Chapter 20 Intestinal Permeability Assessment
 Gut Mucosal Integrity: Mechanisms of Disruption
Gut Mucosal Integrity: Mechanisms of Disruption
A key function of healthy gut mucosa is to prevent bacteria and their endotoxins from reaching portal circulation. For this purpose, a natural physiologic barrier is formed by the mucous layer, the epithelial cells and apical junctional complex, and the lamina propria (Figure 20-1, A). When one or more of these components is impaired, damage to the intestinal barrier may result, allowing luminal toxins to translocate from the gut into the systemic circulation.1,2
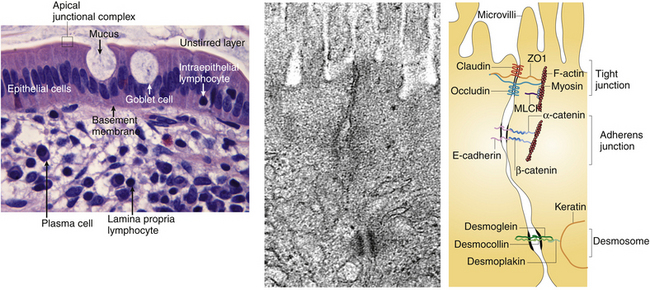
FIGURE 20-1 Anatomy of mucosal barriers.
(Reprinted by permission from Macmillan Publishers Ltd. From Turner J. Intestinal mucosal barrier function in health and disease. Nature Rev Immunol 2009:9:799-809. http://www.nature.com/nri.)
The small intestine consists of specific intrinsic factors that help protect the host from environmental pathogens. These are the enterocytes as well as the paracellular spaces composed of structures called tight junctions, adherens junctions, and desmosomes (Figure 20-1, B). The tight junctions surround the upper portion of the lateral surfaces and the adjacent epithelial cells to create fusion points. This creates distinct fluid compartments that maintain cellular polarity and regulate the passive transepithelial movement of molecules.3
The tight junction zonula occludins (ZO1) is located just below the base of the microvilli and acts as the primary permeability barrier between the external and internal milieus of the body.2 Transmembrane proteins that form part of the tight junction include the claudins and occludin families. Claudins proteins act as sealing proteins, forming ion-selective pores within the tight junction strands, whereas occludin proteins are thought to have adhesive and transducing properties. Both claudins and occludin interact with cytosolic complexes such as actin and myosin, which serve as contractile elements to open and close the paracellular junctions.4 Transmembrane proteins can be affected by cytokines, and minor defects in the paracellular pathway can set up a vicious cycle, whereby antigen presenting cells (e.g., dendritic cells) stimulate the production of Th-1 cells that increase the production of tumor necrosis factor-γ (TNF-γ) and interferon-γ (IFN-γ) and further increase tight junction permeability.3
Gut immunity consists of two distinct mucosal defense mechanisms: the specific system and the nonspecific system. The specific system is composed of secretory immunoglobulin-A (sIgA). The nonspecific system comprises gastric acidity, motility, mucus, digestive enzymes, normal bacterial flora, and bacterial antagonism. Disruptions in gut barrier function may result from impaired host immunity, alterations in gut flora, or direct mucosal injury.5
Ordinarily, normal gut microflora provides “resistance to colonization,” preventing the adherence of pathogenic organisms to the intestinal wall and their subsequent translocation.6 Changes in gut ecology, however, can reduce this natural resistance and encourage the colonization of potentially harmful bacterial species.
Bacteria and the toxins they produce can alter membrane integrity through various mechanisms. Bacteria can secrete enzymes, such as elastase and protease, which hydrolyze the epithelial membrane and cause cells to detach from the epithelial sheet. Bacterial toxins may act on or within membranes and compromise tight junction integrity by binding to membrane receptors. Some toxins can traverse the cell membrane and inhibit cellular growth by altering protein synthesis. They may also alter membrane lipid composition, act as detergents, or form transmembrane pores. The net effect is cellular swelling, lysis, loss of intracellular constituents, and uncontrolled influx of toxic substances.7
Bacterial endotoxins and cytokines have been shown to impair barrier function by activating inducible nitric oxide synthase. Upregulation of this enzyme increases the amount of nitric oxide. Prolonged exposure of the cells to nitric oxide is thought to inhibit cellular respiration, decrease mesenteric blood flow, and alter intestinal permeability.8
Clinical Applications
Because the intestinal mucosa plays a pivotal role in immune defense and nutrient absorption, a wide variety of diseases and conditions are associated with abnormal bowel permeability (Box 20-1). Abnormal permeability is often identified through assessment of the relative rate at which two nondigestible sugars, lactulose and mannitol, permeate the intestinal mucosa. A higher lactulose-to-mannitol (L/M) ratio indicates increased gut permeability (see section on “Measuring Intestinal Permeability” on page x).
Inflammatory Bowel Disease
To date, it is not certain if the altered intestinal permeability seen in patients with Crohn’s disease is a consequence or a cause of the disease.9 Recent research by Takeuchi et al10 favored the notion that increased gut permeability is secondary to intestinal inflammation. In assessing healthy, first-degree relatives of patients with Crohn’s disease, they found a 50% prevalence of subclinical inflammation as evidenced by an increase in fecal calprotectin (a protein marker that reflects white blood cell infiltration into the gut lumen). Expanding this data, Meddings11 proposed a model for the development of inflammatory bowel disease (IBD) that necessitates the presence of at least three conditions: (1) an abnormal immune system that interacts inappropriately with environmental antigens, (2) the presence of an antigenic agent, and (3) a breach in gut barrier function that allows the antigen to reach the immune system.11
Measuring intestinal permeability in asymptomatic patients with Crohn’s disease can help monitor progress and predict disease relapse. A normal lactulose recovery and L/M ratio is associated with good short-term (6 to 12 months) prognosis, whereas increased intestinal permeability is indicative of a relapse within 6 months.10
Celiac Disease
Gut barrier function is altered in nearly all patients with untreated celiac disease. Intestinal permeability testing is therefore an ideal noninvasive screening test, offering a sensitivity of 89%, a specificity of 54%, and negative predictive value of 95% compared with jejunal biopsy.12 Although jejunal biopsy is considered the gold standard for identifying celiac disease, this test is time-consuming, unpleasant, and costly, making it unsuitable as a screening tool for the general population.13 An elevated L/M ratio helps identify which patients should undergo jejunal biopsy when clinical features are compatible with celiac disease. It can also help determine the timing of follow-up biopsy and monitor the recovery of the mucosa.14 Impaired permeability tends to normalize within 5 months when patients strictly adhere to a gluten-free diet. Serial intestinal permeability testing can also be used to monitor dietary compliance in these patients.12
Diabetes
An emerging body of evidence suggests altered gut barrier function may be an etiologic factor in the pathogenesis of type 1 diabetes.4,15–17 Bosi et al18 assessed intestinal permeability in 81 subjects with islet cell autoimmunity, 18 of whom were in the preclinical phase, 28 with newly diagnosed type 1 diabetes, and 35 with a long-term history of the disease. The researchers found an elevation in lactulose and the L/M ratio across all groups, with the highest levels reported in the preclinical group. These results suggest that increased permeability tracks the course of the autoimmune process and precedes the clinical onset of type 1 diabetes.
Alterations in gut permeability in type 1 diabetics are thought to arise from luminal antigens, such as bovine insulin from cow’s milk, gliadin from wheat, enteral viruses (e.g., enteroviruses and rotavirus), and/or disturbed microbiota. The increased antigenic load appears to activate T cells in both the small intestine and the pancreatic lymph nodes, causing β-cell damage through autoimmunity and Th-1 cytokine upregulation.19 Type 2 diabetes does not appear to be associated with primary changes in the intestinal mucosal epithelium.20
Inflammatory Joint Diseases
Numerous studies have shown a correlation between different types of arthritides and increased intestinal permeability.21,22 It is estimated that between 30% and 60% of such patients have occult intestinal inflammation.23 To date, however, it has not been fully determined whether this correlation results from the disease process itself or is due to mucosal damage caused by treatment with nonsteroidal anti-inflammatory drugs (NSAIDs).24–27
Evidence for a primary cause of intestinal barrier dysfunction may be related to gut microbial infections. Specific pathogenic organisms (Campylobacter, Klebsiella, Proteus, Shigella, Salmonella, and Yersinia) have been found in patients with various arthritides who have altered intestinal permeability.28–31 It has been postulated that gut bacterial antigens, when absorbed by the intestinal mucosa, elicit an immune response, with tissue damage occurring from molecular mimicry.32 Measuring intestinal permeability before and after NSAID therapy in patients with inflammatory joint disease helps establish whether mucosal disruption is solely an iatrogenic response.
Food Allergies
The mucosal surface of the small intestine is the largest surface area in the body that comes into direct contact with the external environment and is estimated to be more than 200 m2.33 The advantage of such a vast internal surface area is that dietary nutrients can be rapidly absorbed.34 When there is a breach of gut barrier function, dietary antigens and other luminal toxins can also be translocated across the intestinal wall. Recent research suggests that environmental factors, such as infection or stress, increase gut permeability, which in susceptible individuals can contribute to food allergies.17
For food allergies to manifest, intraluminal antigens must first penetrate the intestinal epithelial barrier before gaining access to mast cells in the subepithelial compartment and triggering hypersensitivity reactions. This pattern has been observed in children with atopic eczema associated with cow’s milk allergy.33 Under normal conditions, most dietary proteins are digested by gastric and pancreatic proteases and brush-border enzymes and transformed into peptides and amino acids before being absorbed by the enterocytes.17,35 In healthy individuals, a small amount of intact proteins in the gut are taken up by enterocytes and broken down inside lysosomal compartments, which prevents undigested proteins from entering the subepithelial compartment.35,36 In atopic individuals, CD23 (the low affinity receptor located on the apical membrane of the epithelial cell) is overexpressed, which leads to the protected transfer of IgE/allergen complexes and transepithelial passage of intact allergens.
Repeated exposure to IgE-mediated antigens places significant demands on the gut mucosa and further increases intestinal permeability, setting up a self-perpetuating cycle that maintains the allergenic state.33,36 In addition to IgE production, the sensitization phase of allergy involves the upregulation of Th-2 cytokines (interleukin-4 [IL-4], IL-5, and IL-13).35 In the animal model, increased production of IL-4 was found to upregulate CD23 expression. This finding may in part explain the role of IL-4 in enhancing intestinal permeability.36
Given the recent scientific findings, a multifactorial approach to food allergy management has been proposed; it includes allergen elimination as well as reducing inflammatory reactions and stabilizing the gut mucosal barrier.33
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


