Chapter 8 Graft Options for Anterior Cruciate Ligament Revision Reconstruction
INTRODUCTION
It has been estimated that approximately 100,000 anterior cruciate ligament (ACL) reconstructions are performed annually in the United States.7 This represents a 30% to 40% increase from previous reports.20 The procedure has a successful outcome in 75% to 95% of cases; however, 8% may experience an unsatisfactory result owing to recurrent instability and graft failure.16 It is reasonable to assume that with an increasing number of ACL reconstructions, orthopaedic surgeons are more likely to encounter a patient with a failed reconstruction. Failure has not been universally defined, but in general, an ACL graft is consider failed when there is an increase in anteroposterior (AP) translation of more than 5.5 mm compared with the contralateral knee during instrumented knee arthrometer testing or when a grade II or III pivot shift test is elicited during physical examination. When planning a revision ACL reconstruction, one of the most important and controversial decisions is the choice of graft. In this chapter, the merits of various graft sources (autografts, allografts, and rarely in North America, synthetic grafts) are discussed as well as the advantages and disadvantages of different anatomic graft types. Finally, the authors present their preferred approach.
SYNTHETIC GRAFTS AND AUGMENTATION DEVICES
Gore-Tex Ligament
The Gore-Tex synthetic ligament consists of woven strands of polytetrafluoroethylene with molded eyelets at each end that allow fixation of the graft to the bone with screws (Fig. 8-1). This prosthetic device is intended to function as a permanent load-bearing implant with an ultimate tensile strength of 5300 N. In 1986, this device received approval from the U.S. Food and Drug Administration (FDA) for limited clinical use in patients with failed ACL reconstruction. The Gore-Tex ligament is placed in an over-the-top location. Femoral fixation in a bone tunnel is contraindicated because abrasion of the ligament at the entrance of the tunnel can cause premature failure. Long-term outcome studies of patients who received this artificial ligament have shown deterioration of results and high failure rates. Paulos and associates39 reported on 188 patients who received Gore-Tex reconstructions followed an average of 48 months postoperatively. Unacceptable results (fair and poor) were obtained in 56% of the patients. Rupture of the ligament occurred in 12% of the cases (Fig. 8-2). Loosening of greater than 3 mm was observed in 34% of the patients, which led to an unacceptable result in nearly one half. Thirty-four percent of the patients suffered from recurrent effusions. The authors concluded that the use of this ligament in primary as well as revision ACL reconstructions led to unacceptable results in 76% of the cases.
Stryker Dacron Ligament
The Stryker Dacron prosthetic ligament (Fig. 8-3) is intended to function as a permanent prosthesis. This device has an ultimate tensile strength of 3600 N and was approved in 1989 for use in failed intra-articular ACL reconstructions. It can be implanted either using an over-the-top technique or through a bone tunnel. Similar to the Gore-Tex ligament, long-term studies have shown high failure rates. Wredmark and Engstrom54 reported 5-year results in a group of 42 patients. During the observation period, the failure rate was nearly 60% based on ligament examination (instrumented knee arthrometer and/or pivot shift test). Eighty percent of the patients had radiographic evidence of ligament rupture. Histologic examination of the failed ligaments during revision surgery revealed an inflammatory or foreign body reaction, and not the expected organized fibrous ingrowth.
Kennedy Ligament Augmentation Device
The ligament augmentation device (LAD) polypropylene graft, in contrast to the grafts described previously, is intended to function as a load-sharing implant to protect a biologic graft while it heals (Fig. 8-4). The FDA released this product in 1986 for use with the Marshall-McIntosh ACL reconstruction, but it has also been used with autogenous bone–patellar tendon–bone (B-PT-B) and hamstring grafts. The device is manufactured in widths of 8 mm and 6 mm, with ultimate tensile strengths of 1730 N and 1500 N, respectively. The Kennedy LAD was perhaps the most widely used prosthetic device in ACL reconstruction. Although complications from its use such as synovitis, breakage, or painful hardware have been reported,5 the device fell out of favor mainly because comparison studies failed to demonstrate any advantage with its use for autograft or allograft augmentation.30,32
ALLOGRAFTS
Graft Options
The wide availability of musculoskeletal allografts in North America has resulted in an ever-increasing utilization of these tissues in orthopaedic surgery. According to data from the American Association of Tissue Banks (AATB), a voluntary accreditation organization that sets standards for tissue banking, approximately 1.5 million bone and soft tissue allografts are distributed each year by AATB-accredited tissue banks in the United States.41 Allografts are appealing during revision ACL reconstruction in which autograft options may be limited or not available. The morbidity associated with autograft harvest is avoided and the operative time is decreased. A variety of allograft tissue is available for ACL reconstruction. The most popular tissues are the Achilles tendon (AT), B-PT-B, quadriceps tendon (QT), hamstring tendons, and the tibialis anterior (TA) tendon. Other less frequently used options are the tibialis posterior tendon, peroneus longus tendon, and fascia lata. These grafts provide a variety of length, width, and strength options and allow for soft tissue or bone fixation. The biomechanical properties of the TA, tibialis posterior (TP), and the peroneus longus (PL) tendon have recently been evaluated. Their ultimate tensile strength when tested as doubled grafts was 3144 N, 3391 N, and 2483 N, respectively. The TA had the greatest stiffness (344 N/mm), followed by the TP (302 N/mm) and the PL (244 N/mm).40 These values compare favorably with historical data of other graft sources.35 In addition, allografts harvested with bone blocks at their end(s) such as the AT, B-PT-B, or QT, may allow greater flexibility during revision reconstructions when bone loss is encountered. In such cases, shaping the bone block(s) of the allograft to match a widened preexisting tunnel or to fill a bone defect from removed hardware can obviate the need for a staged reconstruction. These benefits, however, have to be weighed against certain disadvantages of allografts that are associated with their procurement, processing, and sterilization.
Disease Transmission
One of the major concerns related to the use of allografts is disease transmission. The incidence of infection after allograft knee reconstruction has not been accurately determined. Both bacterial and viral infections have been reported. As of March 11, 2002, the U.S. Centers for Disease Control and Prevention (CDC) had received 26 reports of bacterial infections associated with musculoskeletal tissue allografts. Eighteen of these cases were associated with grafts used for ACL reconstruction. Thirteen (50%) of the 26 patients were infected with Clostridium species (12 with C. septicum, one with C. sordellii). All allografts were processed aseptically, but did not undergo terminal sterilization.3 In response, additional steps were taken to reduce the risk of disease transmission including sterilization methods that kill bacterial spores, swab and destructive cultures, rejection of tissues contaminated by bowel flora, and implementation of time limits for the procurement of the donors (12 hr when the donor is procured at room temperature, 24 hr for refrigerated donors). However, in 2003, an additional case of septic arthritis due to Streptococcus pyogenes after allograft ACL reconstruction was reported, despite adaptation of these measures.2 The risk of viral disease transmission is lower, and most incidents have occurred before the pathogen was known or when the available technology could not identify its presence in the donor owing to a “serologic window.” There have been reports of hepatitis C, and human immunodeficiency virus (HIV) transmission from tendon allografts used in knee reconstruction.1,47 The risk of HIV transmission from musculoskeletal allografts has been estimated to be as low as 1 in 1,667,600.8
Donor Screening and Sterilization Techniques
Currently, allograft donors are initially screened with a detailed medical and social history of the donor and postmortem examination for signs of infectious disease. Blood cultures as well as cultures from the harvested grafts are taken. The AATB requires a serologic examination for HIV type 1 and 2 antibodies, HIV-1 DNA by polymerase chain reaction, hepatitis B surface antigen, hepatitis B core antibody, hepatitis C antibody, human T-cell lymphotropic virus types 1 and 2 antibodies, and syphilis antibody. The introduction of newer testing methods such as nucleic acid testing can decrease the window period associated with conventional antigen and antibody testing and increase the safety of allografts. The Board of Governors of the AATB voted to require AATB-accredited tissue banks to adopt this newer testing method for HIV-1 and hepatitis C by March of 2005.29
Several disinfection and sterilization methods have been used to eliminate bacteria, spores, and viruses from allografts.53 Currently, there is no single standard approach to eliminate pathogens from allografts. Furthermore, some of these methods can alter the biomechanical properties of soft tissue grafts. Aseptic harvesting and processing alone preserve the strength of tendon grafts, but these do not eliminate bacteria, fungi, spores, or viruses. In addition, this method removes blood and lipids from only the surface of the graft. Chemical soaking can eliminate bacteria and fungi, but this process does not eradicate spores or viruses.
Two commonly employed methods for terminal sterilization are gamma irradiation (GI) and ethylene oxide (EO). GI is effective against bacteria at doses of 1.5 to 2.5 Mrad. However, as much as 4.0 Mrad is required to inactivate HIV, and even higher doses may be necessary to kill spores.13 Unfortunately, higher doses of GI result in a substantial decrease of the strength of tendinous grafts. Fideler and colleagues14 studied the effects of GI on B-PT-B allografts. These investigators noted a 15%, 24%, and 46% reduction in all biomechanical properties of the grafts after exposing them to doses of 2.0, 3.0, and 4.0 Mrad of irradiation, respectively. EO has successfully been used to sterilize surgical instruments. However, its use as a method to sterilize ACL allografts has been associated with intra-articular reactions with chronic synovitis, graft failure, and bone dissolution developed from the chemical residues that EO leaves in moist tissues.19
In an attempt to ameliorate the problems associated with each individual method of sterilization, several companies have developed proprietary techniques to process allografts that combine the methods previously discussed or introduce new ones.50 Cryolife, Inc. (Kennisaw, GA) uses slow freezing combined with a dehydrating solvent, such as dimethyl sulfoxide or glycerol, to cryopreserve allografts. After desiccation, the grafts are treated for an extended period of time with an antimicrobial solution. No secondary sterilization technique is used. BioCleanse (Regeneration Technologies, Inc., Alachua, FL) uses a low-temperature chemical sterilization method, which allows better tissue penetration of the chemical and thus eliminates endogenous contamination from the allografts.
Healing of Allografts Postimplantation
Biologic integration and tissue compatibility merit consideration when allografts are used in primary or revision ACL reconstruction. Allografts follow the same healing process observed with autograft reconstruction of an initial period of avascular necrosis, followed by revascularization and cell proliferation.4 However, several studies have documented that this integration occurs at a much slower rate. Jackson and coworkers18 evaluated patellar tendon autografts and fresh frozen allografts 6 months after ACL reconstruction in a goat model. Compared with the allografts, the autografts demonstrated a smaller increase in AP displacement, two-times greater values of maximum force to failure, a significant increase in cross-sectional area, a more rapid loss of large-diameter collagen fibrils, and an increased density and number of small-diameter collagen fibrils. The allografts had a greater decrease in structural properties, a slower rate of biologic incorporation, and a prolonged presence of an inflammatory response. The autografts demonstrated a more robust biologic response, improved stability, and increased strength to failure values.
More recently, Malinin and associates28 studied nine ACL allografts and one autograft, retrieved at autopsy or at the time of revision surgery, to determine the rate and the extent of graft cellular replacement and remodeling. Examination of specimens from 20 days to 10 years after transplantation revealed a pattern of revascularization similar to that reported in previous biopsy studies. However, examination of entire allografts 2 years after transplantation revealed that the central portions of the grafts remained acellular and complete attachment was not present. One specimen retrieved 3.5 years post-transplantation did demonstrate complete attachment. The authors concluded that complete remodeling and cellular replacement of the entire graft may require 3 years or longer.
Similar findings were documented in a study by Beck and coworkers.6 These authors obtained biopsies during arthroscopy from ACL allografts at various times after implantation. Histologic examination of the specimens showed that allografts remain largely avascular and acellular as late as 8 years postimplantation (Fig. 8-5).
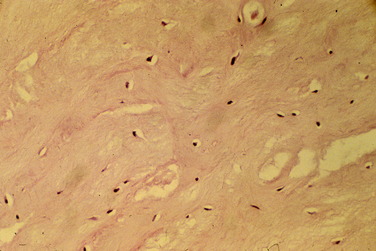
FIGURE 8-5 Histologic examination of specimens from Beck and coworkers6 shows that allografts remain largely avascular and acellular as late as 8 years postimplantation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree