Chapter 5 Biology of Anterior Cruciate Ligament Graft Healing
INTRODUCTION
Anterior cruciate ligament (ACL) tears are common among athletes and may become functionally disabling knee injuries. Reconstruction of a torn ACL in order to restore function and limit injury to the menisci has become a common orthopaedic procedure. Despite advances in surgical techniques and the ability to implant an anatomic, isometric graft, ACL reconstruction is not a universally successful procedure. Rates of recurrent laxity 1 year postoperatively have been reported to be as high as 17%.78
Failure of graft integration and tendon-to-bone healing may be an important cause of recurrent laxity. The healing of tendon to bone is the basic requirement for the long-term survival of the graft.12,24 Whether an autograft or an allograft tendon is used, biomechanical testing has shown that the initial strength of the graft material is superior to that of the intact ACL.12,24 Therefore, the weakest link after reconstruction is not the graft itself, but rather the fixation points until graft osteointegration occurs. The intra-articular portion of the graft must also undergo remodeling and a process of “ligamentization” to form a structure that resembles a native ligament.3,5,28,36,38
Current techniques of ACL reconstruction require tendon-to-bone healing in a surgically created bone tunnel. There are no native sites in humans at which a tendon passes through a bone tunnel and, therefore, no analogous situation to the healing that is required after reconstruction. When a bone-tendon-bone graft is used for ACL reconstruction, graft fixation initially depends on bone-to-bone healing. However, tendon-to-bone healing still remains critical regardless of whether a soft tissue graft or a bone-tendon-bone graft is selected. The length of the tendinous portion of the bone-tendon-bone graft is greater than the intra-articular length of the native ACL, resulting in substantial tendon in the bone tunnel. Aperture fixation to minimize graft micromotion and tunnel widening requires tendon-to-bone healing with any graft.38
NATIVE TENDON-BONE INSERTION
Critical Points NATIVE ACL INSERTION
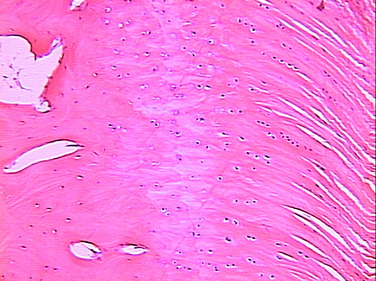
neural elements.4 The ACL inserts to bone via a direct type of insertion, similar to the transition seen from tendon to bone. Microscopic examination of the sites of bony attachment show interdigitation of the collagen fibers with bone through four distinct transition zones: tendon, unmineralized fibrocartilage, mineralized fibrocartilage, and bone (Fig. 5-1).4,17,57,70 This graduated change in stiffness allows for transmission of complex mechanical loads from soft tissue to bone while minimizing peak stresses at any single point along the ligament. Cartilage-specific collagens including types II, IX, X, and XI are found in the fibrocartilage insertion site. Collagen X plays a key role in maintaining the interface between the mineralized and the unmineralized zones.4,17,57,70
TENDON-BONE INSERTION AFTER ACL RECONSTRUCTION
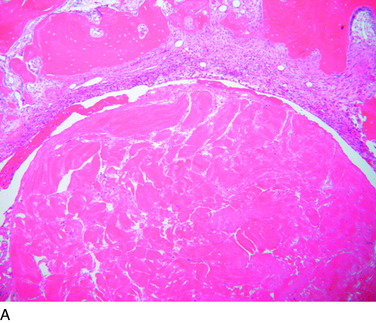
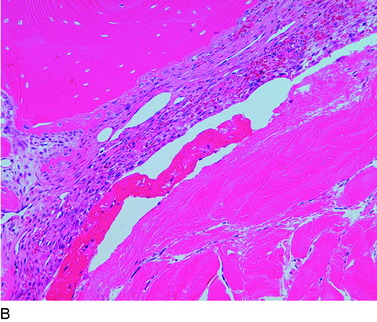
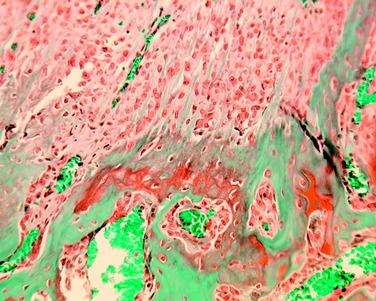
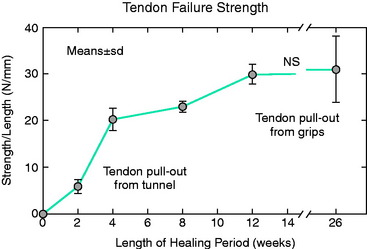
The overall structure, composition, and organization of a native ACL insertion site are not reproduced after ACL reconstruction and reflect an inability to recapitulate the events that occur during embryonic development with current surgical techniques. Rather than regenerating the four organized zones of direct insertion, the graft heals with an interposed zone of vascular, highly cellular granulation tissue between the graft and the tunnel wall (Fig. 5-2).20,21,65 After 3 to 4 weeks, this interface tissue undergoes a maturation process until its matrix consists of oriented, Sharpey-like collagen fibers that bridge the bone to the graft (Fig. 5-3). The number and size of these collagen fibers positively correlate with the pull-out strength of the graft (Fig. 5-4). Graft attachment strength further improves as bone grows into the interface tissue and outer portion of the graft.20,21,65
The maturation process of a tendon graft in a bone tunnel was defined by Kanazawa and coworkers in a rabbit ACL model.34 In the initial postoperative period, the graft-tunnel interface is filled with vascular granulation tissue containing type III collagen. Vascular endothelial growth factor (VEGF) and
Critical Points TENDON-BONE INSERTION AFTER ACL RECONSTRUCTION
fibroblast growth factor (FGF) are expressed, stimulating an influx of macrophages and enlarged fibroblasts. Chondroid cells accumulate along the walls of the bone tunnel and deposit type II collagen. The granulation tissue layer is degraded and becomes indistinct. The chondroid cells are gradually replaced with lamellar bone in a process similar to endochondral ossification.34 The Sharpey-like fibers are composed of type III collagen and extend into the surrounding bone to resist shear stresses. The time interval for this process has been variably reported in the literature, ranging from 8 to 30 weeks.34
CHALLENGES OF TENDON-BONE HEALING AFTER ACL RECONSTRUCTION
The biology of healing between a grafted tendon and a bone tunnel remains incompletely understood. The biologic and biomechanical environments result in the formation of an inferior attachment site that is different from the organized, direct-type ACL insertion. Current work suggests a number of fundamental challenges that are responsible for the suboptimal healing response between tendon and bone instead of regeneration of the insertion site.23 These factors include
MODULATION OF TENDON-BONE HEALING
Technical Factors
Adjustments can be made in the surgical technique of reconstruction to optimize tendon-to-bone healing. The fundamental principle is to maximize the surface area of the tendon-bone interface. Animal studies have shown that increasing the length of the bone tunnel positively correlates with the quality and strength of the reconstruction.80 Minimizing graft tunnel mismatch by achieving as tight a fit as possible also improves healing.22 In addition, maximizing circumferential contact area of the graft and tunnel (i.e., avoiding use of an interference screw) may improve healing.73
Critical Points MODULATION OF TENDON-BONE HEALING
Mechanical Factors
Relative graft-tunnel micromotion may preclude the formation of a firm attachment to the tunnel wall. Yu and Paessler83 compared “aggressive” versus “conservative” rehabilitation protocols in patients after ACL reconstruction with quadrupled hamstring grafts. Significantly greater tunnel widening was reported in the aggressive rehabilitation group, supporting the view that graft-tunnel micromotion is a cause of tunnel widening and that the mechanical environment in the bone tunnel influences tendon-bone healing.
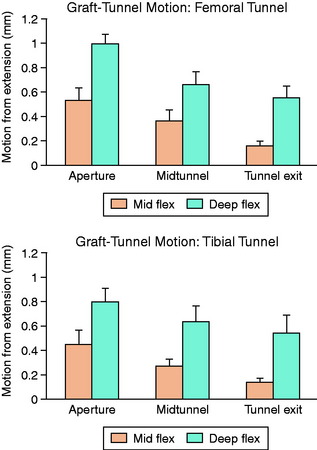
Animal studies have corroborated these clinical observations. Sakai and associates71 compared immediate motion with varying periods of immobilization for up to 6 weeks after ACL reconstruction in a rabbit model. Biomechanical analysis demonstrated a greater load-to-failure of the graft in immobilized animals, and histologic studies demonstrated closer tendon-bone apposition with less fibrovascular interface scar tissue. Rodeo and colleagues66 evaluated the effect of graft-tunnel motion on tendon-to-bone healing in a rabbit ACL reconstruction model. ACL reconstruction was performed in five cadaveric rabbit limbs with aluminum beads fixed to the tendon and bone tunnel. Three-dimensional graft-tunnel motion was quantified using micro–computed tomography (CT) analysis. Motion was observed to be greatest at the tunnel apertures and least at the tunnel exits, adjacent to the graft fixation at the tunnel exit (Fig. 5-5). Histomorphometric analysis demonstrated healing to be slowest at the apertures, with a wider fibrovascular interface tissue present between tendon and bone. An inverse relationship between graft-tunnel motion and healing in the femoral tunnel was demonstrated. In addition, osteoclasts were preferentially observed at the tunnel aperture, supporting the hypothesis that graft-tunnel motion may stimulate osteoclast-mediated bone resorption and secondary tunnel widening.66
Biologic Factors
Modulation of the Inflammatory Response
Immediately after ACL reconstruction and release of the tourniquet, the knee is filled with blood from the drilled bone tunnels. This initiates an acute inflammatory response marked by influx of neutrophils, macrophages, and mesenchymal cells. Important lessons about the effect of this inflammatory response on tissue healing can be learned from the study of fetal wound healing. Wounds in the embryo and early fetus heal by tissue regeneration and result in “scarless healing.” These healing events are characterized by the absence of an acute inflammatory response. Inflammation after trauma, although essential for healing in adults, is also responsible for healing by scar rather than regeneration of native tissue. The rapid inflammatory response that ensues after ACL reconstruction may trigger a cascade of events that ultimately leads to fibrosis rather than tissue regeneration and an anatomic tendon-to-bone insertion.19,39,72
The fibrin clot that forms after surgery allows for a controlled release of cytokines that drive the early response. Transforming growth factor-β (TGF-β) and platelet derived growth factor (PDGF) act together to modulate tissue healing and matrix deposition by recruiting neutrophils and macrophages to the local tissue.19,39,72 Neutrophil influx peaks at about 2 days after the operation and is followed by an influx of macrophages. These monocytes are essential to the early formation of granulation tissue from the clot, initiating the process of soft tissue adherence to bone.35 They also drive the angiogenic phase, providing nutrients and oxygen for the prolonged period of matrix synthesis and remodeling that ensues over the next several weeks. TGF-β secreted by macrophages recruits and stimulates fibroblasts to degrade matrix through matrix metalloproteinases (MMPs). Simultaneously, these fibroblasts synthesize new matrix proteins to replace the early granulation tissue with scar tissue. Tissue inhibitors of matrix metalloproteinases (TIMPs) inhibit MMPs and provide a check-and-balance regulation to this complex process of matrix degradation, synthesis, and remodeling.18
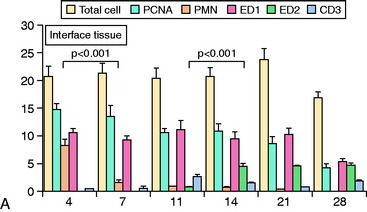
The accumulation of macrophages around the tendon graft in bone has been further characterized in a rat ACL reconstruction model.35 Two distinct subpopulations of macrophages were identified at the tendon-bone interface. ED1+ macrophages derived from the circulation act in a proinflammatory fashion in the early response with neutrophils to migrate into the tissues and remove debris after surgery. This is followed by the accumulation of proregenerative ED2+ macrophages from the local tissues, which promote anabolic tissue healing and scar formation by fibroblasts via TGF-β secretion (Fig. 5-6).35
Given the critical role of macrophages in scar tissue formation, animal studies have investigated the role of macrophage depletion on tendon-bone healing. It has been theorized that depletion may promote formation of a “scarless,” native insertion site. Hays and coworkers26 in the authors’ laboratory demonstrated less scar tissue formation, improved collagen organization, and superior biomechanical strength in a rat ACL reconstruction model when macrophages were depleted by administration of liposomal clodronate, a selective inducer of macrophage apoptosis. Other studies have identified gastric pentadecapeptide BPC 157, a novel anti-inflammatory used in the treatment of inflammatory bowel disease, to improve Achilles tendon-to-surface bone healing and tissue regeneration in a rat Achilles tendon model.37
The impact of inflammatory response suppression, however, has not been uniformly favorable for tendon-bone healing. Cohen and associates11 evaluated the effect of the anti-inflammatory medications indomethacin and celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, on tendon-bone healing in a rat rotator cuff model. The treated group was found to have histologically and biomechanically inferior tendon-bone interfaces at both 4 and 8 weeks relative to controls (Fig. 5-7).
Modulation of Bone Ingrowth
Bone ingrowth plays a critical role in the later stages of tendon-bone healing after ACL reconstruction and is ultimately responsible for the improved biomechanical properties after healing is complete. Animal studies have provided strong evidence to support that increased bone ingrowth positively correlates with graft-bone fixation strength.67 The primary strategies that have been employed include use of osteoinductive agents, osteoconductive agents, and modulation of osteoclast activity.
Osteoinductive agents, particularly from the bone morphogenic protein (BMP) family, have been shown to promote ingrowth of bone from the tunnel into the soft tissue graft. Rodeo and coworkers67 delivered recombinant human BMP-2 (rhBMP-2) on an absorbable type I collagen sponge in a tendon-bone canine model. At all time points, the rhBMP-2–treated limbs healed with increased bone formation around the tendon. Biomechanical testing demonstrated higher tendon pull-out strength in the treated limbs relative to controls at 2 weeks. Ma and colleagues41 delivered rhBMP-2 in an injectable calcium phosphate matrix to the bone tunnel in a rabbit ACL reconstruction model. Histologic analysis revealed a dose-dependent increase in bone formation at the tendon-bone interface and significantly narrower tunnel diameters (15%–45%) relative to the control group. Increased construct stiffness was also seen in the treatment group at 8 weeks postoperatively.41 Martinek and associates44 transected semitendinosus grafts in vitro with adenovirus-BMP-2 (Ad-BMP-2) and compared them with untreated controls in a rabbit ACL reconstruction model. These investigators demonstrated the formation of a fibrocartilaginous interface at the tendon-bone junction in the experimental group that was absent in the controls. Both stiffness and load-to-failure parameters were superior in the treatment group at 8 weeks. BMP-7 has also been shown to improve bone formation at the tendon-bone interface and load to failure at both 3 and 6 weeks postoperatively in an ovine reconstruction model.1,45
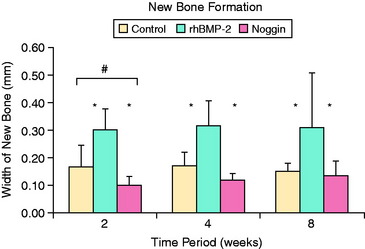
Further support for the role of BMPs in bone ingrowth around tendon graft comes from studies of BMP inhibition. Ma and colleagues41 delivered noggin, a potent inhibitor of all BMP activity, to the healing tendon-bone interface using an injectable calcium phosphate matrix in a rabbit ACL reconstruction model. Noggin significantly inhibited new bone formation at the tendon-bone interface. Furthermore, a significant increase in the width of the fibrous tissue interface between tendon and bone was found in the noggin-treated animals (Fig. 5-8).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree