Extremity vascular injuries continue to pose clinical challenges to trauma and vascular surgeons. More than three fourths of all vascular trauma occurs in the extremity.
1,
2,
3,
4,
5 Although most clinically significant peripheral vascular injuries result from penetrating trauma, the contribution to this cohort from blunt trauma may be higher depending on the injury mechanism and setting.
2,
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14 The accumulated experience with vascular trauma has seen considerable contributions from surgeons in combat settings, where most injuries result from penetrating trauma. Historically, many arterial injuries were managed with simple ligation followed by amputation. Current management yields high rates of successful vascular repair and significantly improved limb salvage rates compared to vessel ligation. In addition, the development of endovascular techniques will impact the future of surgical management for vascular trauma.
Patient Evaluation
The evaluation of trauma victims with vascular injury should follow the basic protocols of Advanced Trauma Life Support (ATLS). A thorough, focused vascular examination is paramount when an arterial injury is suspected. Palpation of the arterial pulse at all levels in the affected limb(s) should begin distally and move proximally, with note made of the quality, strength, and rate at each level. Throughout the examination, the injured limb should be compared to the contralateral limb. Occasionally, hard signs of arterial disruption will be encountered such as active arterial hemorrhage, expanding hematoma, limb ischemia, thrill, bruit, or absent pulses (see
Table 1). Of these, pulse deficit is reportedly the most common.
15 Although compression techniques-including placement
of tourniquets-can be used as temporizing measures for active arterial hemorrhage in the trauma bay, further management should be performed in the operating room. Radiographic or ultrasonographic assessment of a limb with an absent pulse, however, is obligatory if other issues do not require immediate attention in the multiply injured patient.
Evaluation of the affected extremity with a handheld Doppler ultrasound should be performed routinely in all patients with nonpalpable pulses. It is important to make note of the phase pattern of the waveforms. Absent Doppler signals warrant further imaging—either with angiography, duplex ultrasonography, or computed tomography with angiogram (CTA). Every Doppler examination, whether normal or abnormal, should be followed by the Ankle Brachial Index (ABI) measurement. An ABI >1.0 is considered normal. Any measurement <0.9 warrants further evaluation, usually with angiography. Again, it is important to compare findings between limbs.
Combined with ABI, the physical examination (PE) has proved to be an accurate measure of arterial injury.
8,
15,
16,
17,
18,
19 A high negative predictive value has been well documented when the PE is used as a screening test in the trauma setting, with fewer than 1.5% of occult vascular injuries ultimately requiring operative intervention.
16 The robust sensitivity of screening with PE and ABI has been validated by multiple authors for both penetrating and blunt injury. Gonzalez et al.
15 examined the utility of PE for the evaluation of penetrating extremity trauma in 406 patients with 489 vascular injuries. The authors demonstrated a sensitivity of 92% and a specificity of 95%. Their screening protocol included PE for hard signs of vascular injury, assessment of injury trajectory, and Doppler ultrasound. Those patients without hard signs were observed and those with hard signs were either taken directly to the operating room or for angiography. There were four missed injuries, for a negative predictive value of 99%. Miranda et al.
20 reported a positive predictive value of 94.3% and a negative predictive value of 100% for PE in a retrospective review of their institution’s protocol for managing posterior knee dislocations. Thirty-five patients with knee dislocations were either taken emergently to the operating room (8 patients, 23%) because of hard signs of vascular injury, or they were observed with serial examinations (27 patients, 77%). In the latter group, no vascular injuries were diagnosed during hospitalization, nor at a mean follow-up of 13 months (12 patients). Long-term studies have also validated the nonoperative management of occult vascular injuries. In a longitudinal study of patients with penetrating extremity trauma, Dennis et al.,
16 demonstrated that PE alone can be safely used to triage this patient population. The mean follow-up for patients in the group who underwent evaluation only with PE was 5.4 years, and none of the patients surveyed required vascular intervention and none reported symptoms related to vascular insufficiency.
Pathophysiology
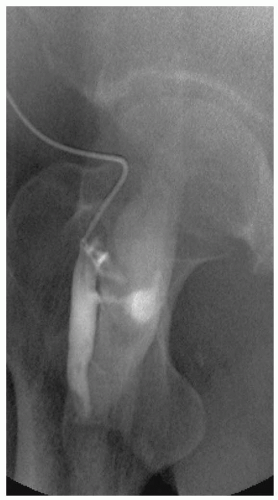
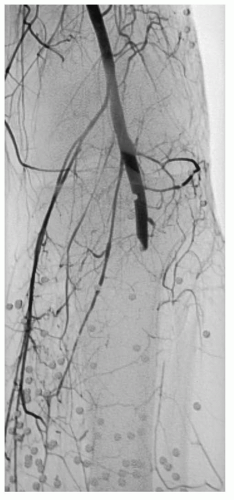
The arterial wall is composed of three anatomic layers-the intima, media, and adventia-and vascular injury can affect one or all vessel layers. Blunt force trauma seen with crush injury can lead to vessel contusion. Hematoma formation occurs between the intima and media or the media and adventia and cause compression and partial or complete vessel occlusion. Disruption between the intima and media can also lead to an intimal flap with subsequent dissection and thrombosis. Whenever the intimal layer is disrupted, the highly thrombogenic internal elastic lamina is exposed. When the inner two layers are disrupted but the adventitia is intact, a pseudoaneurysm forms outside the vessel lumen (see
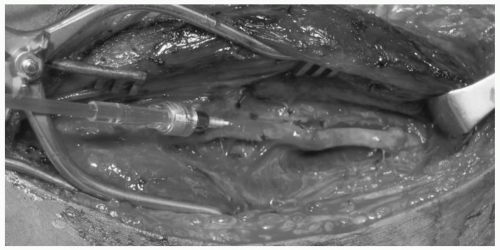
Fig. 1). Stretch injury to the vessel causes disruption between the layers due to shearing forces. These injuries often occur in the setting of posterior knee dislocation with resulting occlusion of the popliteal artery. Vessel lacerations or complete transections are typically the result of penetrating injury from missiles or other projectiles, and less commonly from fractured bone (see
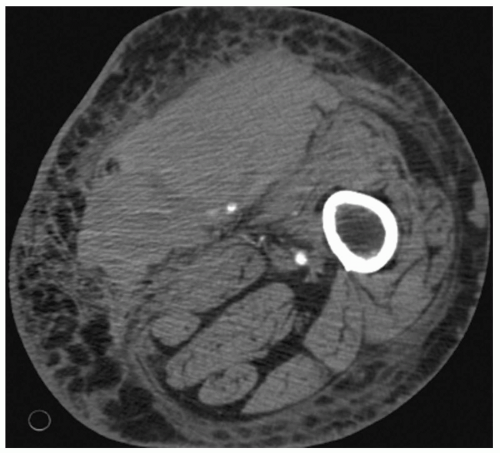
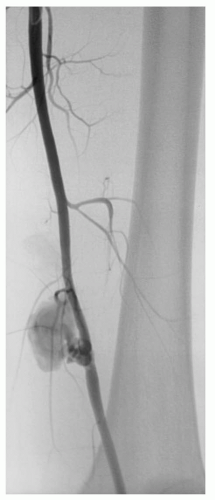
Fig. 4). Arteriovenous fistulae (AVFs) commonly result when concomitant arterial and venous injuries occur in close proximity (see
Figs. 2 and
6).
Etiology, Injury Mechanism, and Location
The etiology of vascular trauma often depends on both the setting in which injuries are reported and the particular vessel injured. For instance, in the civilian setting at an urban trauma center, penetrating injury accounted for 81% of approximately 360 vascular injuries at the Ben Taub General Hospital in Houston, Texas, from 1981 to 1985.
2 Likewise,
recent wartime experience demonstrates that approximately 94% of battlefield vascular injuries (of which 78% were extremity injuries) were the result of penetrating trauma.
1 In contrast, injury to the popliteal artery is the result of blunt mechanisms in most reported cases.
12,
21,
22 In their review of the National Trauma Data Bank to evaluate outcomes from popliteal artery injury, Mullenix et al.
22 noted that 61% of the injuries were the result of blunt mechanisms, with only 39% occurring from penetrating injury. Likewise, Hossny et al.
12 reported that of 39 popliteal artery injuries occurring over a 9-year period, 31 (79%) were the result of blunt trauma.
Penetrating injury most often occurs from gunshot or stab wounds in the civilian setting, and from fragment wounds in combat settings.
1,
8,
11 Surgeons in combat settings also routinely encounter blast injury from improvised explosive devices (IEDs).
1 Blunt injury most often results from motor vehicle collisions (MVCs), falls, and crush injury in both civilian and battlefield environments.
Lower extremity vascular injuries are defined as those occurring below the inguinal ligament. The most commonly injured vessel is the femoral artery.
6,
11 Injuries to the common femoral artery (CFA), profunda femoris, superficial femoral artery (SFA), and popliteal artery both above and below the knee represent the bulk of clinically significant vascular trauma requiring revascularization procedures or surgical exploration (see
Table 2). Because there is more redundancy in the blood supply to the calf and foot, with anterior tibial, posterior tibial, and peroneal arteries all supplying this distribution, one or sometimes two of these vessels can be disrupted without loss of distal perfusion and limb ischemia. The one
caveat to this point is that significant bleeding from any of these vessels or their branches can lead to elevated compartment pressures in the calf and subsequent compartment syndrome. Trauma and vascular surgeons should maintain a high index of suspicion for the compartment syndrome and intervene with fasciotomy as necessary, as discussed later in this chapter.
Vascular injury to the upper extremity is relatively common, representing 30% to 40% of vascular injuries in urban civilian settings.
23,
24 For the purposes of this discussion, the vessels of the upper extremity are defined as coursing from the axilla to the wrist. Injuries to the subclavian vessels are commonly reported together with those of the axillary vessels, and surgical management and associated injuries are similar.
10,
25,
26 Therefore, axillosubclavian vessel injuries are discussed here as well. The most commonly injured vessel in the upper extremity is the brachial artery with an incidence of approximately 50%, followed by the radial and ulnar arteries (˜25% each), with the least common being the axillary and subclavian arteries (˜3% to 5%).
23,
24 The most common mechanism for vascular injury in the upper extremity is penetrating trauma.
10,
23,
24,
25Iatrogenic injuries account for an increasing number of peripheral vascular lesions.
27 The rise in these injuries has come with the growth of percutaneous therapies for coronary and peripheral arterial disease processes. Although many of these injuries can be managed by noninvasive methods (compression and/or fibrin injection under ultrasonographic guidance, endovascular stent grafts, etc.), some require more extensive management. Although a full discussion of the incidence, etiology, prevention, and management of these injuries is beyond the scope of this chapter, they remain an important diagnostic and treatment challenge for vascular surgeons.