Chapter 18 Autoimmune disease
The immune system is closely regulated to minimize the risk of self-antigens being recognized as foreign, leading to immune-mediated damage to an individual’s own cells and tissues. This regulation occurs by mechanisms that induce immune tolerance (Fig. 3.18.1). Suppressor CD8 T cells can also inhibit the immune response, in part by releasing inhibitory cytokines (e.g. IL-10). Autoimmune diseases develop when one or more of these protective mechanisms fails or is bypassed.
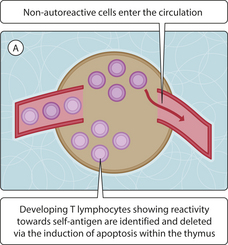
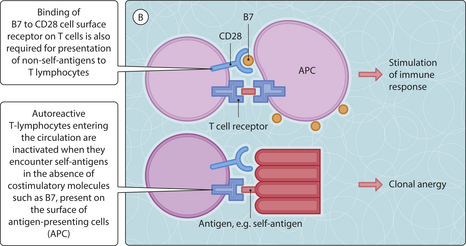
Fig. 3.18.1 Mechanisms for the induction of immune tolerance. (A) Clonal deletion of T-lymphocytes that recognize self-antigens (may also occur for B-cells but this is of less importance); (B) clonal anergy.
Mechanisms of autoimmune disease development
Infective agents
The immune response to some infective agents may be associated with polyclonal reactivation of circulating autoreactive lymphocytes that have previously been inactivated via the clonal inactivation mechanism (Fig. 3.18.1). This, in turn, can promote the development of autoimmune disease.
The role of immunogenetics
Correct functioning of the immune system is reliant upon complex interactions between immunoreactive cells and their targets. These interactions involve molecules present on the surfaces of cells, many of which are transmembrane in nature and, therefore, may also interact with intracytoplasmic components. Many of the genes encoding molecules such as the HLA family and cytokines (or their promoter regions) are highly polymorphic (meaning that one or more variants of the genes exist) and these polymorphisms may alter the detailed structure of the molecules (e.g. the antigen-binding groove of HLA molecules) or possibly the level of expression of molecules (e.g. cytokines). These changes can modulate immune responses, leading to interindividual differences in the response to antigens and the risk of development of forms of aberrant immune response such as those characterizing autoimmune disease. Individuals possessing certain HLA genetic polymorphisms are at increased risk of developing particular autoimmune diseases (Table 3.18.1).
Table 3.18.1 Associations between HLA alleles and autoimmune diseases
| Disease | Associated HLA locus/allele | Relative riska |
|---|---|---|
| Coeliac disease | DQ2 | 250 |
| Ankylosing spondylitis | B27 | > 150 |
| Reactive disease | B27 | > 40 |
| Type 1 diabetes mellitusb | DQ8 | 14 |
| DQ6 | 0.02 | |
| Multiple sclerosis | DR2, DQ6 | 12 |
| Rheumatoid arthritis | DR4 | 9 |
| Haemochromatosis | A3 | 6 |
| Addison’s disease | DR3 | 5 |
| Graves’ disease | DR3 | 4 |
| Myasthenia gravis | DR3 | 2 |
a Relative risk measures frequently of the disease in question in individuals with an HLA locus allele compared with those not carrying the allele.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






