct perpendicular access.
An open procedure may be chosen in the learning curve or when an arthroscopic approach is not practical due to size or location of the lesion. Arthroscopic or open mosaicplasties have the same steps and technique.
Arthroscopic Mosaicplasty
Portal Selection
As has been emphasized previously, perpendicular access to the lesion is critical to proper insertion of the grafts. Take care in making the viewing and working portals. Use a
1.2-mm K-wire or 18-gauge spinal needle initially to locate the portal sites. It should be noted that these portals tend to be more central than the standard portals due to the inward curve of the condyles (Fig. 28-1). For osteochondritis dissecans on the medial femoral condyle, the approach should be from the lateral side. Standard lateral portal is sometimes too oblique. Therefore, use the central patellar tendon portal, which gives good access to the inner positions of both the medial femoral condyle and the lateral femoral condyle.
1.2-mm K-wire or 18-gauge spinal needle initially to locate the portal sites. It should be noted that these portals tend to be more central than the standard portals due to the inward curve of the condyles (Fig. 28-1). For osteochondritis dissecans on the medial femoral condyle, the approach should be from the lateral side. Standard lateral portal is sometimes too oblique. Therefore, use the central patellar tendon portal, which gives good access to the inner positions of both the medial femoral condyle and the lateral femoral condyle.
Defect Preparation
Use a full-radius resector or curette and a knife blade to bring the edges of the defect back to good hyaline cartilage at a right angle. Clean the base of the lesion with an arthroscopic burr (Abrader, Acromionizer) or half-round rasp to viable subchondral bone (Fig. 28-2). Abrasion arthroplasty of the defect site promotes fibrocartilage grouting from the bony base. Because tapping the cutting edge of the guide into the bony base and removal of it can mark the defect site, use the drill guide to determine the number and size of grafts needed (Fig. 28-3). Filling of the defect by same-sized contacting rings allows a filling rate of about 70% to 80%, but use of additional sizes to cover the dead spaces and cutting the grafts into each other can improve the coverage to 90% to 100% (Fig. 28-4). Finally, measure the depth of the defect with the laser marks of the dilator.
Graft Harvest
The medial femoral condyle periphery of the patellofemoral joint above the line of the notch is the preferred arthroscopic harvest site. The lateral femoral condyle above the sulcus terminalis and, in exceptional cases, the notch area can serve as additional donor areas (Fig. 28-5). Grafts harvested from the notch area are less favorable features, as they have concave cartilage caps and less elastic underlying bone. The medial patellofemoral periphery has easier access than the lateral one as fluid distension can promote lateral positioning of the patella and may provide easier perpendicular positioning for the harvesting chisel.
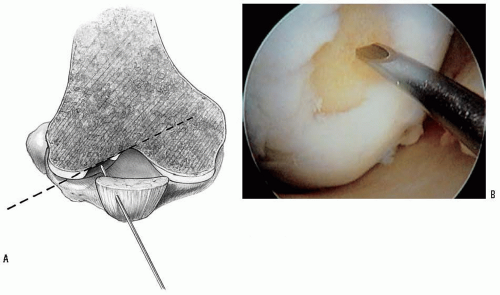 FIGURE 28-1 A: Use of a spinal needle to determine the perpendicular access to the defect. B: Arthroscopic picture of the same step. |
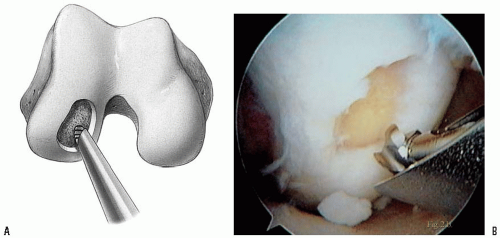 FIGURE 28-2 A: Abrasion arthroplasty of the bony base of the defect. B: Arthroscopic picture of the same step. |
The best view for harvesting grafts is obtained by introducing the scope through the standard contralateral portal. Extend the knee and use the standard ipsilateral portal to check the perpendicular access to the donor site. Extended position should provide perpendicular access to the most superior donor hole. Step-by-step flexion allows the harvest of additional grafts from the lower portions of the patellofemoral periphery. If the standard portals do not allow a perpendicular approach, use a spinal needle or a K-wire to determine the location of additional harvesting portals.
Once the necessary portal has been determined, introduce the proper-sized tube chisel filled with the appropriate harvesting tamp. Once the site has been clearly identified, the chisel is located perpendicular
to the articular surface and driven by a hammer to the appropriate depth. The minimal length of the graft should be at least two times its diameter, but, as a rule, take 15-mm-long grafts to resurface chondral lesions and 25-mm-long plugs for osteochondral defects. It is important to hold the chisel firmly to avoid its shifting at the cartilage-bone interface, producing a crooked graft. By flexing the knee, lower sites can be obtained. The lower limit is the level of the top of the intercondylar notch (sulcus terminalis). Insert the appropriate harvesting tamp into the cross-hole in the tubular chisel and use it as a lever. The chisel should be toggled, not rotated, causing the graft to break free at the chisel tip (Fig. 28-6). Eject the grafts from the chisel by sliding the appropriately sized chisel guard over the cutting end. Use the tamp to push out the graft onto gauze in a salinewetted basin (Fig. 28-7



to the articular surface and driven by a hammer to the appropriate depth. The minimal length of the graft should be at least two times its diameter, but, as a rule, take 15-mm-long grafts to resurface chondral lesions and 25-mm-long plugs for osteochondral defects. It is important to hold the chisel firmly to avoid its shifting at the cartilage-bone interface, producing a crooked graft. By flexing the knee, lower sites can be obtained. The lower limit is the level of the top of the intercondylar notch (sulcus terminalis). Insert the appropriate harvesting tamp into the cross-hole in the tubular chisel and use it as a lever. The chisel should be toggled, not rotated, causing the graft to break free at the chisel tip (Fig. 28-6). Eject the grafts from the chisel by sliding the appropriately sized chisel guard over the cutting end. Use the tamp to push out the graft onto gauze in a salinewetted basin (Fig. 28-7
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








