Chapter 11 Anterior Cruciate Ligament Reconstruction in Skeletally Immature Patients
INTRODUCTION
Prior to the mid-1980s, mid-substance anterior cruciate ligament (ACL) injuries in the skeletally immature athlete were believed to be rare.25 More recently, ACL injury has been reported in 10% to 65% of pediatric knees with acute hemarthroses.19,42 Although the true incidence and prevalence of ACL tears in the pediatric population have not been established, these injuries are now being recognized with increasing frequency.42,44 Explanations for this apparent change may include improved ability to diagnose injury by physical examination and magnetic resonance imaging (MRI) as well as changes in the activity patterns of young athletes. The puzzling question facing clinicians centers on whether youngsters of today are training and conditioning to the point that the ACL has become more susceptible to injury.
Previously, the concern for iatrogenic growth disturbance in children prevented the routine use of anatomic ACL reconstruction that has proved successful in adults. Alternative surgical treatment options include primary repair, extra-articular reconstruction, and transepiphyseal intra-articular reconstruction. Even though these injuries are occurring with increasing frequency, no unanimity exists concerning the appropriate and safest treatment. There are no long-term studies to help determine the optimal management approach. The few studies that have followed skeletally immature patients to physeal closure after transphyseal ACL reconstruction have documented growth comparable with that of age-matched controls without surgery.1,27,39
INDICATIONS
Graf and associates11 reported poor results at 15 months after injury, with new meniscal tears and episodes of instability in seven of eight skeletally immature patients who did not undergo reconstruction or activity limitations after ACL injury. Similarly, in a series of 18 skeletally immature patients examined an average of 51 months after complete ACL tear, Mizuta and colleagues35 found that all patients had symptoms, 6 had meniscal tears, and 11 had developed radiographic changes. Janarv and coworkers16 found that 10 of 23 skeletally immature patients treated with rehabilitation eventually needed reconstruction. McCarroll and associates29 reported superior results with surgical management of complete ACL tears in prepubescent and junior high school patients compared with patients receiving conservative treatment. Of 16 prepubescent patients treated nonoperatively, 9 ceased sports participation, 4 sustained at least one reinjury, and only 3 were able to return to sport. In a separate group of 75 junior high school athletes with mid-substance tears, McCarroll and colleagues30 reported that 37 of 38 patients who were initially treated nonoperatively had instability and 27 (71%) developed meniscal tears. Overall, 92% (55 of 60) of those treated with reconstruction returned to play.
CONTRAINDICATIONS
In the pubescent patient in whom a final growth spurt is anticipated, a soft tissue graft can be used. The exact risk of growth plate injury is unknown but can be extrapolated from the literature on trauma and fractures involving the growth plates around the knee.8 If parents are unwilling to accept the risk of growth plate injury, reconstruction should be delayed until the physes are closed.
CLINICAL EVALUATION
History
A tear of the ACL can result from an impact to the knee or, more commonly, a noncontact twisting or landing injury. The timing of injury, either acute or chronic, should be noted. Knee effusion, pain, and ability to bear weight are important factors in diagnosing an ACL tear. An audible pop heard at the time of injury may be reported. An acute hemarthrosis, particularly in the first 6 to 12 hours after injury, should raise concern for ACL injury. Stanitski and coworkers42 found that 47% of preadolescents with a knee effusion had an ACL injury.
Physical Examination
The most important component in diagnosing pediatric knee injuries is the physical examination.41 The presence of an effusion should be noted. Palpation of the joint line and physeal plates should be performed. Active and passive motion should be documented. Varus and valgus laxity should be checked in full extension and at 30° of flexion. Lachman testing should be assessed for magnitude. Pivot shifting may be quite uncomfortable in the acute setting and is not recommended. If the Lachman examination is positive acutely, the pivot shift test can be deferred until the examination under anesthesia if surgical intervention is chosen. Findings should always be compared with those in the contralateral knee. The patient should be assessed for generalized laxity with the degree of knee hyperextension noted.
PREOPERATIVE PLANNING
Skeletal Maturity
The patient’s level of skeletal maturity should be defined preoperatively. It is generally assumed that girls grow until age 14 ± 1 years and boys until age 16 ± 1 years2; however, age may not be the best indicator of potential growth. Peak growth velocity occurs between the ages of 10 to 11 years for girls and 13 to 14 years for boys. Closure of the triradiate cartilage, which can be seen on a standard radiograph of the pelvis, typically marks the end of peak growth velocity.26 The patient can be assessed physiologically for signs of development, as noted by Tanner and Davies.43 Age at onset of menses can be useful in females. After menarche, girls enter a deceleration phase of growth and typically reach skeletal maturity 18 months later. Family height can be used as an approximate estimate of growth potential as well. The patient can be assessed radiographically to determine skeletal maturity using the Risser sign on the pelvis.37 The Risser sign is a radiographic measurement based on ossification of the iliac apophysis, beginning on the lateral aspect and progressing medially. Divided into four quadrants, the Risser sign proceeds from 0 (no ossification) to Risser 4, in which all four quadrants show ossification of the apophysis. Patients with Risser 0 or 1 have a significant amount of growth remaining, and the Risser 4 patient is skeletally mature. Bone age is another method to determine skeletal maturity by obtaining a radiograph of the hand and wrist and comparing it with the standards in the Gruelich and Pyle atlas.12 The “rule of thirds” suggests that the distal femur and proximal tibia grow an average of 0.9 cm and 0.6 cm, respectively, per year of growth remaining.32 Limb lengths and bilateral lower extremity alignment should be measured and any differences noted.
Associated Intra-articular Pathology
Previous studies have shown that 20% to 100% of pediatric patients who sustain ACL injuries have a concomitant meniscal injury.11,34 Millett and associates34 found that the incidence of medial meniscus tears increases significantly with chronic ACL insufficiency. Of the 22 patients who underwent surgery more than 6 weeks after injury, 72% had associated meniscal tears. If unstable meniscus tears exist, aggressive management is indicated. Every attempt should be made to salvage the meniscus in children and adolescents. Meniscectomy in this age group carries an even more ominous prognosis.18 Manzione and colleagues28 evaluated the results of partial and total meniscectomy in 20 patients with a mean age of 15 years (range, 5–15 yr). At 6-year follow-up, 16 knees had grade I osteoarthritis and 4 had grade II or III changes. Osteochondral injuries may require débridement, drilling, and/or stabilization at the time of ACL reconstruction.
OPERATIVE TECHNIQUES AND CLINICAL OUTCOMES
Primary Repair
DeLee and Curtis7 reported on three patients who underwent primary ACL repair through sutures tied across the physis. At 21 months follow-up, all had clinical laxity and two of the three had episodes of giving-way. Engebretsen and coworkers10 reviewed eight patients 3 to 8 years after primary repair and found that all experienced a decrease in activity level and five of the eight demonstrated instability on clinical examination. Grontvedt and associates13 also noted that primary repair of the ACL in the skeletally immature patient had poor results, similar to those found in adults.
Physeal-Sparing Techniques
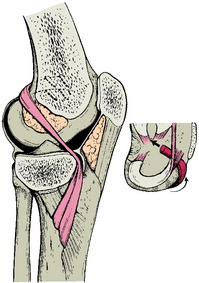
Early reports of physeal-sparing intra-articular reconstruction describe using a hamstring autograft left attached distally, passed into the knee under the anterior portion of the medial meniscus, and fixed to the lateral femoral condyle with staples in the over- the-top position on the femur6 (Fig. 11-1). This technique is nonanatomic and does not reproduce the normal knee ligament kinematics. In a report of nine patients at 36.5 years follow-up, all had a 1+ Lachman and 1+ anterior drawer test. Six of nine patients returned to sports, but with bracing and precautions. No growth disturbances were reported.6
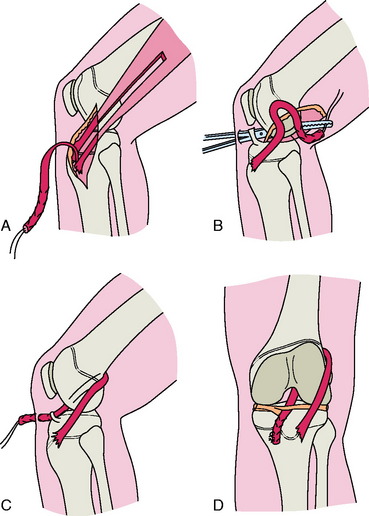
More recently, Kocher and colleagues21 described Micheli and coworkers’ technique,33 which uses a combined intra- and extra-articular reconstruction and an autogenous iliotibial band (ITB) without violation of the physes. An incision is made obliquely from the lateral joint line to the superior border of the ITB. The ITB graft is harvested free proximally and left attached to Gerdy’s tubercle distally and tubularized with a whipstitch. Arthroscopy of the knee is then performed through standard portals. The free end of the graft is brought into the knee in the over-the-top position posteriorly. A second incision is made over the proximal medial tibia. Dissection is carried down to the periosteum, and a curved clamp is passed from this incision into the joint under the intermeniscal ligament. The graft is then brought through the knee, under the intermeniscal ligament anteriorly, and out through the tibial incision. The graft is fixed to the femur with the use of sutures to the lateral femoral condyle at the insertion of the lateral intermuscular septum with the knee in 90° of flexion and external rotation. On the tibial side, the graft is sutured to the periosteum of the proximal tibia just distal to the physis with the knee flexed 20°, and tension is applied to the graft (Fig. 11-2).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree