CHAPTER 34 Endoscopic Carpal Tunnel Release: Chow Technique
Sir James Paget first described median nerve compression after a distal radius fracture in 1854.1,2 In 1880, James Putman, a neurologist from Boston, reported similar symptoms in a group of his patients.3 The first formal description of surgical decompression for treatment of this condition was reported in 1933; this was followed by Phalen’s classic article in 1950.4 Since then, open carpal tunnel release has been established as the gold standard for surgical treatment of carpal tunnel syndrome.
The first two reports in the literature on the topic of endoscopic carpal tunnel release (ECTR), written separately by Chow and by Okutsu and colleagues, were published in the March issue of Arthroscopy in 1989.5,6 In the following year, Chow presented a paper describing his clinical results after ECTR in 149 cases at the 1990 Arthroscopy Association of North America (AANA) Annual Meeting in Orlando, Florida. In the fall of 1990, Agee and associates7 reported to the American Society for Surgery of the Hand Annual Meeting in Toronto, Canada, the clinical results of a multicenter study of the use of Agee’s technique for endoscopic release of the carpal ligament. Since the publication of these three endoscopic carpal ligament release techniques, there has been increasing interest and much debate among surgeons regarding the safety and efficacy of the endoscopic procedures compared with the open technique. Several modifications and variations of the three original ideas have been made since their initial demonstration.
ANATOMY
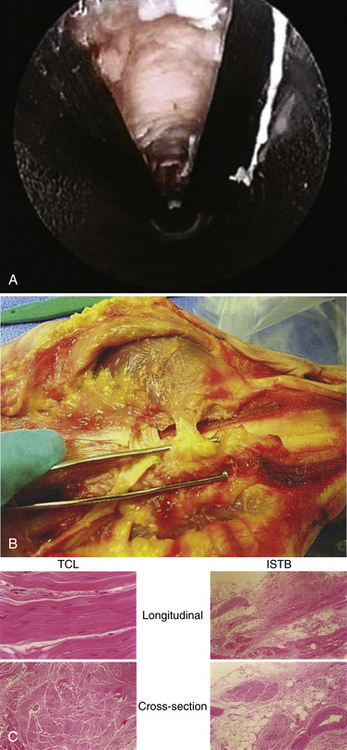
Superficial to the transverse carpal ligament lies a sling of vascular and nervous connective tissue, subcutaneous fat, and densely innervated palmar dermis and epidermis. This sling of soft tissue is an interthenar soft tissue band (ISTB) (Fig. 34-1A and B) that bridges the thenar and hypothenar musculature. It is a histologically distinct structure (see Fig. 34-1C) that lies volar to the transverse carpal ligament. The ISTB and the palmaris brevis muscle should be preserved. The ISTB prevents bowstringing of the flexor tendons after surgery, thereby maintaining their strength during contraction.8–10 It is the goal of ECTR to preserve this natural anatomy while selectively releasing only the transverse carpal ligament.
Carpal tunnel syndrome is a compressive neuropathy of the median nerve at the wrist. It can result from any condition that increases intra–carpal tunnel pressure, such as irritation and swelling of any of the nine flexor tendons and their tenosynovium, edema of the median nerve, anatomic abnormality or scarring of any of the bordering structures of the carpal tunnel, anomalous lumbrical anatomy, an intra–carpal tunnel mass (e.g., a deep ganglion cyst), or stiffening and contracture of the transverse carpal ligament itself.
PATIENT EVALUATION
History and Physical Examination
Carpal tunnel syndrome accounts for approximately 463,673 carpal tunnel releases performed annually in the United States.11 Patients who have developed this syndrome usually present with a history of characteristic symptoms, such as nocturnal pain, paresthesias, and numbness in the median nerve distribution distal to the wrist, and thenar muscular weakness. The physician should be aware of the patient’s general health condition and family history. Congenital diseases or anomalies, connective tissue diseases, systemic and metabolic disorders, and previous injury to the distal forearm or wrist should be taken into consideration.
The physical examination is critical for accurate diagnosis. In an acute case, there is tenderness along the carpal canal area. Light percussion over the median nerve at the wrist produces a sensation of an electric current that radiates to the median nerve distribution; this is known as Tinel’s sign. Phalen’s sign is evoked by holding the wrists at maximum flexion with the dorsal aspects of the hands in full contact (i.e., reverse praying position). This position narrows the carpal canal; if it reproduces the paresthesias in the fingers within 60 seconds, the sign is considered positive. As the pathologic condition advances, less time is necessary to evoke a response. Other indicators include the monofilament test, two-point discrimination, the reverse Phalen’s test, and the tourniquet test. In the late stages, with thenar muscle atrophy, the examiner can observe the muscle wasting in the thenar area.12 Muscle weakness is tested subjectively by resisted palmar abduction of the thumb against the examiner’s index finger and comparison of one hand with the other.
Diagnostic Tests and Imaging
Electromyography and nerve conduction velocity tests aid in the detection of carpal tunnel syndrome. Indications for surgery should not be decided or altered according to the results of nerve conduction velocity tests, especially when the results are normal but the patient has the clinical signs and symptoms of the syndrome.13–15 A delay in the distal latency of the median nerve of 7.0 msec or longer represents significant compression of the median nerve; if such a delay is present, surgery should be considered immediately. The most important aspects in diagnosing carpal tunnel syndrome are the history and the physical examination results. Electrodiagnostic studies of the median nerve are used adjunctively to confirm the diagnosis and perhaps to suggest how the patient will respond to surgery.
Wrist radiography can rule out the possibility of congenital or acquired bone or joint deformity, abnormality, or pathology. Previously sustained fractures of the distal forearm and wrist should be taken into account. A malunited fracture of the distal radius, previously performed surgery in the wrist area, or a hypoplastic or aplastic hook of the hamate16 can produce difficulties for the surgeon during placement of and operation through the slotted cannula. Standard anteroposterior and lateral views of the distal forearm and wrist and a tunnel view of the wrist are required. If a more extensive study is indicated, magnetic resonance imaging (MRI), computed tomography (CT), ultrasound, bone scanning, or arthrography of the wrist may be necessary.17,18
TREATMENT
Open Versus Arthroscopic Management
The indications for the open surgical release of transverse carpal tunnel ligament have been well established, and in most cases they apply to ECTR. The advantages of endoscopic over open carpal tunnel release include no hypertrophic scar or scar tenderness, no pillar pain, less compromise of pinch or grip strength, and an earlier return to work and daily activities. However, ECTR can become a dangerous procedure if it is performed by an inexperienced surgeon.19–21 Significant intraoperative complications have been reported throughout the United States,22–24 raising questions about the value of ECTR. However, ECTR can be performed safely by experienced surgeons. Despite its steep learning curve, the results of ECTR can be satisfying for the patient and the surgeon.25 As the knowledge, technique, and instrumentation have evolved, the safety of ECTR has improved.
Arthroscopic Technique
The original technique was described by Chow as a transbursal approach to the carpal tunnel requiring penetration of the ulnar bursa.5,26 After a multicenter study,27,28 the original technique was modified in an attempt to decrease the complications and improve the learning curve. Conversion to an extrabursal technique made the surgical procedure much easier and safer because it offered better visualization of the proximal transverse carpal ligament.29–31 The following paragraphs describe the extrabursal, dual-portal technique.
Operating Room Setup
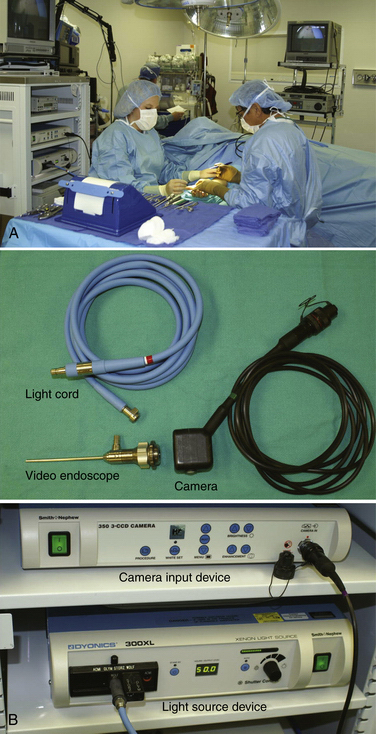
The patient is placed in a supine position, and a hand table is used. Two video monitors are preferred, although some surgeons can manage the procedure with only one. One of the two monitors should face the surgeon, and the other should face the assistant. The surgeon sits on the ulnar side of the patient, and the assistant faces the surgeon (Fig. 34-2A). The arthroscopic equipment consists of a short, 4.0-mm, 30-degree video endoscope. The light post is on the same side as the direction of view, the camera apparatus, the light cord, the camera input device, and the light source (see Fig. 34-2B). Optional equipment includes a digital video recorder and a video printer for the printing of any captured images. Water pump and shaver equipment are not used.
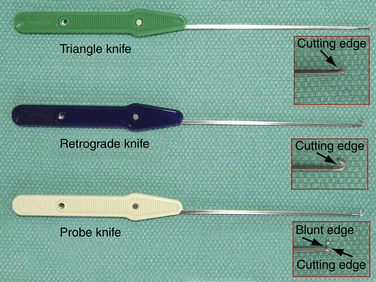
A standard handset should be available. Specific instrumentation for the procedure, designed by Chow, includes an ECTRA System Kit and an ECTRA Disposable Kit (Smith & Nephew Endoscopy, Andover, MA). The ECTRA System Kit includes a video endoscope, slotted cannula, dissecting obturator, curved blunt dissector, palmar arch suppressor, probe, retractors, and hand holder (Fig. 34-3). The dissecting obturator is attached with a detachable handle that can also take some other types of obturators included in the kit (e.g., conical, boat-nose obturator), but the latter are not used routinely. The ECTRA Disposable Kit includes a probe knife, triangle knife, retrograde knife, hand pad, and swabs (Fig. 34-4). These knives allow the surgeon to determine the direction and depth of the cut.

FIGURE 34-3 Instrumentation included in the ECTRA System Kit (Smith & Nephew Endoscopy, Andover, MA).
Standard preparations and draping are performed as usual, without the application of a tourniquet. Before local anesthesia is introduced, a skin marker is used to map landmarks for the entry and exit portals.
Anesthesia
Local anesthesia combined with intravenous medication is recommended for the procedure, because it allows the patient and the surgeon to communicate. An alert patient can inform the surgeon during the procedure about any abnormal sensation in the hand, which can indicate a potential problem caused by a variance of nerve structure in the wrist and palm region.32–38 Usually, when the patient first comes into the operating room, 100 μg of fentanyl citrate (Sublimaze, Baxter Healthcare Corporation, Westlake Village, CA) is given intravenously. This is a narcotic analgesic medication with an onset of 7 to 8 minutes and a peak action of approximately 30 minutes. Usually, the surgical time does not exceed 10 minutes. A 1% solution of Xylocaine (Astra, Westboro, MA) without epinephrine is injected, approximately 2 to 3 mL at the entry portal and 6 to 7 mL at the exit portal (because of the higher degree of sensitivity of the skin on the palmar region). Special care is taken to limit the injection to the skin and subcutaneous tissue and to avoid affecting the nerve by penetrating deeply.