CHAPTER 34 Emerging Technologies in Shoulder Surgery: Trends and Future Directions
In this chapter we give our personal views on some of the current areas of active research in shoulder surgery and offer a perspective of the future directions in each of these areas. We extend our apology to all our colleagues for areas of research that we do not mention in this chapter. We suspect that some of these unmentioned areas of innovation will undoubtedly make a significant impact in the future. When taking on the challenge of predicting the future we focused our comments to areas that, as active clinician investigators, we are working in and therefore have some ability to influence their future and direction.
BIOLOGICAL ENHANCEMENT OF ROTATOR CUFF REPAIR
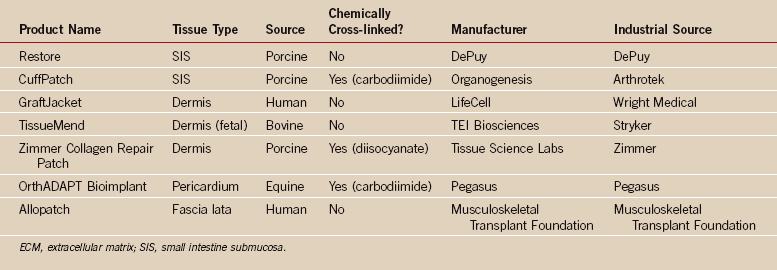
The use of a naturally occurring extracellular matrix (ECM) graft for augmentation of rotator cuff repair has received increased attention in the 21st century. It is a rapidly growing and evolving segment of the orthopaedic implant industry, with a large investment in resources devoted to further development of these materials.1–5 It is likely that these materials and their methods of use will be improved upon such that the clinical results will improve and their use will increase in the future.
Naturally occurring ECM grafts are obtained from either human or animal tissues and are processed in some fashion by the manufacturer to decrease their cellular content with the intent of decreasing the immune response when the graft is placed in the patient (Table 34-1). These tissues also vary by being untreated or treated with agents to cross-link their collagen proteins. Cross-linking also inhibits the immune response and significantly decreases the ability of the host to resorb the graft or incorporate and infiltrate the graft with host cells and tissue.1,4,6
The biological,1,4,6,7 biomechanical,1,4 and early clinical results of these graft materials8–17 are reported in the literature. The differences in biological response that have been defined in animal models suggest that these grafts have very different properties that would be expected to make them behave differently in human application.6 Similarly, these grafts each have different material properties4,5 some that are significantly different from the rotator cuff tendon. It is likely that these differing material and biological properties will influence the indications for their use and the methods of their use in the repair of the rotator cuff. Many of these issues have not been studied, and the widespread use of these grafts for rotator cuff repair is not supported by scientific evidence or significant clinical experience, specifically, randomized clinical trials that define the best indications and use of these products. Despite these less-than-positive early clinical results and the many important clinical questions that remain unanswered, there is ample justification to continue work in the development and clinical testing of these ECM grafts, given the clinical challenges of obtaining reliable primary healing of large chronic rotator cuff repairs in older patients.
Growth factors and stem cell recruitment are critically important in all wound healing as well as in normal tissue turnover. Supplementation of a rotator cuff repair with growth factors applied to the surgical site within a synthetic or resorbable carrier matrix delivered to the repair site by cells modified to produce the growth factor through viral transfection are areas under active investigation in vitro and in preclinical trials.3,11,18–28 In addition, tissue-engineered tendon-like graft materials including a combination of natural or synthetic matrices, differentiated or pluripotential stem cells, and growth factors in tissue culture environment are in various stages of development. The use of in vitro mechanical stimuli to induce a neotendon-like structure is being investigated in vitro and in preclinical animal studies.1,30,31 In some cases, these research strategies result in a better understanding of the factors associated with tendon healing, but in other cases these experiments are performed with the intent of using these products for transplantation.1,29 None of these strategies or products are commercially available, nor are there sufficient data for them to be used in human rotator cuff repair. In our view, it is unlikely that these strategies will be available for clinical use by 2020. It is also likely that these areas of research will, in the longer term, provide new methods to biologically enhance the repaired tendon and improve our clinical results and have a real future after 2020.
Platelets are a rich source of platelet-derived growth factor and transforming growth factor β; both factors are known to be up-regulated in early tendon injury and healing.27 In addition, exogenous use of these factors in in vitro culture and in preclinical trials have shown increases in cell proliferation and in neotendon matrix production.19,20,27 This basic science information, coupled with the ready availability of autologous platelet-rich plasma in the operating room, have spawned early clinical use of this product in rotator cuff repair. Clinical results using this technique are lacking at this time. Platelet gels used in association with ECM grafts are being used based upon the basic science data, and clinical results will need to be supported by randomized clinical trials to prove their clinical efficacy. Given the ease of use, it is likely that a strategy to increase the rate of healing will gain increased use in the near future if clinical efficacy can be demonstrated.
Muscle atrophy and fatty infiltration of the muscle is often associated with large chronic rotator cuff tears. Research has shown that these changes are correlated with changes in the material properties of the muscle and tendon, resulting in difficulty in mobilizing the tendon and increasing tension at the repair site.31–35 Clinical follow-up of patients with successful healing of these tendons demonstrates that these muscle changes are not reversible. This would suggest the need for biological or alternative mechanical measures to help reverse the muscle changes associated with these chronic tears. Studies of chronic tears in animal models have led to a greater understanding of the pathogenesis of these changes31 and most recently to some innovative methods to reverse these changes. It is likely that these innovative methods of treating muscle changes will be incorporated into our clinical practice by 2020.
COMPUTER NAVIGATION AND MINIMALLY INVASIVE SHOULDER REPLACEMENT
There has been a marked increase in the attention given to computer-assisted navigation and less-invasive surgical techniques in hip and knee replacement surgery.36–39 Based upon this trend it would be natural to think that these principles and techniques will spill over to shoulder replacement. It would be easy to predict that the techniques will be available in the near future because several research groups and companies are actively working on developing this area of technology. What is a bit more difficult to predict is the ultimate utility and level of general use of these techniques for shoulder surgery in the next 20 years.
The challenges to the widespread use and adoption of computer-assisted navigation in shoulder arthroplasty relate to the number of surgical cases performed in the United States and worldwide when compared to hip and knee arthroplasty. At present there are approximately 400,000 knee replacements and 200,000 hip replacements performed each year in the United States.40 This compares to approximately 30,000 shoulder replacements performed each year in the United States.40 Most orthopaedic surgeons in general practice perform dozens of hip and knee replacements each year, but these same surgeons generally perform fewer than five shoulder replacements each year. This presents an economic challenge for the orthopaedic-device industry to develop these technologies specifically for shoulder arthroplasty. It also presents a higher threshold for the lower-volume orthopaedic surgeon to adopt these techniques in the early years of development and clinical application of these technologies, particularly when their value and effectiveness have yet to be clearly defined.
Minimally invasive surgery for hip and knee arthroplasty was developed beginning in the 1990s, and the techniques and tools have been applied and proved to be effective in decreasing recovery time.41,42 It is clear for the hip and knee arthroplasty arena that minimally invasive techniques are here to stay and will increase in their general application. In the case of shoulder arthroplasty, the issues of case volume that stall the rapid development and adoption of computer-assisted navigation will also be a negative factor in the development and adoption of these same principles of minimally invasive surgery.
We need to determine the need for minimally invasive techniques and then determine what constitutes minimally invasive surgery for shoulder arthroplasty. The shoulder is typically entered between the pectoralis major and deltoid, and for open surgery this is not likely to be improved upon because morbidity is already minimal. The problems with healing of the subscapularis tenotomy are improved by using a lesser tuberosity osteotomy as the portal to the intra-articular space.43,44 With secure fixation, bone-to-bone healing may prove to be so reliable that the morbidity associated with the subscapularis portal may be minimized.43,44
We can now perform all of these arthroscopic techniques for removal of a loose glenoid, so it may be possible in the future to use them in minimally invasive total shoulder replacement. We predict that minimally invasive total shoulder replacement will be a reality for some surgeons by 2020, but it will take much longer for many surgeons performing a few total shoulder replacements each year to adopt these methods.
MANAGEMENT OF GLENOID BONE LOSS IN SHOULDER ARTHROPLASTY
Glenoid bone loss is relatively common in certain conditions requiring shoulder arthroplasty. These conditions include primary osteoarthritis, postcapsulorrhaphy arthropathy, rheumatoid arthritis, and revision arthroplasty. In many cases, this glenoid bone loss is not concentric. The results of humeral hemiarthroplasty are negatively correlated with the presence of eccentric glenoid bone loss.45 Total shoulder arthroplasty is more difficult, accompanied by a greater rate of complications, and it is less predictable in the presence of glenoid deficiency, especially when subluxation is present.46,47 Therefore, glenoid bone loss in shoulder arthroplasty is an important and unsolved problem.
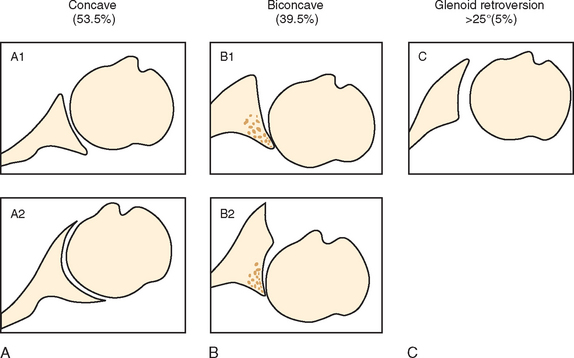
The most common indication for primary shoulder arthroplasty, excluding fractures, is osteoarthritis. Some degree of posterior glenoid bone deficiency has been noted in approximately 40% of osteoarthritic patients.48 Walch and colleagues categorize glenoid morphology in osteoarthritis into three types. Type A (59%) is characterized by concentric glenoid erosion; type B (32%) is characterized by posterior subluxation (B1) and posterior subluxation with asymmetric posterior erosion (B2), and type C (9%) is characterized by glenoid retroversion resulting from glenoid hypoplasia (Fig. 34-1).48
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree