CHAPTER 15 Arthroscopic Osteochondral Transplantation
Techniques for marrow stimulation, such as abrasion arthroplasty, drilling, and microfracture, produce fibrocartilage rather than native hyaline cartilage. Good short- and midterm results have been reported by various authors. However, inconsistent long-term results have perpetuated the interest in finding alternative methods for treating full-thickness chondral lesions. These include autologous chondrocyte implantation, autologous osteochondral transplantation, fresh osteochondral allografting, and bulk allografting, as well as a whole host of synthetics, scaffolds and first-, second-, and third-generation cell-based technologies.1–4 This chapter will focus on the COR system (DePuy Mitek; Raynham, Mass) for arthroscopic osteochondral transplantation.
ANATOMY
In adults, hyaline cartilage normally has a thickness of roughly 8 to 9 mm. As a person ages, the articular cartilage thins. The presence of pathology can accelerate this eroding process. To produce symptoms, articular cartilage must be thinned by more than 75% before subchondral nerve fibers can detect the change in contact surface pressure. However, overall size of the lesion also plays a role in this. A small chondral defect will not produce symptoms because the stress is distributed to the surrounding joint surface. Only when the cartilage defect becomes large enough, and thin enough will the patient become symptomatic.
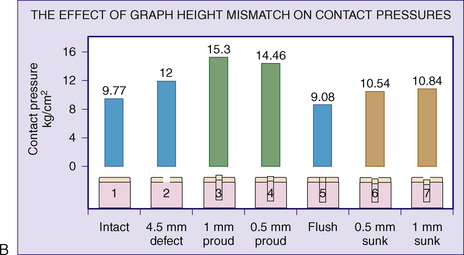
To illustrate this point, Jason and Koh5 have examined the contact pressures across an articular surface with respect to a focal defect and a grafted defect. Various graft height mismatches were modeled (Fig. 15-1), with the results shown in Table 15-1. Not surprisingly, plugs that were flush to their articular surfaces most closely resembled the contact pressure of the normal intact articular surface. However, plugs countersunk by 0.5 to 1.0 mm also closely mimicked the native pressures. Thus, filling a defect to a near-congruent articular surface can reproduce native articular-surfaced pressures. This is the foundation of autogenous osteochondral transplantation.
TABLE 15-1 Effect of Graft Height Mismatch on Contact Pressures
| Condition of Intra-articular Joint Surface | Contact Surface Pressure (kg/cm2) |
|---|---|
| Normal intact surface | 9.77 |
| Open 4.5-mm hole | 12.00 |
| Plug flush to the surface | 9.08 |
| Plug countersunk 0.5 mm to surface | 10.54 |
| Plug countersunk 1.0 mm to surface | 10.84 |
| Plug 0.5 mm proud to the surface | 14.46 |
| Plug 1.0 mm proud to the surface | 15.30 |
Because small chondral defects will not produce symptoms, the goal of osteochondral transplantation is to decrease the size of large defects functionally to become less clinically significant.
PATIENT EVALUATION
History and Physical Examination
A history of an acute injury with a subsequent hemarthrosis can be found in focal chondral and osteochondral injuries. This has been reported to have as high as a 20% incidence in patients with no ligamentous instability. Often, patients can present with mechanical symptoms of locking, catching, pain, and recurrent effusions. Ligamentous and meniscal injury can present additional symptoms and challenges.6–8